Abstract
Aims/hypothesis
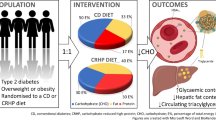
Dietary recommendations for treating type 2 diabetes are unclear but a trend towards recommending a diet reduced in carbohydrate content is acknowledged. We compared a carbohydrate-reduced high-protein (CRHP) diet with an iso-energetic conventional diabetes (CD) diet to elucidate the effects on glycaemic control and selected cardiovascular risk markers during 6 weeks of full food provision of each diet.
Methods
The primary outcome of the study was change in HbA1c. Secondary outcomes reported in the present paper include glycaemic variables, ectopic fat content and 24 h blood pressure. Eligibility criteria were: men and women with type 2 diabetes, HbA1c 48–97 mmol/mol (6.5–11%), age >18 years, haemoglobin >6/>7 mmol/l (women/men) and eGFR >30 ml min−1 (1.73 m)−2. Participants were randomised by drawing blinded ballots to 6 + 6 weeks of an iso-energetic CRHP vs CD diet in an open label, crossover design aiming at body weight stability. The CRHP/CD diets contained carbohydrate 30/50 energy per cent (E%), protein 30/17E% and fat 40/33E%, respectively. Participants underwent a meal test at the end of each diet period and glycaemic variables, lipid profiles, 24 h blood pressure and ectopic fat including liver and pancreatic fat content were assessed at baseline and at the end of each diet period. Data were collected at Copenhagen University Hospital, Bispebjerg and Copenhagen University Hospital, Herlev.
Results
Twenty-eight participants completed the study. Fourteen participants carried out 6 weeks of the CRHP intervention followed by 6 weeks of the CD intervention, and 14 participants received the dietary interventions in the reverse order. Compared with a CD diet, a CRHP diet reduced the primary outcome of HbA1c (mean ± SEM: −6.2 ± 0.8 mmol/mol (−0.6 ± 0.1%) vs −0.75 ± 1.0 mmol/mol (−0.1 ± 0.1%); p < 0.001). Nine (out of 37) pre-specified secondary outcomes are reported in the present paper, of which five were significantly different between the diets, (p < 0.05); compared with a CD diet, a CRHP diet reduced the secondary outcomes (mean ± SEM or medians [interquartile range]) of fasting plasma glucose (−0.71 ± 0.20 mmol/l vs 0.03 ± 0.23 mmol/l; p < 0.05), postprandial plasma glucose AUC (9.58 ± 0.29 mmol/l × 240 min vs 11.89 ± 0.43 mmol/l × 240 min; p < 0.001) and net AUC (1.25 ± 0.20 mmol/l × 240 min vs 3.10 ± 0.25 mmol/l × 240 min; p < 0.001), hepatic fat content (−2.4% [−7.8% to −1.0%] vs 0.2% [−2.3% to 0.9%]; p < 0.01) and pancreatic fat content (−1.7% [−3.5% to 0.6%] vs 0.5% [−1.0% to 2.0%]; p < 0.05). Changes in other secondary outcomes, i.e. 24 h blood pressure and muscle-, visceral- or subcutaneous adipose tissue, did not differ between diets.
Conclusions/interpretation
A moderate macronutrient shift by substituting carbohydrates with protein and fat for 6 weeks reduced HbA1c and hepatic fat content in weight stable individuals with type 2 diabetes.
Trial registration
ClinicalTrials.gov NCT02764021.
Funding
The study was funded by grants from Arla Food for Health; the Novo Nordisk Foundation Center for Basic Metabolic Research, University of Copenhagen; the Department of Clinical Medicine, Aarhus University; the Department of Nutrition, Exercise and Sports, University of Copenhagen; and Copenhagen University Hospital, Bispebjerg.
Similar content being viewed by others

Introduction
Insulin resistance and impaired beta cell function are core defects in type 2 diabetes responsible for the pathophysiological disturbances leading to hyperglycaemia, dyslipidaemia and hypertension [1]. Nutritional therapy constitutes an initial and important treatment, but dietary recommendations are ambiguous. Overweight individuals with type 2 diabetes have been advised to consume a hypocaloric diet low in fat and high in carbohydrates with a low glycaemic index [2, 3], although recent guidelines tend to put less emphasis on macronutrient distribution in favour of individualised diets [4, 5].
Conclusions on the efficacy of low-carbohydrate diets have been conflicting, and in some studies body weight loss has complicated the interpretation of the results [6]. Nevertheless, a lower carbohydrate content of the diet has been found to reduce postprandial and diurnal hyperglycaemia in individuals with type 2 diabetes [7, 8]. We recently extended these findings by demonstrating an immediate beneficial effect of substituting carbohydrates with fat and proteins on postprandial hyperglycaemia, hyperinsulinaemia and dyslipidaemia [9, 10].
Non-alcoholic fatty liver disease (NAFLD) is common in individuals with type 2 diabetes [11]. The histopathological spectrum of NAFLD ranges from non-alcoholic steatosis to non-alcoholic steatohepatitis and fibrosis of the liver [12]. NAFLD has, independent of conventional risk factors, been associated with increased incidence of cardiovascular disease (CVD) in individuals with type 2 diabetes [13].
The present study evaluates the effects on glycaemic control, ectopic fat accumulation and selected risk factors for CVD during 6 weeks of carbohydrate-restricted meal provision in weight stable participants with type 2 diabetes.
