Abstract
Objectives
The aim of the study was to identify HIF-1A genetic variants and their possible association with HIF-1α, VEGF, KDR, RORc and Foxp3 protein levels, and susceptibility to and severity of RA.
Methods
The HIF-1A gene polymorphisms were genotyped for 587 RA patients and 341 healthy individuals. The HIF-1α, VEGF, KDR, RORc and Foxp3serum levels were evaluated.
Results
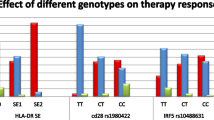
Under the codominant model, the frequency of the rs12434438 GG genotype was lower in RA patients than in controls (P = 0.02). Under the recessive model (AA + AG vs GG), the association was also significant (OR 3.32; CI 1.19–9.24; P = 0.02). Overall, rs12434438 A/G and rs1951795 A/C are in almost completed linkage disequilibrium with D′ = 0.96 and r2 = 0.85. The HIF-1A rs1951795 A allele was associated with rheumatoid factor (P = 0.02) and mean value of erythrocyte sedimentation rate (ESR) (P = 0.05). In RA patients with HIF-1A rs12434439 GG genotype, the parameters of disease activity such as DAS-28, VAS score, Larsen score or HAQ score were lower compared to RA patients with the HIF-1A rs12434439 AA genotype. Moreover, we also observed that Foxp3 serum levels were higher, and RORc2 serum levels were lower in RA patients with rs12434439 GG.
Conclusion
The polymorphic HIF-1A rs12434439 GG genotype may play a protective role for RA development.

Similar content being viewed by others
References
Wang CH, Yao H, Chen LN, Jia JF, Wang L, Dai JY, et al. CD147 induces angiogenesis through a vascular endothelial growth factor and hypoxia-inducible transcription factor 1α-mediated pathway in rheumatoid arthritis. Arthritis Rheum. 2012;64:1818–27.
Konisti S, Kiriakidis S, Paleolog EM. Hypoxia—a key regulator of angiogenesis and inflammation in rheumatoid arthritis. Nat Rev Rheumatol. 2012;8:153–62.
Quiñonez-Flores CM, González-Chávez SA, Pacheco-Tena C. Hypoxia and its implications in rheumatoid arthritis. J Biomed Sci. 2016;23:62.
Deng W, Feng X, Li X, Wang D, Sun L. Hypoxia-inducible factor 1 in autoimmune diseases. Cell Immunol. 2016;303:7–15.
Hu F, Liu H, Xu L, Li Y, Liu X, Shi L, et al. Hypoxia-inducible factor-1α perpetuates synovial fibroblast interactions with T cells and B cells in rheumatoid arthritis. Eur J Immunol. 2016;46:742–51.
Jeon CH, Ahn JK, Chai JY, Kim HJ, Bae EK, Park SH, et al. Hypoxia appears at pre-arthritic stage and shows co-localization with early synovial inflammation in collagen induced arthritis. Clin Exp Rheumatol. 2008;26:646–8.
Maruotti N, Cantatore FP, Crivellato E, Vacca A, Ribatti D. Angiogeneis in rheumatoid arthritis. Histol Histopathol. 2006;21:557–66.
Koch AE, Harlow LA, Haines GK, Amento EP, Unemori EN, Wong WL, et al. Vascular endothelial growth factor. A cytokine modulating endothelial function in rheumatoid arthritis. J Immunol. 1994;152:4149–56.
Ballara S, Taylor PC, Reusch P, Marmé D, Feldmann M, Maini RN, et al. Raised serum vascular endothelial growth factor levels are associated with destructive change in inflammatory arthritis. Arthritis Rheum. 2001;44:2055–64.
Kurosaka D, Hirai K, Nishioka M, Miyamoto Y, Yoshida K, Noda K, et al. Clinical significance of serum levels of vascular endothelial growth factor, angiopoietin-1, and angiopoietin-2 in patients with rheumatoid arthritis. J Rheumatol. 2010;37:1121–8.
Paradowska-Gorycka A, Pawlik A, Romanowska-Prochnicka K, Haladyj E, Malinowski D, Stypinska B, et al. Relationship between VEGF gene polymorphisms and serum VEGF protein levels in patients with rheumatoid arthritis. PLoS One. 2016;11:e0160769.
Dang EV, Barbi J, Yang HY, Jinasena D, Yu H, Zheng Y, et al. Control of T(H)17/T(reg) balance by hypoxia-inducible factor 1. Cell. 2011;146:772–84.
Larsen H, Muz B, Khong TL, Feldmann M, Paleolog EM. Differential effects of Th1 versus Th2 cytokines in combination with hypoxia on HIFs and angiogenesis in RA. Arthritis Res Ther. 2012;14:R180.
Hot A, Zrioual S, Lenief V, Miossec P. IL-17 and tumour necrosis factor α combination induces a HIF-1α-dependent invasive phenotype in synoviocytes. Ann Rheum Dis. 2012;71:1393–401.
Shen J, Li Z, Chen J, Song Z, Zhou Z, Shi Y. SHEsisPlus, a toolset for genetic studies on polyploid species. Sci Rep. 2016;6:24095.
Shi Y, He L. SHEsis, a powerful software platform for analyses of linkage disequilibrium, haplotype construction, and genetic association at polymorphism loci. Cell Res. 2005;15:97–8.
