Abstract
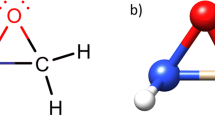
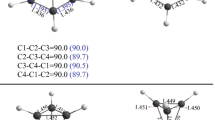
The nature of the bonding in the three-membered ring molecules cyclopropane, thiirane, oxirane, and aziridine has been investigated throughab initio FSGO calculations. The direct correspondence between floating spherical Gaussian orbitals and specific chemical bonds has been used to study the degree of “bond bending”. In accord with chemical intuition, it is demonstrated that the C-C ring bond becomes progressively more bent as the bond length is reduced. C-C bonds are found to be more flexible than C-N (O, S) bonds. The sizes and locations of carbon-heteroatom bond orbitals and C-H bond orbitals are also discussed.
Similar content being viewed by others
References
Clark, D. T.: Theoret. Chim. Acta (Berl.)15, 225 (1969)
Bonaccorsi, R., Scrocco, E., Tomasi, J.: J. Chem. Phys.62, 5270 (1970)
Frost, A.: J. Chem. Phys.47, 3707 (1967)
Frost, A., Rouse, R.: J. Am. Chem. Soc.90, 1965 (1968)
Talaty, E., Schwartz, A., Simons, G.: J. Am. Chem. Soc.97, 972 (1975)
Woods, J., Simons, G.: J. Am. Chem. Soc., in press
Simons, G., Zandler, M., Talaty, E.: J. Am. Chem. Soc.98, 7869 (1976)
Cunningham, G., Boyd, A., Myers, R., Gwenn, W., LeVan, W.: J. Chem. Phys.19, 676 (1951)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Talaty, E.R., Simons, G. Local orbital study of bonding in small rings: Cyclopropane, thiirane, oxirane, and aziridine. Theoret. Chim. Acta 48, 331–335 (1978). https://doi.org/10.1007/PL00020132
Received:
Issue Date:
DOI: https://doi.org/10.1007/PL00020132