Methods
Study design
The study was designed as a 6 + 6 week open label, randomised, crossover-controlled trial. Inclusion criteria were: men and women with type 2 diabetes [14], HbA1c 48–97 mmol/mol (6.5–11%), age >18 years, haemoglobin >6/>7 mmol/l (women/men) and eGFR >30 ml min−1 (1.73 m)−2. Range of eGFR was 56 ml min−1 (1.73 m)−2 to >90 ml min−1 (1.73 m)−2. The lower HbA1c inclusion criterion was adjusted from the initial 58 mmol/mol (7.5%) to 48 mmol/mol (6.5%) to facilitate recruitment. Exclusion criteria were: critical illness, systemic corticosteroid treatment, severe food allergy or intolerance, severe gut disease, alcohol dependence syndrome, injectable diabetes medication, repeated fasting plasma glucose >13.3 mmol/l, urine albumin/creatinine ratio > 300 mg/g, lactation, pregnancy or planning of pregnancy during the study and inability, physically or mentally, to comply with the procedures required by the study protocol. Examinations were initiated in April 2016 and terminated according to the pre-specified study plan in November 2017. All physical examination procedures were performed at Copenhagen University Hospital, Bispebjerg, except the MRI and spectroscopy, which were performed at Copenhagen University Hospital, Herlev. A third-party study nurse randomised the participants by drawing blinded ballots. Block randomisation was applied with block sizes of two and balanced to ensure allocation in a 1:1 ratio.
All participants signed informed consent prior to any study-related procedures. The study was approved by the local Scientific Ethical Committee and carried out in accordance with the Declaration of Helsinki. The study was registered at ClinicalTrials.gov (registration no. NCT02764021).
Dietary interventions
For 12 weeks, participants were provided, free of charge, with five daily meals to fully cover daily energy needs (breakfast 25% of total energy expenditure [TEE%], lunch 30 TEE%, pre-dinner snack 7.5 TEE%, dinner 30 TEE%, post-dinner snack 7.5 TEE%). Consistent with diabetes dietary guidelines provided by the Diabetes and Nutrition Study Group (DNSG) of the EASD [2], the conventional diabetes (CD) diet consisted of 50% energy (E%) carbohydrate, 17 E% protein and 33 E% fat. Macronutrient composition of the carbohydrate-reduced high-protein (CRHP) diet was 30 E% carbohydrate, 30 E% protein and 40 E% fat (electronic supplementary material [ESM] Table 1). Meals were prepared as a 7 day rotational procedure and produced and distributed twice weekly by trained personnel at the Department of Nutrition, Exercise and Sports, Copenhagen University. Participants were restricted to consume study meals and beverages only according to assigned intervention. Ad libitum intake of non-caloric beverages was allowed.
Provision of foods for maintenance of body weight
Daily total energy expenditure (TEE) was estimated by an algorithm based on participant characteristics and body composition measured by dual-energy x-ray absorptiometry (Lunar iDXA, General Electric Healthcare, GE Medical Systems, Belgium), as previously described [9, 15]. Body weight was measured twice weekly. If changes occurred, energy intake was adjusted accordingly, by lowering/increasing portion sizes and/or by adding/retracting with additional 0.5 MJ macronutrient-adjusted meal snacks, to ensure weight stability. Participants were repeatedly instructed to maintain a constant physical activity level. Furthermore, participants were instructed to keep records of any leftovers and were interviewed twice per week to ensure adherence to study diets.
Urine samples
Twenty-four-hour urine samples were collected after 4 weeks on each diet to evaluate compliance to interventions unconfounded by the investigational programme. We used quantification of 24 h urinary urea excretion, based on an enzymatic ultraviolet test (ABX Pentra 400 Urea CP, Horiba ABX SAS, France), which provides a validated measure of protein intake [16]. Moreover, samples were used to assess 24 h urinary glucose excretion by colorimetry (ABX Pentra 400 Glucose PAP CP, Horiba ABX SAS), 24 h urinary albumin excretion by an immunoturbidimetric test (ABX Pentra 400 Micro Albumin 2 CP, Horiba ABX SAS) and 24 h urinary creatinine excretion based on the reaction between creatinine and sodium picrate (ABX Pentra 400 Creatinine 120 CP, Horiba ABX SAS) to calculate albumin/creatinine ratio.
Blood samples
Blood samples were collected after a 12 h overnight fast. Serum was stored at room temperature for 30 min to ensure coagulation before centrifugation. Plasma was collected in EDTA-treated test tubes and immediately centrifuged after sampling. Serum was used for analysis of insulin, C-peptide, NEFA, total cholesterol, HDL-cholesterol, triacylglycerol, apolipoprotein A1 (ApoA-1) and apolipoprotein B (ApoB). IMMULITE 2000 Immunoassay System (Siemens Healthcare, Erlangen, Germany) was used to analyse insulin and C-peptide. Wako NEFA-HR(2) (ASC-ACOD method, Wako Chemicals, Neuss, Germany) was used for fasting NEFA concentrations. Enzymatic colorimetric assays (CHOL2, HDLC4 and TRIGL, Cobas 6000, Roche Diagnostics, Mannheim, Germany) were used to analyse total cholesterol, HDL-cholesterol and triacylglycerol, respectively. LDL-cholesterol was calculated by using the Friedewald equation [17]. Non-HDL-cholesterol was calculated by subtracting HDL-cholesterol from total cholesterol. ApoA-1 and ApoB were analysed using an immunoturbidimetric test (APOAT and APOBT, Cobas 6000, Roche Diagnostics).
Plasma glucose concentrations were analysed immediately after sampling by use of YSI 2300 STAT Plus (Yellow Springs Instruments, Yellow Springs, OH, USA). Whole blood was collected and handled in accordance with local operating procedure for analysis of HbA1c using the Tosoh Automated Glycohemoglobin Analyser HLC-723G8 (G8; Tosoh Bioscience, Japan). Insulin resistance (HOMA2-IR) was calculated by use of the HOMA2 calculator (version 2.2.3; www.dtu.ox.ac.uk/homacalculator/, accessed 14 January 2019) based on fasting plasma glucose and C-peptide.
