Summary
The aim of this study was to assess the cost-effectiveness of treatment with long-acting injectable risperidone compared with previous oral antipsychotic regimens in patients with schizophrenia. The analysis was conducted from the Health Service of Regione Lombardia perspective.
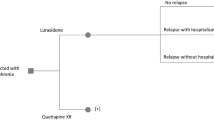
An observational, two-years long, retrospective study was conducted in 28 schizophrenic patients who were undergoing a treatment switch from oral antipsychotics to risperidone RP. The effectiveness of long-acting injectable risperidone compared with previous oral antipsychotics, defined as the absence of relapses (hospitalizations), was assessed at 12 months of treatment. The study considered direct medical costs referred to year 2007 values.
In terms of effectiveness, at 12 months after switching to long-acting injectable risperidone, there was a higher percentage of patients who did not require hospitalization (67.9%) as compared retrospectively with the same period for the previous treatments (28.6%).
Long-acting risperidone showed the best expected cost versus other oral antipsychotic treatments (€9, 191.45 vs €10, 125.57). The difference in favour of risperidone (€934.12) was mainly due to lower costs for hospitalization.
On the basis of these findings, which were obtained in real-world clinical practice, long-acting injectable risperidone is predicted to be the dominant strategy because it results in effective symptom control and direct medical cost savings.
Similar content being viewed by others
Bibliografia
Rouillon F, Toumi M, Dansette GY, et al. Some aspects of the costs of schizophrenia in France. PharmacoEconomics 1997; 11: 578–94
Garattini L, Rossi C, Tediosi F, et al. Direct costs of schizophrenia in Italian community psychiatric services. PharmacoEconomics 2001; 19: 1217–25
Garattini L, Rossi C, Tediosi F, et al. Costi diretti della schizofrenia nei dipartimenti di salute mentale italiani. PharmacoEconomics-Italian Research Articles 2002; 4: 81–89
Mapelli V, Bezzi R, Erlicher A, et al. Costi ed efficacia della cura della schizofrenia con antipsicotici tipici e atipici PharmacoEconomics-Italian Research Articles 2004; 6: 15–30
Ravasio R, Ferrannini L. Costo-efficacia di risperidone iniettabile a rilascio prolungato nel trattamento della schizofrenia in Italia. Un’analisi di confronto versus risperidone orale, olanzapina orale e aloperidolo decanoato. PharmacoEconomics-Italian Research Articles 2006; 8: 19–31
Foster RH, Goa KL. Risperidone. A pharmacoeconomic review of its use in schizophrenia. PharmacoEconomics 1998; 14: 97–133
Tansella M, de Girolamo G. La diffusione dei disturbi mentali nella comunità. In: Cassano GB, Dell’Acqua G, Garattini S, et al. (editors). La salute mentale. Contro il pregiudizio il coraggio delle cure. Roma: Il Pensiero Scientifico Editore, 2001: 7–12
Miller DS, Yatham LN, Lam RW. Comparative efficacy of typical and atypical antipsychotics as add-on therapy to mood stabilizers in the treatment of acute mania. J Clin Psychiatry 2001; 62: 975–80
Kane JM, Marder SR. Psychopharmacologic treatment of schizophrenia. Schizophr Bull 1993; 19: 253–62
Love RC. Novel versus conventional antipsychotic drugs. Pharmacotherapy 1996; 16 (1 Pt 2): 6–10
CCOHTA. Pharmacoeconomic evaluations of clozapine in treatment resistant schizophrenia and risperidone in chronic schizophrenia. Technology overview: pharmaceuticals 1997; 7: 1–15
Cookson RF, Huybrechts KF. Risperidone: an assessment of its economic benefits in the treatment of schizophrenia. Journal of Medical Economics 1998; 1: 103–34
Davies A, Langley PC, Keks NA, et al. Risperidone versus haloperidol: II. Cost-effectiveness. Clin Ther 1998; 20: 196–213
Markovitz JS, Brown CS, Moore TR. Atypical antipsychotic part I: pharmacology, pharmacokinetics, and efficacy. Ann Pharmacother 1999; 33: 73–85
Borison RL, Pathiraja AP, Diamond BI, Meibach RC. Risperidone: clinical safety and efficacy in schizophrenia. Psychopharmacol Bull 1992; 27: 213–18
