Abstract
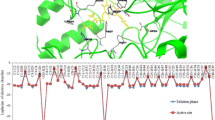
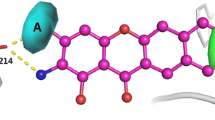
This study has been carried out to understand the nature of conformational flexibility and electrostatic properties of polyhydroxyl coumarins derivatives. When these compounds present in the active site of S. cerevisiae α-glucosidase, the lactone rings of the molecules are flanking out, while all benzene rings are embedded deep inside the binding cavity. These hydroxyl groups can interact with the surrounding amino acids by hydrogen bonds easily. When the hydroxyl groups at the C9 of benzene ring are replaced by methoxy groups, there is no evident influence on the hydrogen bonding interactions with the surrounding amino acid Asp68 and Lys155. However, their Laplacian values of electron densities of hydroxyl O–H bonds are obviously decreased in the active site, which suggests concentrated electron densities. In general, most of the electron densities of chemical bonds become more depleted after docking with the S. cerevisiae α-glucosidase, implying strong interactions with the surrounding amino acids. For polyhydroxyl coumarin derivatives, the global maximum values of the molecular electrostatic potential on molecular vdW surfaces stem from hydrogen atoms of the hydroxyl groups. However, the values are decreased evidently and stem from the different atoms in both phases while methoxy group is introduced. These fine details at electronic level allow to better understand the exact interactions between natural coumarins derivatives and target protein.





Similar content being viewed by others
References
Arnold K, Bordoli L, Kopp J, Schwede T (2006) The SWISS-MODEL workspace: a web-based environment for protein structure homology modeling. Bioinformatics 22:195–201
Azhagesan Renuga P, Poomani K (2013) Exploring the conformation, charge density distribution and the electrostatic properties of galanthamine molecule in the active site of AChE using DFT and AIM theory. Int J Quant Chem 113:1200–1208
Bader RFW (1990) Atoms in molecules: a quantum theory. Oxford University Press, Oxford
Bischoff H (1995) The mechanism of alpha-glucosidase inhibition in the management of diabetes. Clin Invest Med 18(4):303–311
Chaudhry J, Ghosha NN, Roya K, Chandra R (2007) Antihyperglycemic effect of a new thiazolidinedione analog and its role in ameliorating oxidative stress in alloxan-induced diabetic rats. Life Sci 80:1135–1142
Chen H, Ouyang K, Jiang Y, Yang Z, Hu W, Xiong L, Wang N, Liu X, Wang W (2017) Constituent analysis of the ethanol extracts of Chimonanthus nitens Oliv. leaves and their inhibitory effect on α-glucosidase activity. Int J Biol Macromol 98:829–836
Chen J, Mangelinckx S, Ma L, Wang Z, Li W, De Kimpe N (2014) Caffeoylquinic acid derivatives isolated from the aerial parts of Gynura divaricata and their yeast α-glucosidase and PTP1B inhibitory activity. Fitoterapia 99:1–6
Chukwujekwu JC, Rengasamy KR, de Kock CA, Smith PJ, Slavětínská LP, van Staden J (2016) Alpha-glucosidase inhibitory and antiplasmodial properties of terpenoids from the leaves of Buddleja saligna Willd. J Enzyme Inhib Med Chem 31(1):63–66
da Silva Gonçalves A, França TC, Caetano MS, Ramalho TC (2014) Reactivation steps by 2-PAM of tabun-inhibited human acetylcholinesterase: reducing the computational cost in hybrid QM/MM methods. J Biomol Struct Dyn 32:301–307
de Lima WEA, Pereira AF, de Castro AA, da Cunha EFF, Ramalho TC (2016) Flexibility in the molecular design of acetylcholinesterase reactivators: probing representative conformations by chemometric techniques and docking/QM calculations. Lett Drug Des Discov 13:360–371
Delgado A, Corni S, Pittalis S, Rozzi CA (2015) Modeling solvation effects in real-space and real-time within density functional approaches. J Chem Phys 143(14):144111
Devipriya B, Kumaradhas P (2013) Molecular flexibility and the electrostatic moments of curcumin and its derivatives in the active site ofp300: a theoretical charge density study. Chem Biol Interact 204:153–156
