Abstract
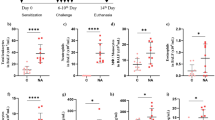
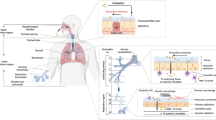
Inhalation of commonly present irritants, such as chlorine and chlorine derivatives, can cause adverse respiratory effects, including irritant-induced asthma (IIA). We hypothesize that due to airway barrier impairment, exposure to hypochlorite (ClO-) can result in airway hypersensitivity. C57Bl/6 mice received an intra-peritoneal (i.p.) injection of the airway damaging agent naphthalene (NA, 200 mg/kg body weight) or vehicle (mineral oil, MO). In vivo micro-computed tomography (CT) images of the lungs were acquired before and at regular time points after the i.p. treatment. After a recovery period of 14 days an intranasal (i.n.) challenge with 0.003% active chlorine (in ClO-) or vehicle (distilled water, H2O) was given, followed by assessment of the breathing frequency. One day later, pulmonary function, along with pulmonary inflammation was determined. Lung permeability was assessed by means of total broncho-alveolar lavage (BAL) protein content and plasma surfactant protein (SP)-D levels. In vivo micro-CT imaging revealed enlargement of the lungs and airways early after NA treatment, with a return to normal at day 14. When challenged i.n. with ClO-, NA-pretreated mice immediately responded with a sensory irritant response. Twenty-four hours later, NA/ClO- mice showed airway hyperreactivity (AHR), accompanied by a neutrophilic and eosinophilic inflammation. NA administration followed by ClO- induced airway barrier impairment, as shown by increased BAL protein and plasma SP-D concentrations; histology revealed epithelial denudation. These data prove that NA-induced lung impairment renders the lungs of mice more sensitive to an airway challenge with ClO-, confirming the hypothesis that incomplete barrier repair, followed by irritant exposure results in airway hypersensitivity.





Similar content being viewed by others
References
Alarie Y, Ferguson JS, Stock MF, Weyel DA, Schaper M (1987) Sensory and pulmonary irritation of methyl isocyanate in mice and pulmonary irritation and possible cyanidelike effects of methyl isocyanate in guinea pigs. Environ Health Perspect 72(June):159–167
Benfante A, Battaglia S, Principe S, Mitri CD, Paternò A, Spatafora M, Scichilone N (2016) Asthmatics with high levels of serum surfactant protein D have more severe disease. Eur Respir J 47(6):1864–1867. https://doi.org/10.1183/13993003.02142-2015
Bernstein JA, Alexis N, Barnes C, Bernstein IL, Nel A, Peden D, Diaz-Sanchez D, Tarlo SM, Williams PB, Bernstein JA (2004) Health effects of air pollution. J Allergy Clin Immunol 114(5):1116–1123. https://doi.org/10.1016/j.jaci.2004.08.030
Brooks SM, Bernstein IL (2011) Irritant-induced airway disorders. Immunol Allergy Clin N Am 31(4):747–768. https://doi.org/10.1016/j.iac.2011.07.002
Brooks SM, Weiss MA, Bernstein IL (1985) Reactive airways dysfunction syndrome (RADS). Persistent asthma syndrome after high level irritant exposures. Chest 88(3):376–384
Buckpitt A, Chang AM, Weir A, Van Winkle L, Duan X, Philpot R, Plopper C (1995) Relationship of cytochrome P450 activity to clara cell cytotoxicity. IV. Metabolism of naphthalene and naphthalene oxide in microdissected airways from mice, rats, and hamsters. Mol Pharmacol 47(1):74–81