Mixed meal testing
As displayed in ESM Fig. 1, a CRHP mixed meal test (MMT) was performed after 6 weeks of CRHP intervention and a CD MMT was performed after 6 weeks of CD intervention in order to test the postprandial responses of the two study diets, i.e. differences in macronutrient composition. Solid phase mixed test meals (ESM Table 1) were produced in five energy levels ranging from 2.25 MJ (598 kcal) to 3.25 MJ (837 kcal) to approximately match 25% of TEE for each participant. All ingredients were weighed out with an accuracy of 0.5 g by trained personnel at the research kitchen, Department of Nutrition, Exercise and Sports, University of Copenhagen. Participants were instructed to avoid any strenuous physical activity 48 h prior to meal tests. Alcohol consumption was prohibited during the entire study. Meal tests were performed after consumption of a standardised CD or CRHP dinner according to assigned intervention and a subsequent 12 h overnight fast. A peripheral i.v. cannula was inserted in an antecubital vein before a solid CRHP or iso-energetic CD meal was served at time 0 and consumed within 25 min. Participants remained sedentary throughout the examination.
Magnetic resonance examination
Magnetic resonance measurements were performed at baseline and at the end of each dietary intervention period using a 3.0 T Ingenia MRI system (Philips Medical Systems, Best, the Netherlands) with a dStream torso coil. Magnetic resonance scanning protocol included planning scans, single voxel spectroscopy (Point RESolved Spectroscopy [PRESS]) for measuring liver fat content and muscle fat content [18, 19], and chemical shift encoding-based water-fat imaging (mDixon) for measuring pancreatic fat content, visceral adipose tissue (VAT), subcutaneous adipose tissue (SAT) and waist perimeter. Means of duplicate analyses of pancreas fat fractions were used to reduce intra-observer variability. VAT and SAT volumes and waist perimeter were measured using a single transverse section of 10 mm thickness acquired at the middle of the third lumbar vertebra, as described in detail previously [20]. All magnetic resonance data were analysed by a research technician blinded to treatment.
Diurnal blood pressure
Twenty-four-hour ambulatory blood pressure (ambulatory blood pressure monitor: Model 90217, Spacelabs Healthcare, WA, USA) was measured at home at baseline and at the end of each dietary treatment period.
Statistical analysis
An a priori calculation of sample size was performed based on earlier published effect size [7]. With 90% power, a sample size of six participants was sufficient to detect a 12.1 mmol/mol (~1.1%) between-diet difference in the primary outcome, HbA1c, in this crossover setting, provided an SD of ±6.6 mmol/mol (~0.6%) and a significance level of 5%. To account for possible dropouts, smaller than expected effect size and multiple pre-specified exploratory analyses of secondary endpoints, a total of 30 participants were included.
The study includes a total of 37 pre-specified secondary outcomes, of which nine, i.e. glycaemic variables, ectopic fat content and 24 h blood pressure, are presented in this paper (for details on all outcomes, please refer to ESM Table 2). Other pre-specified outcomes, i.e. beta cell function, NEFA metabolism, low-grade inflammation, growth hormone axis, heart rate variability, gut and satiety hormones and subjective satiety will, following analyses, be reported subsequently. The statistical analysis of the primary outcome and the secondary outcomes was performed with a significance level of p < 0.05 (two-tailed test). Due to multiple comparisons, a risk of false-positive results may inherently exist.
Results are presented as means (±SEM) or as medians (interquartile range) if normal distribution assumptions were not met as assessed by visual examination. AUC was calculated using the trapezoidal rule. Net AUC was calculated by subtracting area below baseline values from the AUC. A linear mixed effects model was used to evaluate differences in treatment effects between diets adjusted for body weight change for all variables reported, except body weight and BMI, for which a paired sample t test was applied. All linear mixed effects models included body weight change and an interaction between diet and period as fixed effects. To account for inter-participant variability caused by the paired design, participants were applied as random effects. Residuals were visually examined for linearity and normality. If appropriate, data were log transformed to meet model assumptions for the mixed effects model. Depending on the distribution of data, a paired sample t test or a Wilcoxon signed-rank test was used to evaluate differences between baseline circulating and imaging biomarkers. Baseline values were defined as means of the initial measurements at the beginning of the two CRHP and the two CD dietary treatment periods. Due to the crossover design, the final measure of the first 6 week period was used as the baseline for the second 6 week period. Considering the low dropout rate (2 out of 30), only per-protocol analyses were carried out. All available data were included, i.e. n = 28, except for 24 h ambulatory blood pressure monitoring and hepatic and pancreatic fat fractions, which, due to technical failure and problems with diurnal urine sampling, were obtained in 27 participants only. Statistical analyses were performed by use of RStudio (Version 0.99.473; RStudio, Boston, MA, USA) and graphical presentations were made using GraphPad Prism (Version 7.02; GraphPad Software, San Diego, CA, USA).
Results
In addition to the primary outcome, we report on nine secondary and 27 exploratory outcomes, of which five secondary and 11 exploratory were significant at p < 0.05.
Participants
Baseline characterisation of study participants and the study flow diagram are displayed in Table 1 and Fig. 1, respectively. All glucose-lowering, lipid-lowering and antihypertensive medications were stable at least 2 months prior to initiation of the study and remained unchanged throughout the study period (Table 1).
Baseline measurements
Except for fasting triacylglycerol (p = 0.01), none of the circulating biomarkers, MRI- or spectroscopy-derived markers of fat content or anthropometric measurements differed between diets at baseline for each intervention period.
Primary endpoint
HbA1c
Compared with a CD diet, a CRHP diet reduced HbA1c (−6.2 ± 0.8 mmol/mol vs −0.75 ± 1.0 mmol/mol, corresponding to −0.6 ± 0.1% vs −0.1 ± 0.1%; p < 0.001) (Table 2, Fig. 2).