Espinoza-Hicks JC, Nápoles-Duarte JM, Nevárez-Moorillón GV, Camacho-Dávila A, Rodríguez-Valdez LM (2016) Synthesis, electronic, and spectral properties of novel geranylated chalcone derivatives: a theoretical and experimental study. J Mol Model 22(10):253
Etxeberria U, de la Garza AL, Campión J, Martínez JA, Milagro FI (2012) Antidiabetic effects of natural plant extracts via inhibition of carbohydrate hydrolysis enzymes with emphasis on pancreatic alpha amylase. Expert Opin Ther Targets 16:269–297
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ. Gaussian 09, Revision E.01 (2009) Gaussian, Inc. Wallingford CT
Guisasola EEB, Gutierrez LJ, Salcedo RE, Garibotto FM, Andujar SA, Enriz RD, Rodriguez AM (2016) Conformational transition of A beta(42) inhibited by a mimetic peptide. A molecular modeling study using QM/MM calculations and QTAIM analysis. Comput Theor Chem 1080:56–65
Hibbs DE, Hanrahan JR, Hursthouse MB, Knight DW, Overgaard J, Turner P, Piltz RO, Waller MP (2003) Experimental and theoretical charge distribution in (Z)-N-methyl-C-phenylnitrone. Org Biomol Chem 1(6):1034
Hwang SH, Wang Z, Yoon HN, Lim SS (2016) Xanthium strumarium as an inhibitor of α-glucosidase, protein tyrosine phosphatase 1β, protein glycation and ABTS+ for diabetic and its complication. Molecules 1(9):pii: E1241
Jeyaveeran JC, Praveen C, Arun Y, Prince AAM, Perumal PT (2016) Flexible synthesis of isomeric pyranoindolones and evaluation of cytotoxicity towards HeLa cells. J Chem Sci 128:787–802
Kalra S (2014) Alpha glucosidase inhibitors. J Pak Med Assoc 64:474–476
Kessler J, Dračínský M, Bouř P (2013) Parallel variable selection of molecular dynamics clusters as a tool for calculation of spectroscopic properties. J Comput Chem 34:366–371
Khan KM, Qurban S, Salar U, Taha M, Hussain S, Perveen S, Hameed A, Ismail NH, Riaz M, Wadood A (2016) Synthesis, in vitro α-glucosidase inhibitory activity and molecular docking studies of new thiazole derivatives. Bioorg Chem 68:245–258
Kiefer F, Arnold K, Künzli M, Bordoli L, Schwede T (2009) The SWISS-MODEL repository and associated resources. Nucleic Acids Res 37:D387–D392
Kumar S, Narwal S, Kumar V, Prakash O (2011) α-Glucosidase inhibitors from plants: a natural approach to treat diabetes. Pharmacogn Rev 5(9):19–29
Li H, Yao Y, Li L (2017) Coumarins as potential antidiabetic agents. J Pharm Pharmacol. (Epub ahead of print)
Lu T, Chen F (2012) Quantitative analysis of molecular surface based on improved marching tetrahedra algorithm. J Mol Graph Model 38:314–323
Lu T, Chen FW (2012) Multiwfn: a multifunctional wavefunction analyzer. J Comput Chem 33:580–592
Lu T, Manzetti S (2014) Wavefunction and reactivity study of benzo [a] pyrene diol epoxide and its enantiomeric forms. Struct Chem 25:1521–1533
Mary YS, Panicker CY, Thiemann T, Al-Azani M, Al-Saadi AA, Van Alsenoy C, Raju K, War JA, Srivastava SK (2015) Molecular conformational analysis, vibrational spectra, NBO, NLO analysis and molecular docking study of bis [(E)-anthranyl-9-acrylic] anhydride based on density functional theory calculations. Spectrochim Acta A 151:350–359
Milella L, Milazzo S, De Leo M, Vera Saltos MB, Faraone I, Tuccinardi T, Lapillo M, De Tommasi N, Braca A (2016) α-glucosidase and α-amylase inhibitors from arcytophyllum thymifolium. J Nat Prod 79(8):2104–2112
Mohamed TA, Soliman UA, Shaaban IA, Zoghaib WM, Wilson LD (2015) Raman, infrared and NMR spectral analysis, normal coordinate analysis and theoretical calculations of 5-(methylthio)-1,3,4-thiadiazole- 2(3H)-thione and its thiol tautomer. Spectrochim Acta A 150:339–349
Mokale SN, Palkar AD, Dube PN, Sakle NS, Miniyar PB (2016) Design, synthesis and in vivo screening of some novel quinazoline analogs as anti-hyperlipidemic and hypoglycemic agents. Bioorg Med Chem Lett 26:272–276
Morris GM, Huey R, Lindstrom W, Sanner MF, Belew RK, Goodsell DS, Olson AJ (2009) Autodock4 and Autodocktools4: automated docking with selective receptor flexibility. J Comput Chem 16:2785–2791