Buckpitt A, Boland B, Isbell M, Morin D, Shultz M, Baldwin R, Chan K, Karlsson A, Lin C, Taff A, West J, Fanucchi M, Van Winkle L, Plopper C (2002) Naphthalene-induced respiratory tract toxicity: metabolic mechanisms of toxicity. Drug Metab Rev 34(4):791–820. https://doi.org/10.1081/DMR-120015694
Casas L, Zock J-P, Torrent M, García-Esteban R, Gracia-Lavedan E, Hyvärinen A, Sunyer J (2013) Use of household cleaning products, exhaled nitric oxide and lung function in children. Eur Respir J 42(5):1415–1418. https://doi.org/10.1183/09031936.00066313
Crouch EC (2000) Surfactant protein-D and pulmonary host defense. Respir Res 1 (Aug):6. https://doi.org/10.1186/rr19
Devos FC, Maaske A, Robichaud A, Pollaris L, Seys S, Lopez CA, Verbeken E, Tenbush M, Lories R, Nemery B, Hoet P, Vanoirbeek, J (2017) Forced expiration measurements in mouse models of obstructive and restrictive lung diseases. Respir Res 18(1):123. https://doi.org/10.1186/s12931-017-0610-1
Evans RB (2005) Chlorine: state of the art. Lung 183(3):151–167. https://doi.org/10.1007/s00408-004-2530-3
Fujita M, Shannon JM, Ouchi H, Voelker DR, Nakanishi Y, Mason RJ (2005) Serum surfactant protein D is increased in acute and chronic inflammation in mice. Cytokine 31(1):25–33. https://doi.org/10.1016/j.cyto.2005.02.006
Georas SN, Rezaee F (2014) Epithelial barrier function: at the frontline of asthma immunology and allergic airway inflammation. J Allergy Clin Immunol 134(3):509–520. https://doi.org/10.1016/j.jaci.2014.05.049
Greeley MA, Van Winkle LS, Edwards PC, Plopper CG (2010) Airway trefoil factor expression during naphthalene injury and repair. Toxicol Sci 113(2):453–467. https://doi.org/10.1093/toxsci/kfp268
Heijink IH, Noordhoek JA, Timens W, van Oosterhout AJ, Postma DS (2014) Abnormalities in airway epithelial junction formation in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 189(11):1439–1442. https://doi.org/10.1164/rccm.201311-1982LE
Holgate ST (2007) Epithelium dysfunction in asthma. J Allergy Clin Immunol 120(6):1233–1244–1246. https://doi.org/10.1016/j.jaci.2007.10.025
Hox V, Vanoirbeek JA, Callebaut I, Bobic S, De Vooght V, Ceuppens J, Hoet P, Nemery B, Hellings PW (2011) Airway exposure to hypochlorite prior to ovalbumin induces airway hyperreactivity without evidence for allergic sensitization. Toxicol Lett 204(2–3):101–107. https://doi.org/10.1016/j.toxlet.2011.04.017
Hox V, Vanoirbeek JA, Alpizar YA, Voedisch S, Callebaut I, Bobic S, Sharify A et al (2013) Crucial role of transient receptor potential ankyrin 1 and mast cells in induction of nonallergic airway hyperreactivity in mice. Am J Respir Crit Care Med 187(5):486–493. https://doi.org/10.1164/rccm.201208-1358OC
Hoyle GW, Svendsen ER (2016) Persistent effects of chlorine inhalation on respiratory health. Ann N Y Acad Sci. https://doi.org/10.1111/nyas.13139
Jonasson S, Koch B, Bucht A (2013) Inhalation of chlorine causes long-standing lung inflammation and airway hyperresponsiveness in a murine model of chemical-induced lung injury. Toxicology 303(January):34–42. https://doi.org/10.1016/j.tox.2012.10.022
Karagiannis TC, Li X, Tang MM, Orlowski C, El-Osta A, Tang MLK, Royce SG (2012) Molecular model of naphthalene-induced DNA damage in the murine lung. Hum Exp Toxicol 31(1):42–50. https://doi.org/10.1177/0960327111407228
