Abstract
Abstract
Inhaled formoterol is a long-acting selective β2-adrenoceptor agonist, with an onset of action of 5 minutes postdose and a bronchodilator effect that lasts for at least 12 hours.
Statistically significant and clinically relevant (> 120ml) improvements in lung function [assessed using standardized/normalized area under the forced expiratory volume in 1 second (FEV1) versus time curve (AUC FEV1)] were observed with inhaled formoterol 12μg twice daily (the approved dosage in the US) compared with placebo in 12-week and 12-month, randomized, double-blind trials in patients with chronic obstructive pulmonary disease (COPD).
The bronchodilator efficacy of formoterol 12μg twice daily was greater than that of oral slow-release theophylline (individualized dosages) in a 12-month trial or inhaled ipratropium bromide 40μg four times daily in a 12-week trial. Improvement in AUC FEV1 with formoterol, but not theophylline, compared with placebo was observed in patients with irreversible or poorly-reversible airflow obstruction. Formoterol also significantly improved health-related quality of life compared with ipratropium bromide or placebo and significantly reduced symptoms compared with placebo. Combination therapy with formoterol 12μg twice daily plus ipratropium bromide 40μg four times daily was significantly more effective than albuterol (salbutamol) 200μg four times daily plus the same dosage of ipratropium bromide in a 3-week, randomized, double-blind, double-dummy, crossover trial.
Inhaled formoterol was well tolerated in clinical trials. The incidence of investigator-determined drug-related adverse events with inhaled formoterol 12μg twice daily was similar to that with placebo and inhaled ipratropium bromide 40μg four times daily but lower than that with oral slow-release theophylline (individualized dosages). Importantly, there were no significant differences between formoterol and placebo or comparator drugs in cardiovascular adverse events in patients with COPD and corrected QT interval values within the normal range.
In conclusion, inhaled formoterol improved lung function and health-related quality of life and reduced symptoms relative to placebo in clinical trials in patients with COPD. The drug had greater bronchodilator efficacy than oral slow-release theophylline or inhaled ipratropium bromide and showed efficacy in combination with ipratropium bromide. The adverse events profile (including cardiovascular adverse events) with formoterol was similar to that with placebo. Thus, inhaled formoterol may be considered as a first-line option for the management of bronchoconstriction in patients with COPD who require regular bronchodilator therapy for the management of symptoms.
Pharmacodynamic Properties
Inhaled formoterol is a long-acting selective β2-adrenoceptor agonist (β2-agonist); it has a rapid onset of action (5 minutes in single- and multiple-dose studies) and, like salmeterol, maintains a bronchodilator effect for at least 12 hours. The onset of postdose bronchodilator action was faster with formoterol 12μg than with salmeterol 100μg in a double-blind, randomized, placebo-controlled trial.
Formoterol 6 to 24μg improved forced expiratory volume in 1 second (FEV1) compared with baseline and placebo in single-dose crossover trials in patients with chronic obstructive pulmonary disease (COPD), and was at least as effective as salmeterol 50 or 100μg or albuterol (salbutamol) 400μg at improving FEV1. Mean peak FEV1 was reached 1 hour after inhalation of formoterol 12μg; values for this parameter were 1 hour after albuterol 200μg, and 2 to 5 hours after salmeterol 50μg.
Formoterol 4.5 to 18μg twice daily for 1 week prolonged the time to exhaustion on a bicycle ergometer test compared with placebo; results were similar to those for ipratropium bromide 80μg three times daily.
All β2-agonists have the potential to increase heart rate and plasma glucose concentrations, and to decrease plasma potassium concentrations, through effects on extrapulmonary β2 receptors. Dose-dependent increases in heart rate, corrected QT (QTc) interval and plasma glucose concentrations, and dose-dependent decreases in plasma potassium concentrations, were observed with inhaled formoterol 24 to 96μg or salmeterol 100 to 400μg in a double-blind, placebo-controlled, crossover trial in 16 healthy volunteers. In patients with COPD, pre-existing mild to moderate cardiac arrhythmias and hypoxemia [PaO2 (arterial oxygen pressure) <60mm Hg], formoterol 12μg had similar systemic effects to salmeterol 50μg. Complex ventricular arrhythmias were observed in formoterol 12 and 24μg recipients, but not in salmeterol 50μg or placebo recipients.
