Abstract
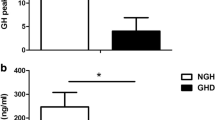
Basal IGF-I levels and the GH response to at least two among provocative stimuli such as clonidine (CLO, Catapresan, 150 mcg/m2 po), GHRH (1 mcg/kg iv)+arginine (ARG, 0.5 g/kg iv infusion during 30 min) and GHRH+pyridostigmine (PD, Mestinon cpr 60 mg po) have been evaluated in 43 children with Prader-Willi syndrome (PWS, 17 males and 26 females, age 3–22 yr, 7 normal weight and 36 obese PWS), in 25 normal short children (NC, 17 males and 8 females, 7.7–18.5 yr) and in 24 children with simple obesity (OB, 14 males, 10 females, 7.7–21.5 yr). Both normal weight and obese PWS had mean IGF-I levels lower than those recorded in NC (p<0.001) and OB (p<0.001). The GH responses to GHRH+ARG and GHRH+PD in NC were similar and higher than that to CLO (p<0.001). In PWS the GH response to GHRH+ARG was higher than that to GHRH+PD (p<0.001) which, in turn, was higher than that to CLO (p<0.001); these responses in PWS were lower than those in normal children (p<0.02) and similar to those in OB. In normal weight PWS the GH responses to GHRH+ARG and to GHRH+PD were similar and higher than to CLO (p<0.05); however, each provocative stimulus elicited a GH rise lower than that in NC (p<0.05). In obese PWS as well as in OB the GH response to GHRH+ARG was higher than that to GHRH+PD (p<0.02) which, in turn, was higher than that to CLO (p<0.001); all GH responses in obese PWS and OB were lower than those in NC (p<0.001) but similar to those in normal weight PWS. In conclusion, patients with PWS show clear reduction of IGF-I levels as well as of the somatotroph responsiveness to provocative stimuli independently of body weight excess. These results strengthen the hypothesis that PWS syndrome is frequently connoted by GH insufficiency.
Similar content being viewed by others
References
Ledbetter D., Riccardi V., Airhart S., Strobel R., Keenen S., Crawford J. Deletion of cromosome 15 as a cause of the Prader Willi Syndrome. N. Engl. J. Med. 1981, 11: 304–325.
Carrozzo R., Rossi E., Christian S., Kittickamron K., Livieri C., Corrias A., Pucci L., Fois A., Simi P., Bosio L., Beccaria L., Zuffardi O., Ledbetter D. Inter and intrachromosomal rearrangements qre bth involved in the origin of 15 q11–q13 deletion. Am. J. Hum. Genet. 1997, 61: 228–232.
Nicholls R., Knoll J., Butker M., Karam S., Lalande M. Genetic imprinting suggested by maternal heterodisomy in nondeletion Prader Willi syndrome. Nature (London) 1989, 342: 281–285.
Holm V.C., Cassidy S.B., Butler M.G., Hanchett J.M., Greenswag L.R., Witman B.Y., Greemberg F. Prader-Willi syndrome: consensus diagnostic criteria. Pediatrics 1993, 91: 398–402.
Donaldson M., Chu C., Cooke A., Wilson A., Greene S., Stephenson J. The Prader Willi syndrome. Arch. Dis. Child. 1994, 70: 58–63.
Hanson J. A view of etiology and pathogenesis of Prader — Willi syndrome. In: Holm V.A., Sulzbacher S.J., Pipe P.L. (Eds.), The Prader-Willi syndrome. University Park Press, Baltimore, 1981, p. 45.
Swaab D., Purba J. S., Hofman M.A. Alterations in the hypothalamic paraventricular nucleus and its oxytocin neurons (putative satiety cells) in Prader-Willi syndrome: a study of five cases. J. Clin. Endocrinol. Metab. 1995, 80: 573–579.
Costeff H., Holm V.A., Ruvalcaba R., Shaver J. Growth hormone secretion in Prader-Willi syndrome. Acta Paediatr. Scand. 1990, 79: 1059–1062.