Dellva MA, Tran P, Tollefson GD, et al. Standard olanzapine versus placebo and ineffective-dose olanzapine in the maintenance of schizophrenia. Psychiatr Serv 1998; 49: 699
www.nice.org.uk/guidance/index.jsp?action=download&o=42140. Data ultimo accesso marzo 2009
Mangalore R, Knapp M. Cost of schizophrenia in England. J Ment Health Policy Econ 2007; 10: 23–41
Oliva-Moreno J. The costs of schizophrenia in Spain. Eur J Health Econ 2006; 7: 182–8
Wu EQ, Birnbaum HG, Shi L, et al. The economic burden of schizophrenia in the United States in 2002. J Clin Psychiatry 2005; 66: 1122–9
Kelly GR, Scott JE, Mamon J. Medication compliance and health education among outpatients with chronic mental disorders. Med Care 1990; 28: 1181–97
Barnes TR. Depot antipsychotic drugs and prevention of psychotic relapse. Clin Neuropharmacol 1991; 14 (Suppl. 2): S1–6
Duncan JC, Rogers R. Medication compliance in patients with chronic schizophrenia: implications for the community management of mentally disordered offenders. J Forensic Sci 1998; 46: 1133–7
Scottish Schizophrenia Research Group. The Scottish First Episode Schizophrenia Study, II: Treatment: pimozide versus flupenthixol. Br J Psychiatry 1987; 150: 334–8
Young JL, Zonana HV Shepler L. Medication noncompliance in schizophrenia: codification and update. Bull Am Acad Psychiatry Law 1986; 14: 105–22
Buchanan A. A two-year prospective study of treatment compliance in patients with schizophrenia. Psychol Med 1992; 22: 787–97
Lieberman JA, Stroup TS, McEvoy JP, et al. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med 2005; 353: 1209–23
Weiden PJ, Olfson M. Cost of relapse in schizophrenia. Schizophr Bull 1995; 21: 419–29
Kane JM, Aguglia E, Altamura AC, et al. Guidelines for depot antipsychotic treatment in schizophrenia. Eur Neuropsychopharmacol 1998; 8: 55–66
Marder SR Overview of partial compliance. J Clin Psychiatry 2003; 64: 3–9
Niaz OS, Haddad PM. Thirty-five months experience of risperidone long-acting injection in aUK psychiatric service including a mirror-image analysis of inpatient care. Acta Psychiatr Scand 2007; 116: 36–46
Taylor DM, Young CL, Mace S, et al. Early clinical experience with risperidone long-acting injection: a prospective, 6-month follow-up of 100 patients. J Clin Psychiatry 2004; 65: 1076–83
Llorca PM, Miadi-Fargier H, Lançon C. Cost-effectiveness analysis of schizophrenic patient care settings: impact of an atypical antipsychotic under long-acting injection formulation. Encephale 2005; 31: 235–46
Chue P. Long-acting risperidone injection: efficacy, safety, and cost-effectiveness of the first long acting atypical antipsychotic. Neuropsychiatr Dis Treat 2007: 3: 13–39
Chue P, Llorca PM, Duchesne I, et al. Hospitalization rates in patients during long-term treatment with long-acting risperidone injection. J Appl Res 2005; 5: 266–74
Olivares JM, Rodriguez-Martinez A, Burón JA, et al. Cost-effectiveness analysis of switching antipsychotic medication to long-acting injectable risperidone in patients with schizophrenia. A 12- and 24-month follow-up from the e-STAR database in Spain. Appl Health Econ Health Policy 2008; 6: 41–53
De Graeve D, Smet A, Mehnert A, et al. Long-acting risperidone compared with oral olanzapine and haloperidol depot in schizophrenia: a Belgian cost-effectiveness analysis. Pharmacoeconomics 2005; 23 (Suppl. 1): 35–47
Laux G, Heeg BM, van Hout BA, et al. Long-acting risperidone compared with oral atypical and conventional depot formulations in Germany. Pharmacoeconomics 2005; 23 (Suppl. 1): 49–61
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ravasio, R., Sanfilippo, L., De Paoli, G. et al. Analisi di costo-efficacia dello switch da un antipsicotico orale a risperidone a rilascio prolungato nel trattamento dei pazienti affetti da schizofrenia. G. Ital. Health Technol. Assess 2, 1–8 (2009). https://doi.org/10.1007/BF03320713
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03320713