Pharmacokinetic Properties
The maximum plasma concentration (92 ng/L) of formoterol was reached within 5 minutes of inhalation of a single supraoptimal dose (120μg) in 12 healthy volunteers. Urinary excretion data suggest that absorption was linear with inhaled formoterol 12 to 96μg in ten healthy volunteers. In vitro plasma protein binding of formoterol was 61 to 64% at concentrations 0.1 to 100 μg/L.
Mean plasma concentrations of the drug at 10 minutes to 6 hours postinhalation were 4.0 to 8.8 ng/L and 8.0 to 17.3 ng/L, respectively, after multiple doses of formoterol 12 or 24μg administered twice daily for 12 weeks in patients with COPD, with some evidence of accumulation of formoterol in the plasma (accumulation index 1.19 to 1.38).
Formoterol is metabolized primarily in the liver by four cytochrome P450 (CYP) isoenzymes (CYP2D6, CYP2C19, CYP2C9 and CYP2A6). These enzymes were not inhibited by the drug at therapeutic concentrations. Following inhalation of formoterol 12 or 24μg by 18 patients with COPD, 7% of the total dose was excreted in the urine as unchanged drug and 6 to 9% of the total dose was eliminated as direct conjugates of formoterol. The mean terminal elimination half-life was determined to be 10 hours (based on plasma concentrations) following inhalation of single-dose formoterol 120μg by 12 healthy volunteers.
Currently, there are no pharmacokinetic data for the use of formoterol in patients with hepatic or renal impairment or in elderly individuals.
Therapeutic Efficacy
Inhaled formoterol has been evaluated as monotherapy or combination therapy for the management of patients with COPD. In clinical trials, COPD was diagnosed using the American Thoracic Society guidelines.
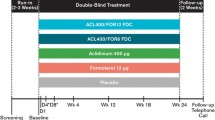
The bronchodilator effect [measured as normalized area under the FEV1 versus time curve (AUC FEV1)] with formoterol 12μg twice daily (n = 194) was significantly greater than that with ipratropium bromide 40μg four times daily (n = 194; p = 0.001) or placebo (n = 200; p < 0.001) in a randomized, double-blind, 12-week trial in patients with COPD. Significant improvements were also observed in mean morning premedication peak expiratory flow (PEF; p < 0.001) and health-related quality of life [all three subsections of the St. George’s Respiratory Questionnaire (SGRQ); p ≤ 0.036], and significant reductions were reported for the use of rescue medication (p ≤ 0.014) and the percentage of ‘bad days’ (days with at least two individual symptom scores of ≥2 and/or a reduction in PEF from baseline of >20%; p < 0.001) in formoterol compared with ipratropium bromide recipients. The differences in health-related quality of life between the two treatments were clinically relevant (exceeding 4 points) for the Activity and the Impacts domains of the SGRQ.
Compared with oral slow-release theophylline (individualized dosages targeted at plasma concentrations of 8 to 20 mg/L), formoterol 12μg twice daily significantly increased standardized AUC12h FEV1 (primary end-point; p = 0.026) and mean morning premedication PEF (p ≤ 0.020) and reduced the percentage of ‘bad days’ (p ≤ 0.035) in a randomized, double-blind (with the exception of the theophylline arm), 12-month trial. A subgroup analysis in this trial indicated that at 3 (p = 0.007) and 12 months (p = 0.002), formoterol (n = 118), but not oral slow-release theophylline (n = 105), produced significant bronchodilation compared with placebo (n = 117) in patients with irreversible or poorly-reversible airflow obstruction (i.e. patients whose FEV1 values increased <15% after receiving albuterol). Both formoterol (p ≤ 0.026) and oral slow-release theophylline (p ≤ 0.013) were significantly more effective than placebo at managing COPD during the night (measured as morning premedication FEV1).
In these two monotherapy trials, inhaled formoterol 24μg twice daily did not provide any additional benefit over the 12μg twice daily dosage in patients with COPD.
The combined efficacy of inhaled formoterol 12μg twice daily plus inhaled ipratropium bromide 40μg four times daily for 3 weeks has been compared with that of albuterol 200μg four times daily for 3 weeks via a pressurized metered-dose inhaler plus inhaled ipratropium bromide 40μg four times daily in a randomized, double-blind, double-dummy, crossover trial in 172 patients with COPD. Formoterol combination therapy was significantly more effective than albuterol combination therapy at increasing mean morning premedication PEF (primary endpoint; p = 0.0003). Combination therapy with formoterol was also more effective according to secondary endpoints, significantly increasing postmedication FEV1 to 6 hours (p< 0.0001), peak postmedication FEV1 (p < 0.0001) and AUC FEV1 (p < 0.0001) and improving symptoms of COPD (measured as mean total symptoms score, p = 0.0042) and the SGRQ symptoms score (p = 0.0408) relative to albuterol combination therapy.