Cappa M., Grossi A., Borrelli P., Ghigo E., Bellone J., Benedetti S., Carta D., Loche S. Growth homone (GH) response to combined Pyridostigmine and GH-releasing hormone administration in patients with Prader — Labhard — Willi Syndrome. Horm. Res. 1993, 39: 51–55.
Angulo M., Castro-Magana M., Mazur B., Canas J., Vitollo P., Sarrantonio M. Growth hormone secretion and effects of growth hormone therapy on growth velocity and weight gain in children with Prader Willi Syndrome. J. Paed. Endocrinol. Metab. 1996, 9: 393–400.
Beccaria L., Bosio L., Sanzari A., Aimaretti G., Ghigo E., Chiumello G. GH secretion in Prader-Labhard — Willi Syndrome: somatotrope responsiveness to GHRH is enhanced by arginine but not by pyridostigmine. J. Paed. Endocrinol. Metab. 1996, 9: 577–583.
Grugni G., Guzzaloni G., Moro D., Bettio D., De Medici C., Morabito F. Reduced growth hormone (GH) responsiveness to combined GH-releasing hormone and pyridostigmine administration in the Prader Willi syndrome. Clin. Endocrinol. 1998, 48: 769–775.
Ranke M.B., Haber P. Growth hormone stimulation tests. In: Ranke M.B. (Eds.), Functional endocrinologic diagnostic in children and adolescents. I.&J., Verlag Mannheim, 1992, p. 61.
Ghigo E., Bellone J., Aimaretti G., Bellone S., Loche S., Cappa M., Bartolotta E., Dammacco F., Camanni F. Reliability of provocative tests to assess growth hormone secretory status. Study in 472 normally growing children. J. Clin. Endocrinol. Metab. 1996, 81: 3323–3327.
Cole T.J. A method for assessing age standardised weight for height in children seen cross sectionally. Ann. Hum. Biol. 1979, 7: 457–473.
Iranmanesh A., Lizarralde G., Veldhuis J.D. Age and relative adiposity are specific negative determinants of the frequency and amplitude of growth hormone (GH) secretory burts and the half-life of endogenous GH in healthy man. J. Clin. Endocrinol. Metab. 1991, 73: 1081–1088.
Veldhuis J.D., Iranmanesh A., Ho K.K.Y., Waters M.J., Johmson M.L., Lizarralde G. Dual defect in pulsatile growth hormone secretion and clearance subserve the hyposomatotropinism of obesity in man. J. Clin. Endocrinol. Metab. 1991, 72: 51–59.
Topper E., Gil-Ad I., Bauman B., Josefaberg Z., Laron Z. Plasma growth hormone response to oral clonidine as compared to insulin hypoglicemia in obese children and adolescents. Horm. Metab. Res. 1984, 1: 127–130.
Loche S., Balzano S., Bozzola M., Moretta A., Pintus S., Faedda A., Muntoni A., Carta D., Pintor C. Secretion of growth hormone releasing hormone in obese children. J. Endocrinol. Invest. 1992, 15: 453–457.
Cordido F., Casanueva F.F., Dieguez C. Cholinergic receptor activation by pyridostigmine restores growth hormone (GH) responsiveness to GH-releasing hormone administration in obese subjects. J. Clin. Endocrinol. Metab. 1989, 68: 290–293.
Rasmussen M.H., Hvidberg A., Juul A., Main K.M., Gotfredsen A., Skakkebaek M., Hilsted J. Massive weight loss restores 24-hour growth hormone release profiles and serum insulin-like growth factor-I in obese subjects. J. Endocrinol. Metab. 1995, 80: 1407–1415.
Tannenbaum G.S., Lapointe M., Gurd W., Finkelstein J.A. Mechanism of impaired growth hormone secretion in genetically obese zucher rats: roles of growth hormone-releasing factor and somatostatin. Endocrinology 1990, 127: 3087–3091.
Ghigo E., Mazza E., Corrias A., Imperiale E., Goffi S., Arvat E., Bellone J., de Sanctis C., Muller E.E., Camanni F. Effect of cholinergic enhancement by pyridostigmine on growth hormone secretion in obese adults and children. Metabolism 1989, 38: 631–633.