Baseline adjusted change in (a) HbA1c (between diets, p < 0.001), (b) fasting triacylglycerol (between diets, p < 0.001), (c) fasting insulin (between diets, p = 0.296) and (d) total fasting body weight (between diets, p = 0.071) during 6 weeks of CRHP and CD treatment. All graphs are presented as means (±SEM). White squares, CRHP diet; black triangles, CD diet
Secondary endpoints
Fasting glucose and postprandial glucose excursions
Compared with a CD diet, a CRHP diet reduced fasting plasma glucose (−0.71 ± 0.20 mmol/l vs 0.03 ± 0.23 mmol/l; p < 0.05; Table 2). When participants were subjected to a meal challenge after 6 weeks of treatment, a CRHP diet reduced postprandial plasma glucose AUC and net AUC by 19.4% and 59.7% (both p < 0.001) as compared with a CD diet (Table 3, Fig. 3).
Postprandial responses to a CRHP diet and a CD diet. Postprandial (a) plasma glucose, (b) insulin, (c) C-peptide, (d) NEFA and (e) triacylglycerol excursions during a 4 h MMT on a CD and a CRHP breakfast (Br) after 6 weeks on dietary treatment. All graphs and AUC/net AUC bars are presented as means (±SEM). Between-diet differences were evaluated by a linear mixed effects model; **p < 0.01, ***p < 0.001. White squares, CRHP diet; black triangles, CD diet
Magnetic resonance
Compared with a CD diet, a CRHP diet reduced liver fat fraction (−2.4% [−7.8% to −1.0%] vs 0.2% [−2.3% to 0.9%]; p < 0.01; Fig. 4) and pancreatic fat fraction (−1.7% [−3.5% to 0.6%] vs 0.5% [−1.0% to 2.0%]; p < 0.05). Changes in muscle fat fraction, VAT and SAT did not differ between diets (Table 2).
Changes in hepatic fat content. Data are presented as medians (interquartile range) with spaghetti-diagrams to present individualised changes in the hepatic fat fraction. The line within each box represents the median, and the top and bottom of the box represent the 75th and 25th percentiles, respectively. The whiskers indicate the maximum and minimum values. The between-diet difference was evaluated by a linear mixed effects model; **p < 0.01
Diurnal blood pressure measurements
Changes in systolic or diastolic 24 h blood pressure during CRHP and CD dietary therapy did not differ significantly between diets (Table 2).
Exploratory endpoints
Metabolic and lipid variables
Changes in fasting insulin, fasting C-peptide and HOMA-IR did not differ between diets (Table 2). Compared with a CD diet, a CRHP diet reduced fasting triacylglycerol (−0.43 mmol/l [−0.77 to −0.16] vs 0.12 mmol/l [0.01 to 0.44]; p < 0.001), total cholesterol (−0.44 ± 0.10 mmol/l vs 0.00 ± 0.10 mmol/l; p < 0.05) and non-HDL-cholesterol (−0.44 ± 0.10 mmol/l vs 0.04 ± 0.09 mmol/l; p < 0.05). Changes in LDL-cholesterol, HDL-cholesterol, ApoA-1, ApoB, ApoA-1/ApoB ratio and fasting NEFA did not differ between diet interventions (Table 2).
Mixed meal testing
Participants underwent mixed meal testing after each of the two 6 week diets. This meal challenge showed that the CRHP diet, when compared with the CD diet, reduced postprandial insulin AUC and net AUC by 8.0% and 11.4% (both p < 0.05), postprandial C-peptide AUC and net AUC by 9.6% and 15.8% (both p < 0.05), postprandial NEFA AUC and net AUC by 9.6% and 12.4% (both p < 0.05), and postprandial triacylglycerol AUC by 30.3% (p < 0.001). No difference was found in postprandial net triacylglycerol AUC between diets due to the difference in fasting value (Table 3, Fig. 3).
Twenty-four-hour urinary excretion
Twenty-four-hour urinary urea excretion was measured as a marker of compliance of protein intake and was 82% higher on a CRHP diet compared with a CD diet (p < 0.001). This corresponded to the 79.4% higher protein content of the CRHP diet. No differences between CRHP and CD diets were found in 24 h urinary glucose excretion, albumin excretion or albumin/creatinine ratio (Table 3).
Anthropometric measurements
Changes in body weight, BMI or waist circumference during CRHP and CD dietary treatment did not differ between diets (Table 2).
Adverse effects
During the CRHP diet intervention, four participants suffered from constipation (one was treated with laxative), one was diagnosed with vitreous degeneration, one was diagnosed with paroxysmal atrial fibrillation, one showed symptoms of bursitis and two complained of sleep disturbances. During CD dietary treatment, one participant complained of sleep disturbances and two suffered from upper respiratory tract infections. Although the literature provides divergent results [21, 22], the higher number of participants suffering from constipation in the CRHP group may relate to the lower content of dietary fibre in the CRHP diet, but other adverse effects seem unrelated to the intervention.
Discussion
In this randomised controlled clinical dietary intervention study with 6 + 6 weeks of full food provision, the consumption of a CRHP diet reduced to a greater extent the primary outcome, HbA1c, than a CD diet, in addition to the following pre-specified secondary outcomes: fasting plasma glucose, postprandial plasma glucose and hepatic and pancreatic fat content. Remaining secondary outcomes, i.e. 24 h blood pressure, muscle adipose tissue, VAT or SAT, revealed no differences between diets. As exploratory analyses, we found that a CRHP diet lowered total cholesterol, non-HDL-cholesterol and fasting triacylglycerol to a greater extent than a CD diet, indicating improved atherogenic lipid profile following carbohydrate restriction. However, these findings must be interpreted with caution due to their exploratory nature.
