Abstract
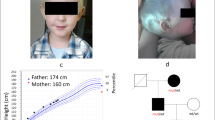
Constitutively activating germline mutations of the TSH receptor (TSH-R) are considered the cause of hereditary non-autoimmune hyperthyroidism. In this study, 10 members (8 affected and 2 unaffected) of an Italian family with hereditary non-autoimmune hyperthyroidism were investigated for the presence of mutations in the TSH-R gene. The clinical features of the disease were also analyzed. PCRamplified fragments of the TSH-R gene were obtained from genomic DNA extracted from peripheral blood leukocytes of each family member and analyzed by direct nucleotide sequencing and restriction analysis. An identical germline TSH-R mutation was detected in all the patients with hyperthyroidism but in none of the unaffected family members. The mutation was heterozygotic and determined the substitution of valine for methionine (codon 463; ATG —>GTG) in the second transmembrane domain of the TSH-R. When expressed in chinese hamster ovary (CHO) cells, the Val463 mutant TSH-R induced constitutive activation of the TSH receptor. Analysis of the clinical features of our family and those of other families with hereditary non-autoimmune hyperthyroidism, including one with the same Val463 mutation, revealed wide variability in the phenotypical expression of the disease. Our findings indicate that an activating germline mutation in the TSHR gene plays a key role in hereditary non-autoimmune hyperthyroidism although the onset of clinical manifestations and the evolution of the disease seem to depend heavily on other factors, thus far unidentified. The absence of a clear correlation between mutant genotypes and phenotypic expression of the disease currently limits the prognostic value of genetic testing in families with hereditary non-autoimmune hyperthyroidism.
Similar content being viewed by others
References
Thomas J.L., Leclere J., Hartemann P. eaet al. Familial hyperthyroidism without evidence of autoimmunity. Acta Endocrinol. (Copenh.) 1982, 100: 512–518.
Leclere J., Bene M.C., Aubert V. et al. Clinical consequences of activating germline mutations of TSH receptor, the concept of toxic hyperplasia. Horm. Res. 1997, 47: 158–162.
Duprez L., Parma J., Van Sande J., et al. Germline mutations in non-autoimmune the thyrotropin receptor gene cause autosomal dominant hyperthyroidism. Nat. Genet. 1994, 7: 396–401.
Tonacchera M., Van Sande J., Cetani F., et al. Functional characteristics of three new germline mutations of the thyrotropin receptor gene causing autosomal dominant toxic thyroid hyperplasia. J. Clin. Endocrinol. Metab. 1996, 81: 547–554.
Schwab K.O., Sohlemann P., Gerlich M., et al. Mutations of the TSH receptor as cause of congenital hyperthyroidism. Exp. Clin. Endocrinol. Diabetes 1996, 104: (4) 124–128.
Fuhrer D., Wonerow P., Willgerodt H., Paschke R. Identification of a new thyrotropin receptor germline mutation (Leu629Phe) in a family with neonatal onset of autosomal dominant nonautoimmune hyperthyroidism J. Clin. Endocrinol. Metab. 1997, 82: 4234–4238.
Gruters A., Schoneberg T., Biebermann H., et al. Severe congenital hyperthyroidism caused by a germ-line neo mutation in the extracellular portion of the thyrotropin receptor. J. Clin. Endocrinol. Metab. 1998, 83: 1431–1436.
Khoo D.H.C., Parma J., Rajasoorya C., Ho S.C., Vassart G. Germline mutation of the thyrotropin receptor gene associated with thyrotoxicosis and mitral valve prolapse in a chinese family. J. Clin. Endocrinol. Metab. 1999, 84: 1459–1462.
Fuhrer D., Mix M., Wonerow P., Richter I., Willgerodt H., Paschke R. Variable phenotype associated with Ser505Asnactivating thyrotropin-receptor germline mutation. Thyroid 1999, 9: 757–761.
Biebermann H., Krude H., Gudermann T., Gruters A. Costitutively activating TSH receptor mutations as a molecular cause of non-autoimmune hyperthyroidism in childhood. Langenbecks Arch. Surg. 2000, 385: 390–392.
