Abstract
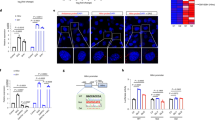
In a previous report from our laboratory, it was reported that hepatic levels of the long non-coding RNA (lncRNA), H19 are decreased in diabetic mice which elevates hepatic gluconeogenesis and glucose output. But, the mechanisms of H19 inhibition in elevating gluconeogenic genes’ transcription and promoting hepatic glucose output were not known. In this study, we aimed to decipher this regulatory role of H19 on glucose metabolism and on FoxO1, an important transcriptional regulator of gluconeogenesis. While H19 inhibition in HepG2 cells increased the levels of FoxO1, its overexpression led to significant inhibition in FoxO1 levels, thereby identifying H19 as an important regulator of FoxO1. Our data also demonstrates that in the absence of H19, there is increased occupancy of p53 on the FoxO1 promoter that possibly is responsible for increased FoxO1 transcription. In vivo silencing of H19 in normal mice caused hyperglycemia, hyperinsulinemia and impaired glucose, insulin, and pyruvate tolerance. Serum triglyceride and cholesterol levels, however, did not show any change. H19 inhibition significantly elevated the hepatic levels of FoxO1 and all the gluconeogenic genes. While fasting increased gluconeogenic genes’ transcription, the levels of H19 were decreased and these patterns reversed upon refeeding the mice. Thus, gluconeogenic genes and H19 levels show inverse patterns of expression, and these results indicate towards an important regulatory role of the lncRNA, H19. It acts as an upstream regulator of gluconeogenesis by regulating the transcription of FoxO1, an important transcription factor of gluconeogenic genes, and hence, regulates hepatic glucose metabolism.
Key messages
-
H19 regulates FoxO1 transcript and protein levels.
-
H19 inhibition increases p53 occupancy on the FoxO1 promoter that promotes FoxO1 transcription.
-
H19 inhibition in vivo induces hyperglycemia and impairs glucose, insulin, and pyruvate tolerance.
-
In vivo H19 inhibition increases the hepatic transcript levels of gluconeogenic genes and FoxO1.
-
Transcript levels of H19 and gluconeogenic genes are inversely regulated during fed and fasted states.






Similar content being viewed by others
References
Magnusson I, Rothman DL, Katz LD, Shulman RG, Shulman GI (1992) Increased rate of gluconeogenesis in type II diabetes mellitus. A 13C nuclear magnetic resonance study. J Clin Invest 90:1323–1327
Basu R, Schwenk WF, Rizza RA (2004) Both fasting glucose production and disappearance are abnormal in people with “mild” and “severe” type 2 diabetes. Am J Physiol Endocrinol Metab 287:E55–E62
Michael MD, Kulkarni RN, Postic C, Previs SF, Shulman GI, Magnuson MA, Kahn CR (2000) Loss of insulin signaling in hepatocytes leads to severe insulin resistance and progressive hepatic dysfunction. Mol Cell 6:87–97
Gastaldelli A, Miyazaki Y, Pettiti M, Buzzigoli E, Mahankali S, Ferrannini E, DeFronzo RA (2004) Separate contribution of diabetes, total fat mass, and fat topography to glucose production, gluconeogenesis, and glycogenolysis. J Clin Endocrinol Metab 89:3914–3921
Oh KJ, Han HS, Kim MJ, Koo SH (2013) CREB and FoxO1: two transcription factors for the regulation of hepatic gluconeogenesis. BMB Rep 46:567–574
Yoon JC, Puigserver P, Chen G, Donovan J, Wu Z, Rhee J, Adelmant G, Stafford J, Kahn CR, Granner DK, Newgard CB, Spiegelman BM (2001) Control of hepatic gluconeogenesis through the transcriptional coactivator PGC-1. Nature 413:131–138