López IP, Piñeiro-Hermida S, Pais RS, Torrens R, Hoeflich A, Pichel JG (2016) Involvement of Igf1r in bronchiolar epithelial regeneration: role during repair kinetics after selective club cell ablation. PloS One 11(11):e0166388. https://doi.org/10.1371/journal.pone.0166388
Maestrelli P, Boschetto P, Fabbri LM, Mapp CE (2009) Mechanisms of occupational asthma. J Allergy Clin Immunol 123(3):531–542–544. https://doi.org/10.1016/j.jaci.2009.01.057
Martin JG, Campbell HR, Iijima H, Gautrin D, Malo JL, Eidelman DH, Hamid Q, Maghni K (2003) Chlorine-induced injury to the airways in mice. Am J Respir Crit Care Med 168(5):568–574. https://doi.org/10.1164/rccm.200201-021OC
McGovern TK, Powell WS, Day BJ, White CW, Govindaraju K, Karmouty-Quintana H, Lavoie N, Tan J, Martin JG (2010) Dimethylthiourea protects against chlorine induced changes in airway function in a murine model of irritant induced asthma. Respir Res 11(Oct):138. https://doi.org/10.1186/1465-9921-11-138
McGovern TK, Goldberger M, Allard B, Farahnak S, Hamamoto Y, O’Sullivan M, Hirota N, Martel G, Rousseau S, Martin JG (2015) Neutrophils mediate airway hyperresponsiveness after chlorine-induced airway injury in the mouse. Am J Respir Cell Mol Biol 52(4):513–522. https://doi.org/10.1165/rcmb.2013-0430OC
Pan T, Nielsen LD, Allen MJ, Shannon KM, Shannon JM, Selman M, Mason RJ (2002) Serum SP-D is a marker of lung injury in rats. Am J Physiol Lung Cell Mol Physiol 282(4):L824–L832. https://doi.org/10.1152/ajplung.00421.2000
Plopper CG, Cranz DL, Kemp L, Serabjit-Singh CJ, Philpot RM (1987) Immunohistochemical demonstration of cytochrome P-450 monooxygenase in clara cells throughout the tracheobronchial airways of the rabbit. Exp Lung Res 13(1):59–68. https://doi.org/10.3109/01902148709064309
Plopper CG, Suverkropp C, Morin D, Nishio S, Buckpitt A (1992) Relationship of cytochrome P-450 activity to clara cell cytotoxicity. I. Histopathologic comparison of the respiratory tract of mice, rats and hamsters after parenteral administration of naphthalene. J Pharmacol Exp Ther 261(1):353–363
Rezaee F, Georas SN (2014) Breaking barriers. New insights into airway epithelial barrier function in health and disease. Am J Respir Cell Mol Biol 50(5):857–869. https://doi.org/10.1165/rcmb.2013-0541RT
Royce SG, Li X, Tortorella S, Goodings L, Chow BS, Giraud AS, Tang ML, Samuel CS (2014a) Mechanistic insights into the contribution of epithelial damage to airway remodeling. Novel therapeutic targets for asthma. Am J Respir Cell Mol Biol 50(1):180–192. https://doi.org/10.1165/rcmb.2013-0008OC
Royce SG, Patel KP, Samuel CS (2014b) Characterization of a novel model incorporating airway epithelial damage and related fibrosis to the pathogenesis of asthma. Lab Investig 94(12):1326–1339. https://doi.org/10.1038/labinvest.2014.119
Shaykhiev R, Crystal RG (2014) Early events in the pathogenesis of chronic obstructive pulmonary disease. Smoking-induced reprogramming of airway epithelial basal progenitor cells. Ann Am Thorac Soc 11 Suppl 5(Dec):S252–S258. https://doi.org/10.1513/AnnalsATS.201402-049AW