Tolerability
Inhaled formoterol was well tolerated in clinical trials in patients with COPD. The percentage of patients experiencing at least one adverse event with inhaled formoterol 12μg twice daily was similar to that with placebo, inhaled ipratropium bromide 40μg four times daily or oral slow-release theophylline (individualized dosages targeted at plasma concentrations of the drug of 8 to 20 mg/L) in randomized, double-blind, comparative trials of 12 weeks’ and 12 months’ duration. Viral infection, exacerbation of COPD, bronchitis, upper respiratory tract infection, dyspnea and headache were the most commonly reported adverse events (i.e. occurring in >5% of formoterol 12μg twice daily recipients); however, the incidence of these events was not significantly different compared with oral slow-release theophylline or placebo.
Drug-related adverse events, serious adverse events and events leading to withdrawal from the study occurred with a similar incidence with inhaled formoterol, placebo or ipratropium bromide. In contrast, drug-related adverse events and withdrawal because of adverse events occurred with a higher incidence in patients receiving oral slow-release theophylline in the 12-month trial.
There were no significant differences in the incidence of cardiovascular adverse events with inhaled formoterol (0.5% of patients) compared with inhaled placebo (2.5%) or ipratropium bromide (2.6%) after 12 weeks’ treatment or in the incidence of serious cardiovascular adverse events with inhaled formoterol (2.4% of patients) compared with placebo (0.9%) or oral slow-release theophylline (2.4%) after 12 months’ treatment in patients with COPD and QTc interval values within the normal range; heart rate and rhythm disorders were infrequent. The incidences of QTc interval prolongation (>0.46s), ECG abnormalities or clinically relevant changes in serum potassium or fasting plasma glucose concentrations were similar with inhaled formoterol 12μg twice daily compared with placebo, inhaled ipratropium bromide or oral slow-release theophylline in clinical trials.
Dosage and Administration
Formoterol, inhaled orally using an Aerolizer™1 inhaler, is indicated in the US for the long-term maintenance treatment of bronchoconstriction associated with COPD (including chronic bronchitis and emphysema). The recommended dosage of formoterol in this patient group is 12μg twice daily approximately 12 hours apart; the total daily dose should not exceed 24μg.
Formoterol should be used with caution in patients with cardiovascular disorders (especially coronary insufficiency, cardiac arrhythmias or hypertension), convulsive disorders or thyrotoxicosis, or hypersensitivity to sympathomimetic amines. Extreme caution is advised if formoterol is used concomitantly with monoamine oxidase inhibitors, tricyclic antidepressants or drugs that are known to prolong the QTc interval, and caution is recommended with the concomitant use of formoterol and non-potassium-sparing diuretics.






Similar content being viewed by others
Notes
Use of tradenames is for product identification purposes only and does not imply endorsement
References
Friedman M, Hilleman DE. Economic burden of chronic obstructive pulmonary disease: impact of new treatment options. Pharmacoeconomics 2001; 19(3): 245–54
Hilleman DE, Dewan N, Malesker M, et al. Pharmacoeconomic evaluation of COPD. Chest 2000; 118: 1278–85
Pauwels RA, Buist AS, Calverley PMA, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: National Heart, Lung, and Blood Institute and World Health Organization Global Initiative for Chronic Obstructive Lung Disease (GOLD): Executive Summary. Respir Care 2001 Aug; 46(8): 798–825
Johnson M, Rennard S. Alternative mechanisms for long-acting β2-adrenergic agonists in COPD. Chest 2001 Jul; 120(1): 258–70
Celli BR, Snider GL, Heffner J, et al. Standards for the diagnosis and care of patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med 1995; 152 (5 Pt 2): S77–120
The COPD Guidelines Group of the Standards of Care Committee of the BTS. BTS guidelines for the management of chronic obstructive pulmonary disease. Thorax 1997; 52Suppl. 5: S1–28