Goyal N, Sivadas A, Shamsudheen KV, Jayarajan R, Verma A, Sivasubbu S, Scaria V, Datta M (2017) RNA sequencing of db/db mice liver identifies lncRNA H19 as a key regulator of gluconeogenesis and hepatic glucose output. Sci Rep 7:8312
Gabory A, Jammes H, Dandolo L (2010) The H19 locus: role of an imprinted non-coding RNA in growth and development. Bioessays 32:473–480
Brannan CI, Dees EC, Ingram RS, Tilghman SM (1990) The product of the H19 gene may function as an RNA. Mol Cell Biol 10:28–36
Shao WJ, Tao LY, Gao C, Xie JY, Zhao RQ (2008) Alterations in methylation and expression levels of imprinted genes H19 and Igf2 in the fetuses of diabetic mice. Comp Med 58:341–346
Ding GL, Wang FF, Shu J, Tian S, Jiang Y, Zhang D, Wang N, Luo Q, Zhang Y, Jin F, Leung PCK, Sheng JZ, Huang HF (2012) Transgenerational glucose intolerance with Igf2/H19 epigenetic alterations in mouse islet induced by intrauterine hyperglycemia. Diabetes 61:1133–1142
Venkatraman A, He XC, Thorvaldsen JL, Sugimura R, Perry JM, Tao F, Zhao M, Christenson MK, Sanchez R, Yu JY, Peng L, Haug JS, Paulson A, Li H, Zhong XB, Clemens TL, Bartolomei MS, Li L (2013) Maternal imprinting at the H19-Igf2 locus maintains adult haematopoietic stem cell quiescence. Nature 500:345–349
Barsyte-Lovejoy D, Lau SK, Boutros PC, Khosravi F, Jurisica I, Andrulis IL, Tsao MS, Penn LZ (2006) The c-Myc oncogene directly induces the H19 noncoding RNA by allele-specific binding to potentiate tumorigenesis. Cancer Res 66:5330–5337
Berteaux N, Lottin S, Monte D, Pinte S, Quatannens B, Coll J, Hondermarck H, Curgy JJ, Dugimont T, Adriaenssens E (2005) H19 mRNA-like noncoding RNA promotes breast cancer cell proliferation through positive control by E2F1. J Biol Chem 280:29625–29636
Matouk IJ, DeGroot N, Mezan S, Ayesh S, Abu-lail R, Hochberg A, Galun E (2007) The H19 non-coding RNA is essential for human tumor growth. PLoS One 2:e845. https://doi.org/10.1371/journal.pone.0000845
Gao Y, Wu F, Zhou J, Yan L, Jurczak MJ, Lee HY, Yang L, Mueller M, Zhou XB, Dandolo L, Szendroedi J, Roden M, Flannery C, Taylor H, Carmichael GG, Shulman GI, Huang Y (2014) The H19/let-7 double-negative feedback loop contributes to glucose metabolism in muscle cells. Nucleic Acids Res 42:13799–13811
Zhang W, Patil S, Chauhan B, Guo S, Powell DR, Le J, Klotsas A, Matika R, Xiao X, Franks R et al (2006) FoxO1 regulates multiple metabolic pathways in the liver: effects on gluconeogenic, glycolytic, and lipogenic gene expression. J Biol Chem 281:10105–10117
Goldstein I, Yizhak K, Madar S, Goldfinger N, Ruppin E, Rotter V (2013) p53 promotes the expression of gluconeogenesis-related genes and enhances hepatic glucose production. Cancer Metab 1(1):9
Kung CP, Murphy ME (2016) The role of the p53 tumor suppressor in metabolism and diabetes. J Endocrinol, 231:R61–R75
Pope C, Mishra S, Russell J, Zhou Q, Zhong X-B (2017) Targeting H19, an imprinted long non-coding RNA, in hepatic functions and liver diseases. Diseases 5:11
Iempridee T (2017) Long non-coding RNA H19 enhances cell proliferation and anchorage-independent growth of cervical cancer cell lines. Exp Biol Med 242:184–193
Wang S, Wu X, Liu Y, Yuan J, Yang F, Huang J, Meng Q, Zhou C, Liu F, Ma J, Sun S, Zheng J, Wang F (2016) Long noncoding RNA H19 inhibits the proliferation of fetal liver cells and the Wnt signaling pathway. FEBS Lett 590:559–570