In agreement with previous findings [7,8,9], postprandial plasma glucose was significantly lower on a CRHP diet as compared with a CD diet, which was in part explained by the dose–response relationship between dietary carbohydrates and glucose excursions. Recently, we demonstrated that a CRHP diet acutely reduced postprandial blood glucose level and insulinaemia in individuals with type 2 diabetes [9]. Although the applied MMT method is unsuitable to address changes or adaptions to study diets over time, the present findings confirm that the effect, i.e. the reduction in postprandial blood glucose level and insulinaemia, is maintained after 6 weeks of a highly controlled CRHP dietary treatment. Importantly, postprandial glycaemia, compared with fasting glycaemia, contributes relatively more to HbA1c in individuals with well-controlled type 2 diabetes [23]. As no differences between diets were found in HOMA-IR, and only a modest decrease in fasting plasma glucose was observed on the CRHP diet, the reduction in HbA1c may primarily be due to a persistent reduction in postprandial blood glucose level during the CRHP dietary treatment. Yet, other mechanisms may contribute to the improved glycaemic control on the CRHP diet, e.g. the relatively higher content of monounsaturated fatty acids [24] and specific food items such as dairy products, nuts, etc. [25, 26]. We further speculate that the higher content of fat in the CRHP diet may slow the rate of gastric emptying and, in turn, decrease the rate of glucose delivery to the circulation.
Our study is in line with a growing body of evidence supporting the short-term beneficial effect of carbohydrate restriction on glycaemic control [27, 28]. However, inconclusive results exist regarding the long-term effects of such lower-carbohydrate diets [29,30,31]. Low compliance to adhere to various study diets in longer-term studies may have played a central role in the absence of significant effects. In the present study, the duration of food provision was limited to 6 weeks, which was not an adequate time to achieve steady state for changes in HbA1c. However, in contrast to the CD diet, the CRHP diet gradually reduced mean HbA1c by 6.2 mmol/mol (0.6%), suggesting that HbA1c may be even further reduced over a longer diet period. In this context, the UK Prospective Diabetes Study found that a 10 mmol/mol (~1%) reduction in HbA1c over 10 years significantly decreased the risk of microvascular complications in individuals with type 2 diabetes [32]. Hence, the demonstrated decrement in HbA1c in relation to full CRHP food provision is most likely of clinical relevance.
Individuals with type 2 diabetes are at high risk of CVD and hypertension, and lipid disturbances are recognised to play a significant role in the development and progression of CVD [33]. We found that a CRHP diet improved blood lipids towards a less atherogenic profile by reducing total cholesterol, fasting triacylglycerol and non-HDL-cholesterol. However, blood lipid analyses were exploratory only and long-term studies are needed to reproduce these findings and to evaluate whether a CRHP diet will eventually affect the risk of both micro- and macrovascular complications.
Studies have reported that hepatic de novo lipogenesis is elevated in hyperinsulinaemic individuals [34, 35] and in individuals consuming a low-fat high-carbohydrate diet [35]. In the present study, a moderate reduction in dietary carbohydrate induced a substantial decrease in postprandial glucose and insulin concentrations and a concomitant decline in the hepatic fat fraction. Together, these findings indicate that the present shift in macronutrient composition may modulate the rate of de novo lipogenesis and subsequently reduce the hepatic fat fraction.
An elevated supply of fatty acids to the liver is considered essential for hepatic fat accumulation [36]. The reduction in fasting and postprandial triacylglycerol concentrations during the CRHP diet may result from reduced de novo lipogenesis due to the persistent reduction in insulin excursions. Moreover, lipoprotein lipase (LPL) is activated by insulin to promote hydrolysis of dietary-derived chylomicrons and triacylglycerol-rich lipoproteins, subsequently leading to a ‘spill-over’ of fatty acids, which thereby contributes to the circulating NEFA pool [37]. Thus, a reduction in de novo lipogenesis and the lower NEFA levels may both contribute to lower the hepatic fat fraction found following the CRHP diet. Population-based studies suggest the 1H-MRS cut-off value for hepatic steatosis to be 5.6% [38]. However, newer data based on the correlation between liver biopsies and proton magnetic resonance spectroscopy (1H-MRS) suggest that a lower cut-off value of approximately 1.8–3.0% is more appropriate [39, 40]. Irrespective of the defined upper limit of hepatic steatosis, all participants with steatosis (except one) showed a decrease in liver fat content on the CRHP intervention (Fig. 4). As steatosis has previously been linked to an attenuated ability to suppress endogenous glucose production [41], the observed decrease in liver fat content on the CRHP diet may also improve glucose homeostasis.
No clear consensus exists regarding the pathophysiological aspects and clinical implications of fat accumulation in the pancreas [42, 43]. In the present study, a CRHP diet was found to decrease pancreatic fat content as compared with a CD diet, but whether the reduction in pancreatic fat is mechanistically related to the improved glucose metabolism is yet to be clarified.
The safety of substituting dietary carbohydrates with fat and proteins is debated [44, 45]. A large prospective cohort study, performed in mainly non-diabetic individuals, concluded that substituting carbohydrates with animal protein and fat was associated with an increased all-cause mortality [46], but a lower mortality risk was found when carbohydrates were substituted for plant protein and fat, emphasising that food sources may be pivotal [46]. However, these associations should be interpreted with caution as the causality cannot be established by observational studies.
High-protein diets have been proposed to have potential deleterious effect on renal function [47]. A meta-analysis of randomised controlled trials found that low-protein diets improved GFR in individuals with diabetic nephropathy [48]. In the present study, the two diets did not affect 24 h urinary albumin excretion or albumin/creatinine ratio. This finding is consistent with previous studies investigating the long-term effect of low-carbohydrate, high-protein diets on renal markers in individuals with type 2 diabetes without overt kidney disease [49, 50].
Among the limitations of this study are the unblinded design, the short duration of intervention, the sex imbalance and the lack of objective quantification of physical activity level. The study was designed to address numerous endpoints (ESM Table 2). As stated, this possesses an important limitation due to multiplicity and the risk of false-positive findings. Hence, secondary, and especially exploratory, endpoints must be interpreted with caution and further studies are warranted to reproduce and confirm our present findings. We chose not to use a washout period between the two 6 week periods. There were several reasons for this. First, HbA1c reflects the average glucose level over the previous 2–3 months, and to obtain equilibrium on HbA1c a similar washout period is required. Second, the participants’ eating habits during a washout period might be influenced by the first study period. This could, subsequently, influence baseline values of the second study period. Third, we wanted to minimise the dropout rate by reducing the already very large burden of the study for the participants. The strengths of the present study were the low dropout rate, the equal control of body weight in the two diet groups and the high adherence to study diets ensured by full meal provision and close surveillance of study participants. Moreover, compliance was confirmed by quantification of 24 h urinary urea excretion.