Siafakas NM, Vermeire P, Pride NB, et al. Optimal assessment and management of chronic obstructive pulmonary disease (COPD). Eur Respir J 1995; 8: 1398–420
Madison MJ, Irwin RS. Chronic obstructive pulmonary disease. Lancet 1998; 352: 467–73
Barnes PJ. Chronic obstructive pulmonary disease. N Engl J Med 2000; 343(4): 269–80
Cazzola M, Santangelo G, Piccolo A, et al. Effect of salmeterol and formoterol in patients with chronic obstructive pulmonary disease. Pulm Pharmacol 1994 Apr; 7(2): 103–7
Faulds D, Hollingshead LM, Goa KL. Formoterol: a review of its pharmacological properties and therapeutic potential in reversible obstructive airways disease. Drugs 1991 Jul; 42(1): 115–37
Bartow RA, Brogden RN. Formoterol: an update of its pharmacological properties and therapeutic efficacy in the management of asthma. Drugs 1998; 55(2): 303–22
Novartis Pharmaceuticals. Foradil aerolizer: formoterol fumarate inhalation powder [web page]. Available from URL: http://www.pharma.us.novartis.com/product/pi/pdf/foradil.pdf [Accessed 2002 Mar 4]
Anderson GP. Formoterol: pharmacology, molecular basis of agonism, and mechanism of long duration of a highly potent and selective β2-andrenoceptor agonist bronchodilator. Life Sci 1993; 52: 2145–60
Cazzola M, Di Perna F, Califano C, et al. Formoterol Turbuhaler (F) vs salbutamol Diskus (S) in patients with partially reversible stable COPD. Am J Respir Crit Care Med 1999; 159 (3 Suppl. 2): A798
Çelik G, Kayacan O, Beder S, et al. Formoterol and salmeterol in partially reversible chronic obstructive pulmonary disease: a crossover, placebo-controlled comparison of onset and duration of action. Respiration 1999; 66(5): 434–9
Kottakis J, Cioppa GD, Creemers J, et al. Faster onset of bronchodilation with formoterol than with salmeterol in patients with stable, moderate to severe COPD: results of a randomized, double-blind, clinical study. Can Res J 2002; 9(2): 107–15
Cazzola M, Matera MG, Santangelo G, et al. Salmeterol and formoterol in partially reversible severe chronic obstructive pulmonary disease: a dose-response study. Respir Med 1995 May; 89(5): 357–62
Kottakis I, Bouros D, Siafakas N, et al. Effects of formoterol and salmeterol on inspiratory capacity: results from a single-dose, 5-period cross-over study in stable, poorly reversible, stage II and III COPD patients [abstract]. Am J Respir Crit Care Med 2001 Apr; 163(Suppl. 2): S278
Guhan AR, Cooper S, Oborne J, et al. Systemic effects of formoterol and salmeterol: a dose-response comparison in healthy subjects. Thorax 2000; 55: 650–6
Benhamou D, Cuvelier A, Muir JF, et al. Rapid onset of bronchodilation in COPD: a placebo-controlled study comparing formoterol (Foradil Aerolizer) with salbutamol (Ventodisk). Respir Med 2001 Oct; 95(10): 817–21
Sichletidis L, Kottakis J, Marcou S, et al. Bronchodilatory responses to formoterol, ipratropium, and their combination in patients with stable COPD. Int J Clin Pract 1999 Apr-1999 31; 53(3): 185–8
Rossi A, Kristufek P, Levine BE, et al. Comparison of the efficacy, tolerability and safety of formoterol dry powder and oral slow-release theophylline in the treatment of COPD. Chest 2002; 121(4): 1058–69
Dahl R, Greefhorst LA, Nowak D, et al. Inhaled formoterol dry powder versus ipratropium bromide in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2001 Sep 1; 164(5): 778–84
Liesker JWW, van de Velde V, Meysmann M, et al. The effects of formoterol on excercise capacity in COPD patients [abstract]. Eur Respir J 1999; 14: 3795
Bowden JJ, Sulakvelidze I, McDonald DM. Inhibition of neutrophil and eosinophil adhesion to venules of rat trachea by β2-adrenergic agonist formoterol. J Appl Physiol 1994 Jul; 77(1): 397–405
Anderson R, Feldman C, Theron AJ, et al. Anti-inflammatory, membrane-stabilizing interactions of salmeterol with human neutrophils in vitro. Br J Pharmacol 1996 Apr; 117(7): 1387–94
Rabe KF, Giembycz MA, Dent G, et al. Salmeterol is a competitive agonist at β-adrenoceptors mediating inhibition of respiratory burst in guinea-pig eosinophils. Eur J Pharmacol 1993 Feb 9; 231(2): 305–8
