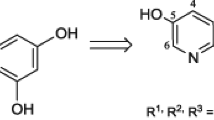
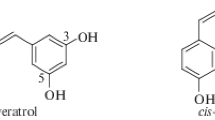
Abstract
To continue the research on the preparation of resveratrol structural analogs containing the 3-pyridinol fragment, a series of derivatives with an ethyl radical sterically shielding the hydroxyl group was synthesized. It was shown that the ethyl group introduction has an ambiguous effect on the radical scavenging and antioxidant properties of the stilbazoles studied, which is probably related to the structural features of the resulting radical intermediates. The correlation between the radical scavenging and antioxidant properties of the derivatives studied is established. A number of compounds have been identified that exhibit an antioxidant effect on the mitochondrial membranes lipid peroxidation model, better than the natural prototype and 2-ethyl-6-methylpyridin-3-ol with a related structure used in clinical practice.








Similar content being viewed by others
References
Berman AY, Motechin RA, Wiesenfeld MY, Holz MK (2017) The therapeutic potential of resveratrol: a review of clinical trials. NPJ Precis Oncol 1:35
Bhatti JS, Bhatti GK, Reddy PH (2017) Mitochondrial dysfunction and oxidative stress in metabolic disorders—a step towards mitochondria based therapeutic strategies. Biochim Biophys Acta 1863:1066–1077
Bonnefont-Rousselot D (2016) Resveratrol and cardiovascular diseases. Nutrients 8:250–273
Chesnokova NB, Beznos OV, Pavlenko TA, Zabozlaev AA, Pavlova MV (2015) Effects of hydroxypyridine derivatives mexidol and emoxypin on the reparative processes in rabbit eye on the models of corneal epithelial defect and conjunctival ischemia. Bull Exp Biol Med 158:346–348
Corcelli A, Saponetti MS, Zaccagnino P, Lopalco P, Mastrodonato M, Liquori GE, Lorusso M (2010) Mitochondria isolated in nearly isotonic KCl buffer: focus on cardiolipin and organelle morphology. Biochim Biophys Acta 1798:681–687
Daina A, Zoete V (2016) A BOILED-Egg to predict gastrointestinal absorption and brain penetration of small molecules. ChemMedChem 11:1117–1121
Devasagayam TPA, Pushpendran CK, Eapen J (1983) Differences in lipid peroxidation of rat liver rough and smooth microsomes. Biochim Biophys Acta 750:91–97
Egan WJ, Merz Jr. KM, Baldwin JJ (2000) Prediction of drug absorption using multivariate statistics. J Med Chem 43:3867–3877
Elimadi A, Bouillot L, Sapena R, Tillement J-P, Morin D (1998) Dose-related inversion of cinnarizine and flunarizine effects on mitochondrial permeability transition. Eur J Pharm 348:115–121
Ertl P, Rohde B, Selzer P (2000) Fast calculation of molecular polar surface area as a sum of fragment-based contributions and its application to the prediction of drug transport properties. J Med Chem 43:3714–3717
Fauconneau B, Waffo-Teguo P, Huguet F, Barrier L, Decendit A, Merillon J-M (1997) Comparative study of radical scavenger and antioxidant properties of phenolic compounds from vitis vinifera cell cultures using in vitro tests. Life Sci 61:2103–2110
Garipov MR, Strelnik AD, Shtyrlin NV, Nagimova AI, Naumov AK, Morozov OA, Balakin KV, Shtyrlin YG (2018) Synthesis and nonlinear optical properties of pyridoxine-based stilbazole derivatives and their azo-analogs. Synth Commun 48:768–777
Gülçin I (2010) Antioxidant properties of resveratrol: a structure-activity insight. Innov Food Sci Emerg Technol 11:210–218
He F (2011) Bradford protein assay. Bio 101:e45
Indo HP, Yen H-C, Nakanishi I, Matsumoto K, Tamura M, Nagano Y, Matsui H, Gusev O, Cornette R, Okuda T, Minamiyama Y, Ichikawa H, Suenaga S, Oki M, Sato T, Ozawa T, Clair DKS, Majima HJ (2015) A mitochondrial superoxide theory for oxidative stress diseases and aging. J Clin Biochem Nutr 56:1–7
Jardim FR, de Rossi FT, Nascimento MX, da Silva Barros RG, Borges PA, Prescilio IC, de Oliveira MR (2018) Resveratrol and brain mitochondria: a review. Mol Neurobiol 55:2085–2101
Khan OS, Bhat AA, Krishnankutty R, Mohammad RM, Uddin S (2016) Therapeutic potential of resveratrol in lymphoid malignancies. Nutr Cancer 68:365–373
Khanduja KL, Bhardwaj A (2003) Stable free radical scavenging and antiperoxidative properties of resveratrol compared in vitro with some other bioflavonoids. Indian J Biochem Biophys 40:416–22
Li Q-S, Li Y, Deora GS, Ruan B-F (2019) Derivatives and analogues of resveratrol: recent advances in structural modification. Mini-Rev Medicinal Chem 19:809–825
Markus MA, Morris BJ (2008) Resveratrol in prevention and treatment of common clinical conditions of aging. Clin Inter Aging 3:331–339
Pisoschi AM, Pop A (2015) The role of antioxidants in the chemistry of oxidative stress: a review. Eur J Med Chem 97:55–74
Poulsen MM, Fjeldborg K, Ornstrup MJ, Kjær TN, Nøhr MK, Pedersen SB (2015) Resveratrol and inflammation: Challenges in translating pre-clinical findings to improved patient outcomes. Biochim Biophys Acta 1852:1124–1136
Ray PD, Huang B-W, Tsuji Y (2012) Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell Signal 24:981–990
Semenov AV, Balakireva OI, Tarasova IV, Burtasov AA, Semenova EV, Petrov PS, Minaeva OV, Pyataev NA (2018) Synthesis, theoretical, and experimental study of radical scavenging activity of 3-pyridinol containing trans-resveratrol analogs. Med Chem Res 27:1298–1308
Semenov AV, Balakireva OI, Tarasova IV, Semenova EV, Minaeva OV (2019) Spectroscopic properties of some hydroxylated 2-stilbazole derivatives. J Fluoresc 29:1301–1309
Sies H, Berndt C, Jones DP (2017) Oxidative stress. Annu Rev Biochem 86:715–748
Singh N, Agrawal M, Doré S (2013) Neuroprotective properties and mechanisms of resveratrol in in vitro and in vivo experimental cerebral stroke models. ACS Chem Neurosci 4:1151–1162
Wakabayashi S, Kiyohara Y, Kameda S, Uenishi J, Oae S (1990) Ligand coupling of 2-pyridyl sulfoxides having an sp2 stereocenter at the α-position: a novel preparation of α-stilbazoles. Heteroat Chem 1:225–232
Westphal CH, Dipp MA, Guarente L (2007) A therapeutic role for sirtuins in diseases of aging? Trends Biochem Res 32:555–560
Funding
The reported study was funded by RFBR according to the research project No. 18-43-130004.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Semenov, A.V., Balakireva, O.I., Tarasova, I.V. et al. Synthesis, radical scavenging, and antioxidant activity of stilbazolic resveratrol analogs. Med Chem Res 29, 1590–1599 (2020). https://doi.org/10.1007/s00044-020-02585-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-020-02585-6