In conclusion, we found that a CRHP diet, as compared with a CD diet, decreased hepatic and pancreatic fat content and improved glycaemic control in well-controlled participants with type 2 diabetes during 6 weeks of food provision and stable body weight conditions. This calls for future studies to elucidate the long-term beneficial effects and feasibility of CRHP diets in a real-life setting.
Data availability
Data may be shared upon reasonable request and approval by the Danish Data Protection Agency.
Abbreviations
- CD:
-
Conventional diabetes
- CRHP:
-
Carbohydrate-reduced high-protein
- CVD:
-
Cardiovascular disease
- E%:
-
Energy per cent
- MMT:
-
Mixed meal test
- NAFLD:
-
Non-alcoholic fatty liver disease
- SAT:
-
Subcutaneous adipose tissue
- TEE:
-
Total energy expenditure
- VAT:
-
Visceral adipose tissue
References
Kahn SE, Cooper ME, Del Prato S (2014) Pathophysiology and treatment of type 2 diabetes: perspectives on the past, present, and future. Lancet 383(9922):1068–1083. https://doi.org/10.1016/S0140-6736(13)62154-6
Mann JI, De Leeuw I, Hermansen K et al (2004) Evidence-based nutritional approaches to the treatment and prevention of diabetes mellitus. Nutr Metab Cardiovasc Dis 14(6):373–394. https://doi.org/10.1016/S0939-4753(04)80028-0
Ajala O, English P, Pinkney J (2013) Systematic review and meta-analysis of different dietary approaches to the management of type 2 diabetes. Am J Clin Nutr 97(3):505–516. https://doi.org/10.3945/ajcn.112.042457
American Diabetes Association (2018) Lifestyle management: standards of medical care in diabetes—2018. Diabetes Care 41(Suppl 1):S38–S50. https://doi.org/10.2337/dc18-S004
Davies MJ, D’Alessio DA, Fradkin J et al (2018) Management of hyperglycemia in type 2 diabetes, 2018.A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 61(12):2461–2498. https://doi.org/10.1007/s00125-018-4729-5
Wheeler ML, Dunbar SA, Jaacks LM et al (2012) Macronutrients, food groups, and eating patterns in the management of diabetes: a systematic review of the literature, 2010. Diabetes Care 35(2):434–445. https://doi.org/10.2337/dc11-2216
Nuttall FQ, Schweim K, Hoover H, Gannon MC (2008) Effect of the LoBAG30 diet on blood glucose control in people with type 2 diabetes. Br J Nutr 99(3):511–519. https://doi.org/10.1017/S0007114507819155
Coulston AM, Hollenbeck CB, Swislocki AL, Chen YD, Reaven GM (1987) Deleterious metabolic effects of high-carbohydrate, sucrose-containing diets in patients with non-insulin-dependent diabetes mellitus. Am J Med 82(2):213–220. https://doi.org/10.1016/0002-9343(87)90058-1
Samkani A, Skytte MJ, Kandel D et al (2018) A carbohydrate-reduced high-protein diet acutely decreases postprandial and diurnal glucose excursions in type 2 diabetes patients. Br J Nutr 119(8):910–917. https://doi.org/10.1017/S0007114518000521
Samkani A, Skytte MJ, Anholm C et al (2018) The acute effects of dietary carbohydrate reduction on postprandial responses of non-esterified fatty acids and triglycerides: a randomized trial. Lipids Health Dis 17(1):295. https://doi.org/10.1186/s12944-018-0953-8
Cusi K, Sanyal AJ, Zhang S et al (2017) Non-alcoholic fatty liver disease (NAFLD) prevalence and its metabolic associations in patients with type 1 diabetes and type 2 diabetes. Diabetes Obes Metab 19(11):1630–1634. https://doi.org/10.1111/dom.12973
Hashimoto E, Taniai M, Tokushige K (2013) Characteristics and diagnosis of NAFLD/NASH. J Gastroenterol Hepatol 28(Suppl 4):64–70. https://doi.org/10.1111/jgh.12271
Targher G, Bertolini L, Rodella S et al (2007) Nonalcoholic fatty liver disease is independently associated with an increased incidence of cardiovascular events in type 2 diabetic patients. Diabetes Care 30(8):2119–2121. https://doi.org/10.2337/dc07-0349
American Diabetes Association (2018) Classification and diagnosis of diabetes: standards of medical care in diabetes—2018. Diabetes Care 41(Suppl 1):S13–S27. https://doi.org/10.2337/dc18-S002
Nielsen S, Hensrud DD, Romanski S, Levine JA, Burguera B, Jensen MD (2000) Body composition and resting energy expenditure in humans: role of fat, fat-free mass and extracellular fluid. Int J Obes Relat Metab Disord 24(9):1153–1157. https://doi.org/10.1038/sj.ijo.0801317
Bingham SA (2003) Urine nitrogen as a biomarker for the validation of dietary protein intake. J Nutr 133(Suppl 3):921S–924S. https://doi.org/10.1093/jn/133.3.921S
Friedewald WT, Levy RI, Fredrickson DS (1972) Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 18(6):499–502
Chabanova E, Fonvig CE, Bojsoe C, Holm JC, Thomsen HS (2017) 1H MRS assessment of hepatic fat content: comparison between normal- and excess-weight children and adolescents. Acad Radiol 24(8):982–987. https://doi.org/10.1016/j.acra.2017.02.010
Chabanova E, Bille DS, Thisted E, Holm JC, Thomsen HS (2012) MR spectroscopy of liver in overweight children and adolescents: investigation of 1H T2 relaxation times at 3T. Eur J Radiol 81(5):811–814. https://doi.org/10.1016/j.ejrad.2011.02.017
Fonvig CE, Bille DS, Chabanova E, Nielsen TR, Thomsen HS, Holm JC (2012) Muscle fat content and abdominal adipose tissue distribution investigated by magnetic resonance spectroscopy and imaging in obese children and youths. Pediatr Rep 4(1):e11. https://doi.org/10.4081/pr.2012.e11