Li Z, Zhang Z, He Z, Tang W, Li T, Zeng Z, et al. A partition-ligation-combination-subdivision EM algorithm for haplotype inference with multiallelic markers. Cell Res. 2009;19:519–23.
Wipff J, Dieude P, Avouac J, Tiev K, Hachulla E, Granel B, et al. Association of hypoxia-inducible factor 1A (HIF1A) gene polymorphisms with systemic sclerosis in a French European Caucasian population. Scand J Rheumatol. 2009;38:291–4.
Li P, Cao Q, Shao PF, Cai HZ, Zhou H, Chen JW. at al. Genetic polymorphisms in HIF1A are associated with prostate cancer risk in a Chinese population. Asian J Androl. 2012;14:864–9.
López-Reyes A, Rodríguez-Pérez JM, Fernández-Torres J, Martínez-Rodríguez N, Pérez-Hernández N, Fuentes-Gómez AJ, et al. The HIF1A rs2057482 polymorphism is associated with risk of developing premature coronary artery disease and with some metabolic and cardiovascular risk factors. The genetics of atherosclerotic disease (GEA) Mexican study. Exp Mol Pathol. 2014;96:405–10.
Fernández-Torres J, Hernández-Díaz C, Espinosa-Morales R, Camacho-Galindo J, Galindo-Sevilla Ndel C, López-Macay Á, et al. Polymorphic variation of hypoxia inducible factor-1 A (HIF1A) gene might contribute to the development of knee osteoarthritis: a pilot study. BMC Musculoskelet Disord. 2015;16:218.
Wang X, Ren H, Zhao T, Ma W, Dong J, Zhang S, et al. Single nucleotide polymorphism in the microRNA-199a binding site of HIF1A gene is associated with pancreatic ductal adenocarcinoma risk and worse clinical outcomes. Oncotarget. 2016;7:13717–29.
Yu ZG, Wang BZ, Cheng ZZ. The association of genetic polymorphisms of hypoxia inducible factor-1alpha and vascular endothelial growth factor with increased risk of chronic obstructive pulmonary disease: a case-control study Kaohsiung. J Med Sci. 2017;33:433–441.
Wei WT, Li B, Chen M, Jia HR, Zhang HX. Associations between HIF-1α polymorphisms C1772T and G1790A and susceptibility to chronic obstructive pulmonary disease. Genet Mol Res. 2015;14:17341–7.
Bi YX, Yu L, Jin GX. Correlation between polymorphisms of hypoxia-inducible factor-1α Pro582Ser and type 2 diabetic nephropathy. Genet Mol Res. 2015;14:14503–9.
Liao SH, Liu WZ, Liu T, Sun Y, Feng X. Zhou HF.Potential signaling pathway of hypoxia-inducible factor in lung cancer and its gene polymorphism with lung cancer risk. J Recept Signal Transduct Res. 2015;35(4):233–7.
Guo X, Li D, Chen Y, An J, Wang K, Xu Z, et al. SNP rs2057482 in HIF1A gene predicts clinical outcome of aggressive hepatocellular carcinoma patients after surgery. Sci Rep. 2015;5:11846.
Hu F, Shi L, Mu R, Zhu J, Li Y, Ma X, et al. Hypoxia-inducible factor-1α and interleukin 33 form a regulatory circuit to perpetuate the inflammation in rheumatoid arthritis. PLoS One. 2013;8:e72650.
Wu J, Cui H, Zhu Z, Wang L, Li H, Wang D. Effect of HIF1α on Foxp3 expression in CD4+ CD25− T lymphocytes. Microbiol Immunol. 2014;58:409–415.
Clambey ET, McNamee EN, Westrich JA, Glover LE, Campbell EL, Jedlicka P, et al. Hypoxia-inducible factor-1 alpha-dependent induction of FoxP3 drives regulatory T-cell abundance and function during inflammatory hypoxia of the mucosa. Proc Natl Acad Sci USA. 2012;109:E2784–2793.
hi LZ, Wang R, Huang G, Vogel P, Neale G, Green DR, et al. HIF1alpha-dependent glycolytic pathway orchestrates a metabolic checkpoint for the differentiation of TH17 and Treg cells. J Exp Med. 2011;208:1367–1376.
Kao JK, Hsue YT, Lin CY. Role of new population of peripheral CD11c(+) CD8(+) T cells and CD4(+) CD25(+) regulatory T cells during acute and remission stages in rheumatoid arthritis patients. J Microbiol Immunol Infect. 2007;40:419–27.
Acknowledgements
The technical assistance of Wieslawa Frankowska and Teresa Golaszewska is gratefully acknowledged. We are also grateful to all the RA patients and healthy subjects whose cooperation made this study possible.
Funding
The work was supported by Grant S/13, National Institute of Geriatrics, Rheumatology and Rehabilitation.
Author information
Authors and Affiliations
Contributions
APG conceived, designed and performed experiments, analyzed and interpreted data and wrote the first draft of the manuscript. BS performed experiments and statistical analysis. AP contributed to drafting the manuscript. KRP, EH and MO were involved in the classification of patients with rheumatoid arthritis, clinical check of patients and treatment control. All authors read and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: John Di Battista.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Paradowska-Gorycka, A., Stypinska, B., Pawlik, A. et al. HIF-1A gene polymorphisms and its protein level in patients with rheumatoid arthritis: a case–control study. Inflamm. Res. 67, 423–433 (2018). https://doi.org/10.1007/s00011-018-1134-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-018-1134-y