Okada C, Sugiyama H, Eda R, et al. Effect of formoterol on Superoxide anion generation from bronchoalveolar lavage cells after antigen challenge in guinea pigs. Am J Respir Cell Mol Biol 1993 May; 8(5): 509–17
Seider N, Abinader EG, Oliven A. Cardiac arrhythmias after inhaled bronchodilators in patients with COPD and ischemic heart disease. Chest 1993; 104: 1367–73
Cazzola M, Imperatore F, Salzillo A, et al. Cardiac effects of formoterol and salmeterol in patients suffering from COPD with preexisting cardiac arrhythmias and hypoxemia. Chest 1998 Aug; 114(2): 411–5
Lecaillon JB, Kaiser G, Palmisano M, et al. Pharmacokinetics and tolerability of formoterol in healthy volunteers after a single high dose of Foradil dry powder inhalation via aerolizer. Eur J Clin Pharmacol 1999; 55: 131–8
D’Urzo AD, De Salvo MC, Ramirez-Rivera A, et al. In patients with COPD, treatment with a combination of formoterol and ipratropium is more effective than a combination of salbutamol and ipratropium: a 3-week, randomized, double-blind, within-patient, multicenter study. Chest 2001 May; 119(5): 1347–56
Novartis Pharmaceuticals Corporation. Novartis’ Foradil gains FDA approval for the treatment of chronic obstructive pulmonary disease (COPD). Media Rel 2001 Sep 27
Jones PW, Quirk FH, Baveystock CM. The St George’s Respiratory Questionnaire. Respir Med 1991; 85Suppl. B: 25–31
Anthonisen NR, Connett JE, Kiley JP, et al. Effects of smoking intervention and the use of an inhaled anticholinergic bronchodilator on the rate of decline of FEV1. Lung Health Study. JAMA 1994; 272: 1497–505
Bonay M, Bancal C, Crestani B. Benefits and risks of inhaled corticosteriods in chronic obstructive pulmonary disease. Drug Saf 2002; 25(1): 57–71
Burge S. Should inhaled corticosteroids be used in the long term treatment of chronic obstructive pulmonary disease? Drugs 2001; 61(11): 1535–44
Pauwels RA, Löfdahl C-G, Laitinen LA, et al. Long-term treatment with inhaled budesonide in persons with mild chronic obstructive pulmonary disease who continue smoking. N Engl J Med 1999; 340: 1948–53
Vestbo J, Sørensen T, Lange P, et al. Long-term effect of inhaled budesonide in mild and moderate chronic obstructive pulmonary disease: a randomised controlled trial. Lancet 1999; 353: 1819–23
Burge PS, Calverley PMA, Jones PW, et al. Randomised, doubleblind, placebocontrolled study of fluticasone propionate in patients with moderate to severe chronic obstructive pulmonary disease: the ISOLDE trial. BMJ 2000; 320: 1297–303
Jarvis B, Markham A. Inhaled salmeterol: a review of its efficacy in chronic obstructive pulmonary disease. Drugs Aging 2001; 18(6): 441–72
Cazzola M, Vinciguerra A, Di Perna F, et al. Early reversibility to salbutamol does not always predict bronchodilation after salmeterol in stable chronic obstructive pulmonary disease. Respir Med 1998; 92: 1012–6
Dowling RB, Johnson M, Cole PJ, et al. Effect of salmeterol on Haemophilus influenzae infection of respiratory mucosa in vitro. Eur Respir J 1998; 11: 86–90
Ferguson GT. Update on pharmacologic therapy for chronic obstructive pulmonary disease. Clin Chest Med 2000; 21(4): 723–38
Hanania NA, Ramsdell J, Payne K, et al. Improvements in airflow and dyspnea in COPD patients following 24 weeks treatment with salmeterol 50μg and fluticasone propionate 250μg alone or in combination via the Diskus [abstract no. 884786]. Am J Respir Crit Care Med 2001 Apr; 163Suppl. 2: A279
Wire P, Yates J, Fischer T, et al. The combination of fluticasone propionate and salmeterol administered with a single diskus is safe and effective twice-daily maintenance treatment for COPD [abstract no. 887942]. Chest 2001 Oct; 120 Suppl.: 162S–3S
Mahler DA, Wong E, Giessel G, et al. Improvements in FEV1 and symptoms in COPD patients following 24 weeks of twice daily treatment with salmeterol 50/fluticasone propionate 500 combination [abstract no. 884785]. Am J Respir Crit Care Med 2001 Apr; 163Suppl. 2: 279
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cheer, S.M., Scott, L.J. Formoterol. Am J Respir Med 1, 285–300 (2002). https://doi.org/10.1007/BF03256622
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03256622