Abstract
Purpose
Postoperative quadriceps muscle strength was lowered after tourniquet application during total knee arthroplasty (TKA). Furthermore, tourniquet application results in higher proteolytic activity within vastus medialis cells, without influence on the amount and function of mitochondria. The effects of the commonly utilized intraoperative tourniquet on gene expression within the human skeletal muscle cells are barely examined. The purpose of the present study was to analyze the gene expression within the skeletal muscle cells after tourniquet-induced ischemia to identify differential expressed genes (DEGs) and pathways.
Methods
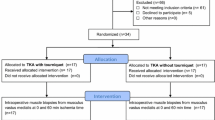
As part of a randomized, controlled, monocentric trial (Clinical-Trials.gov NCT02475603) 20 patients, scheduled to undergo primary total knee arthroplasty (TKA), were included. Written informed consent was received and the patients were randomly assigned to Group A (TKA with tourniquet) (n = 10) and Group B (TKA without tourniquet) (n = 10). A muscle biopsie of (5 × 5 × 5 mm) 125 mm3 were obtained from the vastus medialis exactly 60 min after performing the surgical approach.
After preparation of a muscle homogenate, RNA extraction was performed (RNeasy Plus Universal Mini Kit Qiagen) and RNA integrity (RIN) was determined (Agilent 2100 Bioanalyzer, RNA 6000 Pico Kit). Gene expression profiling was performed using a validated method (GeneChip™ Human Transcriptome Array 2.0; Affymetrix). Statistical analysis (SPSS-Version 24; SAS JMP10 Genomics, Version 6) included the number of significant DEGs (p < 0.05), the number of DEGs with relative difference > 25% and the number of significant pathway (p < 0.05). The serum C-reactive protein (CRP) and the white blood cell (WBC) count were also perioperatively measured. The protocol was approved by our Institutional Ethics Committee (File reference 2012-334N-MA).
Results
Tourniquet application resulted in a total of 3555 (13.8%) statistically significant DEGs within vastus medialis cells. 76 DEGs (29 upregulated, 47 downregulated) revealed a relative difference of more than 25%. Statistically significant changes occurred in 59 (25.8%) of 229 analyzed pathways. Furthermore, there was no clinically meaningful difference between the groups with regard to CRP and WBC count.
Conclusions
Tourniquet induced ischemia results in significant changes of the gene expression within cells of vastus medialis including metabolism, genetic information processing and cellular processes. The identified altered expression of genes and pathways might serve as pharmacotherapeutical targets; although further research is needed to clarify the underlying biological processes.
Clinical relevance
These findings add further knowledge and should raise the awareness of surgeons about the effects of tourniquet induced ischemia at the gene expression level. Additional high-quality research may be warranted to examine the short and long term clinical significance of the present data.
Level of evidence
Level I.

Similar content being viewed by others
Data availability
The raw and normalized data are deposited in the Gene Expression Omnibus database (https://www.ncbi.nlm.nih.gov/geo/; accession no. GSE124595).
References
Ratchford SM, Bailey AN, Senesac HA, Hocker AD, Smolkowski K, Lantz BA, et al. Proteins regulating cap-dependent translation are downregulated during total knee arthroplasty. Am J Physiol Regul Integr Comp Physiol. 2012;302(6):R702–R711711.
Mizner RL, Snyder-Mackler L. Altered loading during walking and sit-to-stand is affected by quadriceps weakness after total knee arthroplasty. J Orthop Res Off Publ Orthop Res Soc. 2005;23(5):1083–90.
Finch E, Walsh M, Thomas SG, Woodhouse LJ. Functional ability perceived by individuals following total knee arthroplasty compared to age-matched individuals without knee disability. J Orthop Sports Phys Ther. 1998;27(4):255–63.
Mizner RL, Petterson SC, Snyder-Mackler L. Quadriceps strength and the time course of functional recovery after total knee arthroplasty. J Orthop Sports Phys Ther. 2005;35(7):424–36.
Walsh M, Woodhouse LJ, Thomas SG, Finch E. Physical impairments and functional limitations: a comparison of individuals 1 year after total knee arthroplasty with control subjects. Phys Ther. 1998;78(3):248–58.
Yoshida Y, Mizner RL, Ramsey DK, Snyder-Mackler L. Examining outcomes from total knee arthroplasty and the relationship between quadriceps strength and knee function over time. Clin Biomech. 2008;23(3):320–8.
Dennis DA, Kittelson AJ, Yang CC, Miner TM, Kim RH, Stevens-Lapsley JE. Does tourniquet use in TKA affect recovery of lower extremity strength and function? A randomized trial. Clin Orthop Relat Res. 2015;474:4393–8.
Li C, Zeng Y, Shen B, Kang P, Yang J, Zhou Z, et al. A meta-analysis of minimally invasive and conventional medial parapatella approaches for primary total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc Off J ESSKA. 2015;23(7):1971–85.
Kvederas G, Porvaneckas N, Andrijauskas A, Svensen CH, Ivaskevicius J, Mazunaitis J, et al. A randomized double-blind clinical trial of tourniquet application strategies for total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc Off J ESSKA. 2013;21(12):2790–9.
Lohmann-Jensen R, Holsgaard-Larsen A, Emmeluth C, Overgaard S, Jensen C. The efficacy of tourniquet assisted total knee arthroplasty on patient-reported and performance-based physical function: a randomized controlled trial protocol. BMC Musculoskelet Disord. 2014;15:110.