Muñoz MA, Joseph-Nathan P (2009) DFT-GIAO 1H and 13C NMR prediction of chemical shifts for the configurational assignment of 6beta-hydroxyhyoscyamine diastereoisomers. Magn Reson Chem 47(7):578–584
Murray JS, Politzer P (1998) Electrostatic potentials: chemical applications. In: Schleyer PvR (ed) Encyclopedia of computational chemistry. Wiley, West Sussex, p 912–920
Murray JS, Politzer P (2011) The electrostatic potential: an overview. Wiley Interdiscip Rev 1:153–163
Nurul Islam M, Jung HA, Sohn HS, Kim HM, Choi JS (2013) Potent α-glucosidase and protein tyrosine phosphatase 1B inhibitors from Artemisia capillaris. Arch Pharm Res 36(5):542–552
Pan G, Zhao L, Xiao N, Yang K, Ma Y, Zhao X, Fan Z, Zhang Y, Yao Q, Lu K, Yu P (2016) Total synthesis of 8-(6″-umbelliferyl)-apigenin and its analogs as anti-diabetic reagents. Eur J Med Chem 122:674–683
Parthasarathy K, Praveen C, Jeyaveeran JC, Prince AA (2016b) Gold catalyzed double condensation reaction: synthesis, antimicrobial and cytotoxicity of spirooxindole derivatives. Bioorg Med Chem Lett 26(17):4310–4317
Parthasarathy K, Praveen C, Saranraj K, Balachandran C, Senthil Kumar P (2016a) Synthesis, antimicrobial and cytotoxic evaluation of spirooxindole[pyrano-bis-2H-l-benzopyrans]. Med Chem Res 25:2155–2170
Pearson K (1901) On lines and planes of closest fit to systems of points in space. Philos Mag 6:559–572
Prabavathi N, Nilufer A (2015) Quantum chemical calculations on elucidation of molecular structure and spectroscopic insights on 2-amino-4-methoxy-6-methylpyrimidine and 2-amino-5-bromo-6- methyl-4-pyrimidinol-a comparative study. Spectrochim Acta A 136(Pt B):192
Praveen C, Ananth DB (2016) Design, synthesis and cytotoxicity of pyrano[4,3-b]indol-1(5H)-ones: a hybrid pharmacophore approach via gold catalyzed cyclization. Bioorg Med Chem Lett 26(10):2507–2512
Praveen C, Dheenkumar P, Perumal PT (2013) Super acid catalysed sequential hydrolysis/cycloisomerization of o-(acetylenic)benzamides under microwave condition: synthesis, antinociceptive and antiinflammatory activity of substituted isocoumarins. J Chem Sci 125:71–83
Qi YJ, Zhao YM, Lu HN, Wang XE, Jin NZ (2016a) Comparative analysis of the bonding modes between two antidiabetic drugs with beta-glucosidases from different species. Indian J Pharm Sci 78(4):525–536
Qi YJ, Zhao YM, Lu HN, Jin NZ (2016b) Exploring molecular flexibility and the interactions of Quercetin derivatives in the active site of α-glucosidase using molecular docking and charge density analysis. Comput Theor Chem 1094:55–68
Renuga Parameswari A, Rajalakshmi G, Kumaradhas P (2015) A combined molecular docking and charge density analysis is a new approach for medicinal research to understand drug-receptor interaction: curcumin-AChE model. Chem Biol Interact 225:21–31
Sabu MC, Kuttan R (2002) Anti-diabetic activity of medicinal plants and its relationship with their antioxidant property. J Ethnopharmacol 81(2):155–160
Sakkiah S, Thangapandian S, Lee KW (2012) Pharmacophore modeling, molecular docking, and molecular dynamics simulation approaches for identifying new lead compounds for inhibiting aldose reductase 2. J Mol Model 18(7):3267–3282
Selvaraju K, Jothi M, Kumaradhas P (2012) Understanding the charge density distribution and the electrostatic properties of hexadecane molecular nanowire under electric field using DFT and AIM theory. Comput Theor Chem 992:9–17
Singh RN, Baboo V, Rawat P, Kumar A, Verma D (2012) Molecular structure, spectral studies, intra and intermolecular interactions analyzes in a novel ethyl 4-[3-(2-chloro-phenyl)-acryloyl]- 3,5-dimethyl-1H-pyrrole-2-carboxylate and its dimer: A combined DFT and AIM approach. Spectrochim Acta A 94:288–301
Sun H, Zhang Y, Ding W, Zhao X, Song X, Wang D, Li Y, Han K, Yang Y, Ma Y, Wang R, Wang D, Yu P (2016) Inhibitory activity evaluation and mechanistic studies of tetracyclic oxindole derivatives as α-glucosidase inhibitors. Eur J Med Chem 123:365–378
Tomasi J, Mennucci B, Cammi R (2005) Quantum mechanical continuum solvation models. Chem Rev 105(8):2999–3093.