Li X, Gray SG, Flam F, Pietsch T, Ekstrom TJ (1998) Developmental-dependent DNA methylation of the IGF2 and H19 promoters is correlated to the promoter activities in human liver development. Int J Dev Biol 42:687–693
Le F, Wang LY, Wang N, Li L, Li le J, Zheng YM, Lou HY, Liu XZ, Xu XR, Sheng JZ et al (2013) In vitro fertilization alters growth and expression of Igf2/H19 and their epigenetic mechanisms in the liver and skeletal muscle of newborn and elder mice. Biol Reprod 88:75
Leighton PA, Ingram RS, Eggenschwiler J, Efstratiadis A, Tilghman SM (1995) Disruption of imprinting caused by deletion of the H19 gene region in mice. Nature 375:34–39
Eriksson JG, Forsen TJ, Osmond C, Barker DJ (2003) Pathways of infant and childhood growth that lead to type 2 diabetes. Diabetes Care 26:3006–3010
Nilsson E, Matte A, Perfilyev A, de Mello VD, Kakela P, Pihlajamaki J, Ling C (2015) Epigenetic alterations in human liver from subjects with type 2 diabetes in parallel with reduced folate levels. J Clin Endocrinol Metab 100:E1491–E1501
Murphy R, Ibáñez L, Hattersley A, Tost J (2012) IGF2/H19 hypomethylation in a patient with very low birthweight, preocious pubarche and insulin resistance. BMC Med Genet 13:42
Jitrapakdee S (2012) Transcription factors and coactivators controlling nutrient and hormonal regulation of hepatic gluconeogenesis. Int J Biochem Cell Biol 44:33–45
Matsumoto M, Pocai A, Rossetti L, Depinho RA, Accili D (2007) Impaired regulation of hepatic glucose production in mice lacking the forkhead transcription factor Foxo1 in liver. Cell Metab 6:208–216
Puigserver P, Rhee J, Donovan J, Walkey CJ, Yoon JC, Oriente F, Kitamura Y, Altomonte J, Dong H, Accili D, Spiegelman BM (2003) Insulin-regulated hepatic gluconeogenesis through FOXO1-PGC-1alpha interaction. Nature 423:550–555
Abdul-Wahed A, Gautier-Stein A, Casteras S, Soty M, Roussel D, Romestaing C, Guillou H, Tourette JA, Pleche N, Zitoun C, Gri B, Sardella A, Rajas F, Mithieux G (2014) A link between hepatic glucose production and peripheral energy metabolism via hepatokines. Mol Metab 3:531–543
Xu X, Ji S, Li W, Yi B, Li H, Zhang H, Ma W (2017) LncRNA H19 promotes the differentiation of bovine skeletal muscle satellite cells by suppressing Sirt1/FoxO1. Cell Mol Biol Lett 22:10
Geisler CE, Hepler C, Higgins MR, Renquist BJ (2016) Hepatic adaptations to maintain metabolic homeostasis in response to fasting and refeeding in mice. Nutr Metab (Lond) 13:62
Acknowledgments
The authors acknowledge CSIR (NG and DK) and UGC (ST), New Delhi, India, for the fellowships. The authors thank the team of the Animal House facility of CSIR-IGIB for the assistance during the in vivo experiments. We thank Mr. Ananda K Mukherjee, CSIR-Institute of Genomics and Integrative Biology for his help in the chromatin-immunoprecipitation experiments.
Funding
This work was supported by funding from the Council of Scientific and Industrial Research (CSIR), New Delhi, India (BSC0123).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Neha Goyal and Shweta Tiwary are co-first authors
Rights and permissions
About this article
Cite this article
Goyal, N., Tiwary, S., Kesharwani, D. et al. Long non-coding RNA H19 inhibition promotes hyperglycemia in mice by upregulating hepatic FoxO1 levels and promoting gluconeogenesis. J Mol Med 97, 115–126 (2019). https://doi.org/10.1007/s00109-018-1718-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-018-1718-6