Fuhrer D., Warner J., Sequeira M., Paschke R., Gregory J., Ludgate M. Novel TSHR germline mutation (Met463Val) masquerading as Graves’ disease in a large Welsh kindred with hyperthyroidism. Thyroid 2000, 10: 1035–1041.
Duprez L., Parma J., Van Sande J., et al. TSH receptor mutations and thyroid disease. Trends Endocrinol. Metab. 1998, 9: 133–140.
Fuhrer D., Paschke R. Thyroid-stimulating hormone receptor mutations: update and clinical implications. Curr. Opin. Endocrinol. Diab. 2000, 7: 288–294.
Camacho P., Gordon D., Chiefari E., et al. A Phe 486 thyrotropin receptor mutation in an autonomously functioning follicular carcinoma that was causing hyperthyroidism. Thyroid 2000, 10: 1009–1012.
Gross B., Misrahi M., Sar S., Milgrom E. Composite structure of the human thyrotropin receptor gene. Biochem. Biophys. Res. Commun. 1991, 177: 679–687.
Russo D., Arturi F., Wicker R. et al. Genetic alterations in thyroid hyperfunctioning adenomas. J. Clin. Endocrinol. Metab. 1995, 80: 1347–1351.
Sanger F., Nicklen S., Coulson R. Dna sequencing with chain terminating inhibition. Proc. Natl. Acad. Sci. USA 1977, 82: 488–492.
Chen C., Okayama H. High-efficiency transformation of mammalian cells by plasmid DNA. Mol. Cell. Biol. 1987, 7: 2745–2752.
Foti D., Russo D., Costante G., Filetti S. The biological activity of bovine and human thyrotropin is differently affected by trypsin treatment of human thyroid cells: thyroid-stimulating antibody is related to human thyrotropin. J. Clin. Endocrinol. Metab. 1991, 73: 710–716.
Labarca C., Paigen K. A simple, rapid and sensitive DNA assay procedure. Anal. Biochem. 1980, 102: 344–352.
Russo D., Arturi F., Chiefari E., Filetti S. Molecular insights into TSH receptor abnormality and thyroid disease. J. Endocrinol. Invest. 1997, 20: 36–47.
Spambalg D., Sharifi N., Elisei R., Gross J.L., Medeiros-Neto G., Fagin J.A. Structural studies of the thyrotropin receptor and Gsa in human thyroid cancers: low prevalence of mutations predicts infrequent involment in malignant transformation. J. Clin. Endocrinol. Metab. 1996, 81: 3898–3901.
Russo D., Tumino S., Arturi F., et al. Detection of an activating mutation of the thyrotropin receptor in a case of an autonomously hyperfunctioning thyroid insular carcinoma. J. Clin. Endocrinol. Metab. 1997, 82: 753–738.
Russo D., Wong M., Costante G. et al. A Val 677 activating mutation of the thyrotropin receptor in a Hurtle cell thyroid carcinoma associated with thyrotoxicosis. Thyroid 1999, 9: 13–17.
Derwahl M. Editorial: TSH receptor and Gsa gene mutations in the pathogenesis of toxic thyroid adenomas — A note of caution. J. Clin. Endocrinol. Metab. 1996, 81: 2783–2785.
Arturi F., Capula C., Chiefari E., Filetti S., Russo D. Thyroid hyperfunctioning adenomas with and without Gsp/TSH receptor mutations show similar clinical features. Exp. Clin. Endocrinol. Diab. 1998, 106: 234–236.
Corvilain B., Van Sande J., Dumont J.E., Vassart G. Somatic and germline mutations of the TSH receptor and thyroid disease. Clin. Endocrinol. 2001, 55: 143–158.
Filetti S., Bidart J.M., Arturi F., Caillou B., Russo D., Schlumberger M. Sodium/iodide symporter: a key transport system in thyroid cancer cell metabolism. Eur. J. Endocrinol. 1999, 141: 443–457.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arturi, F., Chiefari, E., Tumino, S. et al. Similarities and differences in the phenotype of members of an Italian family with hereditary non-autoimmune hyperthyroidism associated with an activating TSH receptor germline mutation. J Endocrinol Invest 25, 696–701 (2002). https://doi.org/10.1007/BF03345103
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03345103