Soyka MB, Wawrzyniak P, Eiwegger T, Holzmann D, Treis A, Wanke K, Kast JI, Akdis CA (2012) Defective epithelial barrier in chronic rhinosinusitis: the regulation of tight junctions by IFN-γ and IL-4. J Allergy Clin Immunol 130(5):1087–1096.e10. https://doi.org/10.1016/j.jaci.2012.05.052
Tuck SA, Ramos-Barbón D, Campbell H, McGovern T, Karmouty-Quintana H, Martin JG (2008) Time course of airway remodelling after an acute chlorine gas exposure in mice. Respir Res 9(Aug):61. https://doi.org/10.1186/1465-9921-9-61
Van Winkle LS, Buckpitt AR, Nishio SJ, Isaac JM, Plopper CG (1995) Cellular response in naphthalene-induced Clara cell injury and bronchiolar epithelial repair in mice. Am J Physiol 269(6 Pt 1):L800–L818
Van Winkle LS, Johnson ZA, Nishio SJ, Brown CD, Plopper CG (1999) Early events in naphthalene-induced acute Clara cell toxicity: comparison of membrane permeability and ultrastructure. Am J Respir Cell Mol Biol 21(1):44–53. https://doi.org/10.1165/ajrcmb.21.1.3630
Vandenplas O, Wiszniewska M, Raulf M, de Blay F, Gerth R, van Wijk G, Moscato B, Nemery et al (2014) EAACI position paper: irritant-induced asthma. Allergy 69(9):1141–1153. https://doi.org/10.1111/all.12448
Velde GV, Poelmans J, Langhe E, Hillen A, Vanoirbeek J, Himmelreich U, Lories RJ (2016) Longitudinal micro-CT provides biomarkers of lung disease that can be used to assess the effect of therapy in preclinical mouse models, and reveal compensatory changes in lung volume. Dis Models Mech 9(1):91–98. https://doi.org/10.1242/dmm.020321
White CW, Martin JG (2010) Chlorine gas inhalation: human clinical evidence of toxicity and experience in animal models. Proc Am Thorac Soc 7(4):257–263. https://doi.org/10.1513/pats.201001-008SM
Wigenstam E, Elfsmark L, Koch B, Bucht A, Jonasson S (2016) Acute respiratory changes and pulmonary inflammation involving a pathway of TGF-β1 induction in a rat model of chlorine-induced lung injury. Toxicol Appl Pharmacol 309(Oct):44–54. https://doi.org/10.1016/j.taap.2016.08.027
Winkler C, Atochina-Vasserman EN, Holz O, Beers MF, Erpenbeck VJ, Krug N, Roepcke S, Lauer G, Elmlinger M, Hohlfeld JM (2011) Comprehensive characterisation of pulmonary and serum surfactant protein D in COPD. Respir Res 12(Mar):29. https://doi.org/10.1186/1465-9921-12-29
Xiao C, Puddicombe SM, Field S, Haywood J, Broughton-Head V, Puxeddu I, Haitchi HM et al (2011) Defective epithelial barrier function in asthma. J Allergy Clin Immunol 128(3):549–556.e12. https://doi.org/10.1016/j.jaci.2011.05.038
Yildirim AO, Veith M, Rausch T, Müller B, Kilb P, Van Winkle LS, Fehrenbach H (2008) Keratinocyte growth factor protects against Clara cell injury induced by naphthalene. Eur Respir J 32(3):694–704. https://doi.org/10.1183/09031936.00155107
Acknowledgements
This project was supported by a grant from the University of Leuven Research Council (GOA/14/011, STG/15/024 and C24/17/061) and a grant of the Flemish Research Foundation (Research Grant 1504912N).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Jeroen A.J. Vanoirbeek and Peter H.M. Hoet shared responsibility.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Van Den Broucke, S., Pollaris, L., Vande Velde, G. et al. Irritant-induced asthma to hypochlorite in mice due to impairment of the airway barrier. Arch Toxicol 92, 1551–1561 (2018). https://doi.org/10.1007/s00204-018-2161-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-018-2161-8