Harris LW, Shadgan B, Reid D, Powers SK, O’Brien PJ. Tourniquet-induced skeletal muscle oxidative injury during lower limb orthopaedic trauma surgery. Bone Jt J. 2011;93-B(IV):557.
Meier WA, Marcus RL, Dibble LE, Foreman KB, Peters CL, Mizner RL, et al. The long-term contribution of muscle activation and muscle size to quadriceps weakness following total knee arthroplasty. J Geriatr Phys Ther. 2009;32(2):79–82.
Bailey AN, Hocker AD, Vermillion BR, Smolkowski K, Shah SN, Jewett BA, et al. MAFbx, MuRF1, and the stress-activated protein kinases are upregulated in muscle cells during total knee arthroplasty. Am J Physiol Regul Integr Comp Physiol. 2012;303(4):R376–R386386.
Jawhar A, Hermanns S, Ponelies N, Obertacke U, Roehl H. Tourniquet-induced ischaemia during total knee arthroplasty results in higher proteolytic activities within vastus medialis cells: a randomized clinical trial. Knee Surg Sports Traumatol Arthrosc Off J ESSKA. 2016;24(10):3313–21.
Jawhar A, Ponelies N, Schild L. Effect of limited ischemia time on the amount and function of mitochondria within human skeletal muscle cells. Eur J Trauma Emerg Surg Off Publ Eur Trauma Soc. 2016;42(6):767–73.
Muyskens JB, Hocker AD, Turnbull DW, Shah SN, Lantz BA, Jewett BA, et al. Transcriptional profiling and muscle cross-section analysis reveal signs of ischemia reperfusion injury following total knee arthroplasty with tourniquet. Physiol Rep. 2016;4(1):e12671.
Cheng YJ, Chien CT, Chen CF. Oxidative stress in bilateral total knee replacement, under ischaemic tourniquet. J Bone Jt Surg Br. 2003;85(5):679–82.
Murphy T, Walsh PM, Doran PP, Mulhall KJ. Transcriptional responses in the adaptation to ischaemia-reperfusion injury: a study of the effect of ischaemic preconditioning in total knee arthroplasty patients. J Transl Med. 2010;8:46.
Dai M, Wang P, Boyd AD, Kostov G, Athey B, Jones EG, et al. Evolving gene/transcript definitions significantly alter the interpretation of GeneChip data. Nucleic Acids Res. 2005;33(20):e175.
Team RC. R: a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing; 2017.
Team R. RStudio: integrated development for R. Boston: RStudio Inc; 2015.
Sergushichev A. An algorithm for fast preranked gene set enrichment analysis using cumulative statistic calculation. bioRXiv. 2016. https://doi.org/10.1101/060012.
Paoni NF, Peale F, Wang F, Errett-Baroncini C, Steinmetz H, Toy K, et al. Time course of skeletal muscle repair and gene expression following acute hind limb ischemia in mice. Physiol Genom. 2002;11(3):263–72.
Westman B, Weidenhielm L, Rooyackers O, Fredriksson K, Wernerman J, Hammarqvist F. Knee replacement surgery as a human clinical model of the effects of ischaemia/reperfusion upon skeletal muscle. Clin Sci. 2007;113(7):313–8.
Sjoholm H, Gidlof A, Larsson J, Sahlin K. The effect of long-term circulatory occlusion on pH and energy metabolism of the quadriceps muscle in man. Clin Sci. 1985;68(5):597–600.
Mathru M, Dries DJ, Barnes L, Tonino P, Sukhani R, Rooney MW. Tourniquet-induced exsanguination in patients requiring lower limb surgery. An ischemia-reperfusion model of oxidant and antioxidant metabolism. Anesthesiology. 1996;84(1):14–22.
Ouellet D, Moffet H. Locomotor deficits before and two months after knee arthroplasty. Arthritis Rheum. 2002;47(5):484–93.
Hocker AD, Boileau RM, Lantz BA, Jewett BA, Gilbert JS, Dreyer HC. Endoplasmic reticulum stress activation during total knee arthroplasty. Physiol Rep. 2013;1(3):e00052.
Sha Y, Xu YQ, Zhao WQ, Tang H, Li FB, Li X, et al. Protective effect of ischaemic preconditioning in total knee arthroplasty. Eur Rev Med Pharmacol Sci. 2014;18(10):1559–666.
Tsunoda K, Sonohata M, Kugisaki H, Someya S, Honke H, Komine M, et al. The effect of air tourniquet on interleukin-6 levels in total knee arthroplasty. Open Orthop J. 2017;11:20–8.
Acknowledgements
We thank our staff of the management office for the randomisation of the patients.
Funding
This study was supported by the Medical Faculty Mannheim, Heidelberg University, Germany.
Author information
Authors and Affiliations
Contributions
Conceptualization: AJ, NP, UO. Data curation: AJ, DB, CT, NP. Methodology: AJ, DB, CS, CT, NP. Project administration: AJ, NP, UO. Resources: AJ, DB, CS, CT, NP, UO. Software: CS, CT. Supervision: AJ, NP, UO. Writing—original draft: AJ. Writing—review/editing: NP, AJ.
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no competing interests exist.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jawhar, A., Brenner, D., De La Torre, C. et al. Gene expression analysis of vastus medialis cells after tourniquet-induced ischemia during total knee arthroplasty: a randomized clinical trial. Eur J Trauma Emerg Surg 47, 233–240 (2021). https://doi.org/10.1007/s00068-019-01196-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00068-019-01196-0