Abstract
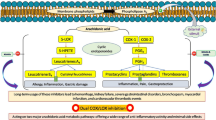
Objective: In order to elucidate potential anti-inflammatory activities of Zeel comp. N and its constituents, the inhibition of the synthesis of Leukotriene B4 (LTB4) and Prostaglandin (PGE2) by 5-lipoxygenase (5-LOX) and cyclo-oxygenase 1 and 2 (COX 1 and 2) respectively were examined in vitro.
Materials: Human HL-60 cells, differentiated for 6–8 days with DMSO (1.2% v/v) were used for the 5-LOX assay. The COX activity assays were carried out with purified enzymes, COX 1 (ram seminal vesicles), COX 2 (sheep placenta) and with human THP-1 cells, differentiated for 24 h with PMA (50 nM).
Methods: LTB4 and PGE2 production in the 5-LOX and COX assays respectively were determined by enzyme linked immunoassays.
Results: A reconstituted Zeel comp. N combination as well as its constituent mother tinctures of Arnica montana, Sanguinaria canadensis and Rhus toxicodendron (Toxicodendron quercifolium) showed distinct inhibitory effects on the production of LTB4 by 5-LOX (IC50 values of 10, 20, 2 and 5 µg/ml respectively) and on the synthesis of PGE2 by COX 1 (IC50 values of 50, 80, 40 and 20 µg/ml respectively) and COX 2 enzymes (IC50 values of 60, 110, 50 and 20 µg/ml respectively). The mother tincture of Solanum dulcamara inhibited the production of PGE2 by COX 1 (IC50 40 µg/ml) and COX 2 (IC50 150 µg/ml) but not production of leukotriene LTB4 by 5-LOX.
Conclusions: The observed dual inhibition of both LOX- and COX-metabolic pathways may offer an explanation for the reported clinical efficacy and the favorable gastrointestinal tolerability of the original remedy Zeel comp. N.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Additional information
Received 10 April 2003; returned for revision 27 May 2003; accepted by W. B. van den Berg 24 November 2003
Rights and permissions
About this article
Cite this article
Jäggi, R., Würgler, U., Grandjean, F. et al. Dual inhibition of 5-lipoxygenase/cyclooxygenase by a reconstituted homeopathic remedy; possible explanation for clinical efficacy and favourable gastrointestinal tolerability. Inflamm. res. 53, 150–157 (2004). https://doi.org/10.1007/s00011-003-1236-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-003-1236-y