Yang J, Wang HP, Zhou L, Xu CF (2012) Effect of dietary fiber on constipation: a meta analysis. World J Gastroenterol 18(48):7378–7383. https://doi.org/10.3748/wjg.v18.i48.7378
Ho KS, Tan CY, Mohd Daud MA, Seow-Choen F (2012) Stopping or reducing dietary fiber intake reduces constipation and its associated symptoms. World J Gastroenterol 18(33):4593–4596. https://doi.org/10.3748/wjg.v18.i33.4593
Monnier L, Colette C (2006) Contributions of fasting and postprandial glucose to hemoglobin A1c. Endocr Pract 12(Suppl 1):42–46. https://doi.org/10.4158/EP.12.S1.42
Schwingshackl L, Strasser B, Hoffmann G (2011) Effects of monounsaturated fatty acids on glycaemic control in patients with abnormal glucose metabolism: a systematic review and meta-analysis. Ann Nutr Metab 58(4):290–296. https://doi.org/10.1159/000331214
Soedamah-Muthu SS, de Goede J (2018) Dairy consumption and cardiometabolic diseases: systematic review and updated meta-analyses of prospective cohort studies. Curr Nutr Rep 7(4):171–182. https://doi.org/10.1007/s13668-018-0253-y
Josse AR, Kendall CW, Augustin LS, Ellis PR, Jenkins DJ (2007) Almonds and postprandial glycemia—a dose-response study. Metabolism 56(3):400–404. https://doi.org/10.1016/j.metabol.2006.10.024
Tay J, Luscombe-Marsh ND, Thompson CH et al (2014) A very low-carbohydrate, low-saturated fat diet for type 2 diabetes management: a randomized trial. Diabetes Care 37(11):2909–2918. https://doi.org/10.2337/dc14-0845
Yamada Y, Uchida J, Izumi H et al (2014) A non-calorie-restricted low-carbohydrate diet is effective as an alternative therapy for patients with type 2 diabetes. Intern Med 53(1):13–19. https://doi.org/10.2169/internalmedicine.53.0861
van Zuuren EJ, Fedorowicz Z, Kuijpers T, Pijl H (2018) Effects of low-carbohydrate- compared with low-fat-diet interventions on metabolic control in people with type 2 diabetes: a systematic review including GRADE assessments. Am J Clin Nutr 108(2):300–331. https://doi.org/10.1093/ajcn/nqy096
Snorgaard O, Poulsen GM, Andersen HK, Astrup A (2017) Systematic review and meta-analysis of dietary carbohydrate restriction in patients with type 2 diabetes. BMJ Open Diabetes Res Care 5(1):e000354. https://doi.org/10.1136/bmjdrc-2016-000354
Meng Y, Bai H, Wang S, Li Z, Wang Q, Chen L (2017) Efficacy of low carbohydrate diet for type 2 diabetes mellitus management: a systematic review and meta-analysis of randomized controlled trials. Diabetes Res Clin Pract 131:124–131. https://doi.org/10.1016/j.diabres.2017.07.006
UK Prospective Diabetes Study (UKPDS) Group (1998) Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 352(9131):837–853
Gaede P, Vedel P, Larsen N, Jensen GV, Parving HH, Pedersen O (2003) Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N Engl J Med 348(5):383–393. https://doi.org/10.1056/NEJMoa021778
Pramfalk C, Pavlides M, Banerjee R et al (2016) Fasting plasma insulin concentrations are associated with changes in hepatic fatty acid synthesis and partitioning prior to changes in liver fat content in healthy adults. Diabetes 65(7):1858–1867. https://doi.org/10.2337/db16-0236
Schwarz JM, Linfoot P, Dare D, Aghajanian K (2003) Hepatic de novo lipogenesis in normoinsulinemic and hyperinsulinemic subjects consuming high-fat, low-carbohydrate and low-fat, high-carbohydrate isoenergetic diets. Am J Clin Nutr 77(1):43–50. https://doi.org/10.1093/ajcn/77.1.43
Donnelly KL, Smith CI, Schwarzenberg SJ, Jessurun J, Boldt MD, Parks EJ (2005) Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J Clin Invest 115(5):1343–1351. https://doi.org/10.1172/JCI23621
Miles JM, Nelson RH (2007) Contribution of triglyceride-rich lipoproteins to plasma free fatty acids. Horm Metab Res 39(10):726–729. https://doi.org/10.1055/s-2007-990273
Szczepaniak LS, Nurenberg P, Leonard D et al (2005) Magnetic resonance spectroscopy to measure hepatic triglyceride content: prevalence of hepatic steatosis in the general population. Am J Physiol Endocrinol Metab 288(2):E462–E468. https://doi.org/10.1152/ajpendo.00064.2004
van Werven JR, Marsman HA, Nederveen AJ et al (2010) Assessment of hepatic steatosis in patients undergoing liver resection: comparison of US, CT, T1-weighted dual-echo MR imaging, and point-resolved 1H MR spectroscopy. Radiology 256(1):159–168. https://doi.org/10.1148/radiol.10091790
Nasr P, Forsgren MF, Ignatova S et al (2017) Using a 3% proton density fat fraction as a cut-off value increases sensitivity of detection of hepatic steatosis, based on results from histopathology analysis. Gastroenterology 153(1):53–55. https://doi.org/10.1053/j.gastro.2017.03.005
Seppala-Lindroos A, Vehkavaara S, Hakkinen AM et al (2002) Fat accumulation in the liver is associated with defects in insulin suppression of glucose production and serum free fatty acids independent of obesity in normal men. J Clin Endocrinol Metab 87(7):3023–3028. https://doi.org/10.1210/jcem.87.7.8638
Begovatz P, Koliaki C, Weber K et al (2015) Pancreatic adipose tissue infiltration, parenchymal steatosis and beta cell function in humans. Diabetologia 58(7):1646–1655. https://doi.org/10.1007/s00125-015-3544-5
Heni M, Machann J, Staiger H et al (2010) Pancreatic fat is negatively associated with insulin secretion in individuals with impaired fasting glucose and/or impaired glucose tolerance: a nuclear magnetic resonance study. Diabetes Metab Res Rev 26(3):200–205. https://doi.org/10.1002/dmrr.1073