Ghigo E., Procopio M., Maccario M., Bellone J., Arvat E., Campana S., Boghen M.F., Camanni F. Repetitive GHRH administration fails to increase to GHRH in obese subjects. Evidence for a somatotrope defect in obesity? Horm. Metab. Res. 1993, 25: 305–308.
Maccario M., Procopio M., Grottoli S., Oleandri S.E., Boffano G.M., Taliano, M., Camanni F., Ghigo E. Effects of acipimox, an antilipolytic drug, on the growth hormone (GH) response to GH-releasing hormone alone or combined with arginine in obesity. Metabolism 1996, 45: 1–6.
Cordido F., Peino R., Penalva A., Alvarez C.V., Casanueva F.F., Dieguez C. Impaired growth hormone secretion in obese subjects is partially reversed by acipimox-mediated plasma free fatty acid depression. J. Clin. Endocrinol. Metab. 1996, 81: 914–918.
Frystyk J., Skjaerbaek E., Mogensen C.E., Orskov H. Free insulin-like growth factor in human obesity. Metabolism 1995, 44 (Suppl. 4): 37–44.
Loche S., Cappa M., Borrelli P., Faedda A., Crinò A., Cella S.G., Corda R., Muller E.E., Pintor C. Reduced growth hormone response to growth hormone-releasing hormone in children with simple obesity: evidence for a somatomedin-C mediated inhibition. Clin. Endocrinol. (Oxf.) 1987, 27: 145–150.
Rudman D., Kutner M.H., Rogers C.M., Lubin M.F., Fleming G.A., Bain R.P. Impaired growth hormone secretion in adult population: relation to age and adiposity. J. Clin. Invest. 1981, 67: 1361–1369.
Ghigo E., Bellone J., Mazza E., Imperiale E., Procopio M., Valente F., Lala R., de Sanctis C., Camanni F. Arginine potentiates the GHRH- but not the pyridostigmine- induced GH secretion in normal short children. Further evidence for a somatostatin suppressing effect of arginine. Clin. Endocrinol. (Oxf.) 1990, 32: 763–767.
Ghigo E., Arvat E., Goffi S., Nicolosi M., Arvat E., Procopio M., Bellone J., Valente F., Ghigo M.C., Camanni F. Repetitive GHRH and arginine administration to explore the maximal secretory capacity of somatotrope cells during lifespan. Clin. Exp. Endocrinol. 1991, 10: 191–198.
Blum W.F., Albertson-Wiekland K., Rosberg S., Ranke M.B. Serum levels of insulin-like growth factor (IGF-I) and IGF-I binding protein 3 spontaneous growth hormone secretion. J. Clin. Endocrinol. Metab. 1993, 76: 1610–1616.
Grosso S., Cioni M., Buoni S., Peruzzi L., Pucci L., Berardi P. Growth hormone secretion in Prader-Willi syndrome. J. Endocrinol. Invest. 1998, 21: 418–422.
Valetto M.R., Bellone J., Baffoni C., Savio P., Aimaretti G., Gianotti L., Arvat E., Camanni F., Ghigo E. Reproducibility of the growth hormone response to stimulation with growth hormone-releasing plus arginine during lifespan. Eur. J. Endocrinol. 1996, 135: 568–572.
Cappa M., Loche S., Salvadori R., Faedda A., Borrelli P., Cella S.G., Pintor C., Muller E.E. The GH response to pyridostigmine plus GHRH is not influenced by pubertal maturation. J. Endocrinol. Invest. 1991, 14: 41–45.
Author information
Authors and Affiliations
Consortia
Rights and permissions
About this article
Cite this article
Corrias, A., Bellone, J., Beccaria, L. et al. GH/IGF-I axis in Prader-Willi syndrome: Evaluation of IGF-I levels and of the somatotroph responsiveness to various provocative stimuli. J Endocrinol Invest 23, 84–89 (2000). https://doi.org/10.1007/BF03343684
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03343684