Toobaei Z, Yousefi R, Panahi F, Shahidpour S, Nourisefat M, Doroodmand MM, Khalafi-Nezhad A (2015) Synthesis of novel poly-hydroxyl functionalized acridine derivatives as inhibitors of α-glucosidase and α-amylase. Carbohydr Res 411:22–32
van Bochove MA, Bickelhaupt FM (2008) Nucleophilic substitution at C, Si and P: how solvation affects the shape of reaction profiles. Eur J Org Chem 4:649–654
van Bochove MA, Swart M, Bickelhaupt FM (2007) Nucleophilic substitution at phosphorus centers (SN2@P). ChemPhysChem 8:2452–2463
van Bochove MA, Swart M, Bickelhaupt FM (2009) Stepwise walden inversion in nucleophilic substitution at phosphorus. Phys Chem Chem Phys 11:259–267
Vijayalakshmi KP, Suresh CH (2008) Theoretical studies on the carcinogenic activity of diol epoxide derivatives of PAH: proton affinity and aromaticity as decisive descriptors. Org Biomol Chem 6:4384–4390
Wang G, Peng Z, Wang J, Li J, Li X (2016) Synthesis, biological evaluation and molecular docking study of N-arylbenzo[d]oxazol-2-amines as potential α-glucosidase inhibitors. Bioorg Med Chem 24(21):5374–5379
Wang L, Li T, Shen Y, Song Y (2016) A theoretical study of the electronic structure and charge transport properties of thieno[2,3-b]benzothiophene based derivatives. Phys Chem Chem Phys 18(12):8401
Wang P, Hao J, Zhang X, Wang C, Guan H, Li M (2016) Synthesis of furostanol glycosides: discovery of a potent α-glucosidase inhibitor. Org Biomol Chem 14:9362–9374
Wang ZW, Wang JS, Luo J, Kong LY (2013) α-Glucosidase inhibitory triterpenoids from the stem barks of Uncaria laevigata. Fitoterapia 90:30–37
Yang HY, Murphy-Ullrich JE, Song YH (2016) Molecular Insights for the role of key residues of calreticulin in its binding activities. Biophys J 110:p53a
Yar M, Bajda M, Shahzadi L, Shahzad SA, Ahmed M, Ashraf M, Alam U, Khan IU, Khan AF (2014) Novel synthesis of dihydropyrimidines for a-glucosidase inhibition to treat type 2 diabetes: in vitro biological evaluation and in silico docking. Bioorg Chem 54:96–104
Yearley EJ, Zhurova EA, Zhurov VV, Pinkerton AA (2007) Binding of genistein to the estrogen receptor based on an experimental electron density study. J Am Chem Soc 129(48):15013
Zhao DG, Zhou AY, Du Z, Zhang Y, Zhang K, Ma YY (2015) Coumarins with α-glucosidase and α-amylase inhibitory activities from the flower of Edgeworthia gardneri. Fitoterapia 107:122–127
Acknowledgements
This work was supported by the Fundamental Research Funds for the Central Universities (31920170007) and the Innovative Training Program for College Students (201710742084).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Qi, Y., Lu, H., Jin, N. et al. Understanding of the conformational flexibility and electrostatic properties of coumarin derivatives in the active site of S. cerevisiae α-glucosidase. Med Chem Res 27, 607–617 (2018). https://doi.org/10.1007/s00044-017-2086-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-017-2086-4