Feinman RD, Pogozelski WK, Astrup A et al (2015) Dietary carbohydrate restriction as the first approach in diabetes management: critical review and evidence base. Nutrition 31(1):1–13. https://doi.org/10.1016/j.nut.2014.06.011
Brouns F (2018) Overweight and diabetes prevention: is a low-carbohydrate-high-fat diet recommendable? Eur J Nutr 57(4):1301–1312. https://doi.org/10.1007/s00394-018-1636-y
Seidelmann SB, Claggett B, Cheng S et al (2018) Dietary carbohydrate intake and mortality: a prospective cohort study and meta-analysis. Lancet Public Health 3(9):e419–e428. https://doi.org/10.1016/S2468-2667(18)30135-X
Marckmann P, Osther P, Pedersen AN, Jespersen B (2015) High-protein diets and renal health. J Ren Nutr 25(1):1–5. https://doi.org/10.1053/j.jrn.2014.06.002
Nezu U, Kamiyama H, Kondo Y, Sakuma M, Morimoto T, Ueda S (2013) Effect of low-protein diet on kidney function in diabetic nephropathy: meta-analysis of randomised controlled trials. BMJ Open 3(5):e002934. https://doi.org/10.1136/bmjopen-2013-002934
Tay J, Thompson CH, Luscombe-Marsh ND et al (2015) Long-term effects of a very low carbohydrate compared with a high carbohydrate diet on renal function in individuals with type 2 diabetes: a randomized trial. Medicine 94(47):e2181. https://doi.org/10.1097/MD.0000000000002181
Tay J, Thompson CH, Luscombe-Marsh ND et al (2018) Effects of an energy-restricted low-carbohydrate, high unsaturated fat/low saturated fat diet versus a high-carbohydrate, low-fat diet in type 2 diabetes: a 2-year randomized clinical trial. Diabetes Obes Metab 20(4):858–871. https://doi.org/10.1111/dom.13164
Acknowledgements
The authors sincerely thank the study participants for their efforts in making this study possible. Further, we thank the kitchen personnel at the Department of Nutrition, Exercise and Sports, University of Copenhagen. Preliminary results from the present study were presented by MJS and AS as poster presentations at the 77th Scientific Session of the ADA, 2017, and at the 54th Annual Meeting of the EASD, 2018.
Funding
The study was funded by grants from: Arla Food for Health; the Novo Nordisk Foundation Center for Basic Metabolic Research, University of Copenhagen; the Department of Clinical Medicine, Aarhus University; the Department of Nutrition, Exercise and Sports, University of Copenhagen; and Copenhagen University Hospital, Bispebjerg. Dietary ingredients were partly provided by Arla Foods, JAN Import A/S, Royal Greenland and Danish Crown. The study sponsors, Arla Food for Health, Arla Foods, JAN Import A/S, Royal Greenland and Danish Crown, were informed, but not involved in the design of the study; the collection, analysis, and interpretation of data; writing the report; or the decision to submit the report for publication. Co-authors from the University of Copenhagen and Aarhus University were involved in the study, as detailed in the contribution statement.
Author information
Authors and Affiliations
Contributions
MJS and AS conducted the study, acquired and analysed the data, and drafted the manuscript. ADP and MNT contributed to acquisition of data. EC produced and analysed magnetic resonance data and contributed to drafting of the manuscript. AA, JF, JJH, HST, SM and TML contributed to the conception and the design of the study and contributed to the production and the interpretation of data. In addition, TML supervised the food production and distribution. SBH and TK conceived and designed the study, obtained funding, supervised the study and contributed to interpretation of data. TK is the guarantor of the manuscript. All authors critically reviewed the manuscript and approved the version to be published.
Corresponding author
Ethics declarations
AA is a member of advisory boards/consultant for: BioCare Copenhagen, Denmark; the Dutch Beer Institute, the Netherlands; Gelesis, USA; Groupe Éthique et Santé, France; McCain Foods Limited, USA; Navamedic, Denmark; Novo Nordisk, Denmark; Pfizer, USA; Saniona, Denmark; Weight Watchers, USA; and is a recipient of travel grants and honoraria as speaker for a wide range of Danish and international concerns. AA is co-owner and a member of the board of the consultancy company Dentacom ApS, Denmark; co-founder and co-owner of UCPH spin-outs Mobile Fitness A/S, Flaxslim ApS and Personalized Weight Management Research Consortium ApS (Gluco-diet.dk). AA is co-inventor of a number of patents owned by UCPH, in accordance with Danish law. AA is co-author of a number of diet and cookery books, including books on personalised diet. AA is not an advocate or activist for specific diets, and is not strongly committed to any specific diet, e.g. veganism, Atkins diet, gluten-free diet, high-animal protein diet or dietary supplements. TML is an advisor for ‘Sense’ diet programme. None of the other authors have conflicts of interest to declare.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM
(PDF 115 kb)
Rights and permissions
About this article
Cite this article
Skytte, M.J., Samkani, A., Petersen, A.D. et al. A carbohydrate-reduced high-protein diet improves HbA1c and liver fat content in weight stable participants with type 2 diabetes: a randomised controlled trial. Diabetologia 62, 2066–2078 (2019). https://doi.org/10.1007/s00125-019-4956-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-019-4956-4