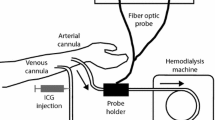
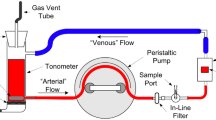
Abstract
The effects of fluorescein, methylene blue, and indocyanine green on hemodynamic variables and on pulse oximetry and co-oximetry measurements of arterial hemoglobin oxygen saturation (SaO2) and oxyhemoglobin percentage (% HbO2) were evaluated in 16 anesthetized dogs in vitro by cooximetry (% HbO2) and in vivo by pulse oximetry (SaO2). The light absorbance (optical density) in plasma (range 500 to 800 nm) was measured by a spectrophotometer. Fluorescein did not affect oximetry measurements, plasma light absorbance in the range measured, or hemodynamic variables. Methylene blue caused dose-dependent decreases in measurements made with both forms of oximetry for up to 30 minutes, the decrease being greater and longer lasting with pulse oximetry (P < 0.05). Hemodynamic measurements in 5 dogs showed that methylene blue (1 to 5 mg/kg) increased arterial pressure transiently, after which cardiac output, stroke index, and left ventricular stroke work index decreased and left ventricular end-diastolic pressure and systemic and pulmonary vascular resistances increased (P < 0.05 with 5 mg/kg). Methemoglobin concentration measured by co-oximetry increased significantly (to 19.9 ± 1.4%, P < 0.05) 1 minute after 5 mg/kg of methylene blue was injected. Methylene blue had a dose- and time-dependent effect on plasma light absorbance, and this effect peaked in the 660- to 670-nm range. The data do not distinguish the relative contributions of physiology (hemodynamic change), chemistry (methemoglobin production), and physics (optical properties) to the decrease in pulse oximetry and co-oximetry measurements that follows injection of methylene blue. Indocyanine green affected neither hemodynamic variables nor co-oximetry readings but decreased pulse oximetry readings for up to 10 minutes dose dependently. With doses of 0.1 to 3.0 mg/kg of indocyanine green plasma light absorbance peaked at 805 nm but increased dose dependently for up to 30 minutes at 660 nm. Methylene blue and indocyanine green (and, by implication, other dyes with plasma light absorbance peaks in the 600- to 1,000-nm range) artifactually alter oximetric detection of arterial hemoglobin oxygen saturation and oxyhemoglobin percentage.
Similar content being viewed by others
References
Kessler MR, Eide T, Humayun B, Poppers PJ. Spurious pulse oximeter desaturation with methylene blue injection. Anesthesiology 1986;65:435–436
Scheller MS, Unger RJ, Kelner MJ. Effects of intravenously administered dyes on pulse oximetry readings. Anesthesiology 1986;65:550–551
Yoshiya I, Shimada Y, Tanaka K. Spectrophotometric monitoring of arterial oxygen saturation in the fingertip. Med Biol Eng Comput 1980;18:27–32
Yelderman M, New W Jr. Evaluation of pulse oximetry. Anesthesiology 1983;59:349–352
Marmion DM. Handbook of U.S. colorants for foods, drugs, and cosmetics, 2nd ed. New York: Wiley-Inter-science, 1984:324
Kirk-Othmer. Encyclopedia of chemical technology, 2nd ed. vol 5. New York: Wiley, 1963:764
Prys-Roberts C. Measurement of cardiac output and regional blood flow. In: Prys-Roberts C, ed. The circulation in anaesthesia: applied physiology and pharmacology. Oxford: Blackwell Scientific, 1980:539
Operator’s manual. IL 282 CO-Oximeter. 79282. Lexington, MA: Instrumentation Laboratory, November 1977: 1/1-1/2
Kramer K, Elam JO, Saxton GA, Elam WN, Jr. Influence of oxygen saturation, erythrocyte concentration and optical depth upon the red and near-infrared light transmittance of whole blood. Am J Physiol 1951;165:229–246
Nilsson JN. Oximetry. Physiol Rev 1960;40:1–26
Harvey SC. Antiseptics and disinfectants; fungicides; ectoparasiticides. In: Gilman AG, Goodman LS, Gilman A, eds. The pharmacological basis of therapeutics, 6th ed. New York: Macmillan, 1985:980
Dees TM, Rumsfeld JA, Miller WF, Chapman CB. Clinical measurement of circulation time. A comparison of magnesium sulphate and Evans blue dye in normal subjects. J Appl Physiol 1957;10:451–454
Broadbent JC, Wood EH. Indicator-dilution curves in acyanotic congenital heart disease. Circulation 1954; 9:890–902
Davidson I, Henry JB. Clinical diagnosis by laboratory methods, 15th ed. Philadelphia: Saunders, 1974:112–113
Kiese M. Methemoglobinemia: a comprehensive treatise. Boca Raton, FL: CRC Press, 1974:9, 19–20, 51, 73–75
Young DS, Pestaner LC, Gibberman V. Effects of drugs on clinical laboratory tests. Clin Chem 1975;21:1D–432D
Rentsch G, Wittekind D. Methylene blue and erythrocytes in the living animal. Contribution to the toxicology of methylene blue and formation of Heinz-bodies. Toxicol Appl Pharmacol 1967;11:87
Stossel TP, Jennings RB. Failure of methylene blue to produce methemoglobinemia in vivo. Am J Clin Pathol 1966;45:600
Haggard HW, Greenberg LA. Methylene-blue: a synergist, not an antidote for carbon monoxide. JAMA 1933; 100:2001
van Slyke DD, Hiller A. Gasometric determination of methemoglobin. J Biol Chem 1929;84:205
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sidi, A., Paulus, D.A., Rush, W. et al. Methylene Blue and Indocyanine Green Artfactually Lower Pulse Oximetry Readings of Oxygen Saturation. Studies in Dogs. J Clin Monitor Comput 3, 249–256 (1987). https://doi.org/10.1007/BF03337379
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03337379