Abstract
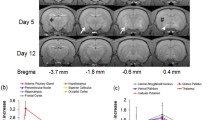
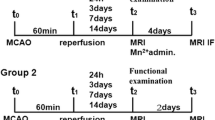
Manganese enhanced magnetic resonance imaging (MRI) is a novel neuroimaging technique that can be usedin vivo to trace neuronal tract and to study brain functions dynamically. In this study, manganese enhanced MRI was used to trace the neuronal tracts between the laminar structures of the olfactory bulb (OB) in rat and to study the so-called “calcium overload” phenomenon in a rat model of cerebral ischemia. High spatial resolution images of the OB were obtained and used to measure the transportation rate of Mn2+ among the laminar structures of the OB, which was shown to be approximately 0.2 mm/h under resting condition. In the rat focal ischemia study, it was found that the total area of brain regions with Mn2+ accumulation (representing brain regions with calcium overload) was only 55%±15% of the area of the ischemic brain regions shown by diffusion-weighted imaging (DWI). Manganese enhanced MRI might be more accurate than DWI in detecting the ischemic core at the early stage of experimental cerebral ischemia.
Similar content being viewed by others
References
Lin, Y. J., Koretsky, A. P., Manganese ion enhances T1-weighted MRI during brain activation: an approach to direct imaging of brain function, Magn. Reson. Med., 1997, 38(3): 378–388.
Hunter, D. R., Komai, H., Haworth, R. A. et al., Comparison of Ca2+, Sr2+, and Mn2+ fluxes in mitochondria of the perfused rat heart, Circ. Res., 1980, 47(5): 721–727.
Pautler, R. G., Koretsky, A. P., Tracing odor-induced activation in the olfactory bulbs of mice using manganese-enhanced magnetic resonance imaging, Neurolmage, 2002, 16(2): 441–448.
Narita, K., Kawasaki, F., Kita, H., Mn and Mg influxes through Ca channels of motor nerve terminals are prevented by verapamil in frogs, Brain Res., 1990, 510(2): 280–295.
Drapeau, P., Nachshen, D. A., Manganese fluxes and manganese-dependent neurotransmitter release in presynaptic nerve endings isolated from rat brain, J. Physiol., 1984, 348: 493–510.
Geraldes, C. F., Sherry, A. D., Brown, R. D., Magnetic field dependence of solvent proton relaxation rates induced by Gd3+ and Mn2+ complexes of various polyaza macrocyclic ligands: Implication for NMR imaging, Magn. Reson. Med., 1986, 3(2): 242–250.
Pautler, R. G., Silva, A. C., Koretsky, A. P., In vivo neuronal tract tracing using manganese-enhanced magnetic resonance imaging, Magn. Reson. Med., 1998, 40(5): 740–748.
Van der Linden, A., Verhoye, M., Van Meir, V. et al., In vivo manganese-enhanced magnetic resonance imaging reveals connections and functional properties of the songbird vocal control system, Neuroscience, 2002, 112(2): 467–474.
Tindemans, I., Verhoye, M., Balthazart, J. et al., In vivo dynamic ME-MRI reveals differential functional responses of RA- and area X-projecting neurons in the HVC of canaries exposed to conspecific song, Eur. J. Neurosci., 2003, 18(12): 3352–3360.
Van Meir, V., Verhoye, M., Absil, P. et al., Differential effects of testosterone on neuronal populations and their connections in a sensorimotor brain nucleus controlling song production in song-birds: A manganese enhanced-magnetic resonance imaging study, Neuroimage, 2004, 21(3): 914–923.
Duong, T. Q., Silva, A. C., Lee, S. P. et al., Functional MRI of calcium-dependent synaptic activity: Cross correlation with CBF and BOLD measurements, Magn. Reson. Med., 2000, 43(3): 383–392.
Aoki, C., Ebisu, T., Tanaka, C. et al., Detection of the anoxic depolarization of focal ischemia using manganese-enhanced MRI, Magn. Reson. Med., 2003, 50(1): 7–12.
Longa, E. Z., Weinstein, P. R., Carlson, S. et al., Reversible middle cerebral artery occlusion without craniectomy in rats, Stroke, 1989, 20(1): 84–91.
McGivern, J., Scholfield, C. N., Action of general anaesthetics on unclamped Ca2+-mediated currents in unmyelinated axons of rat olfactory cortex, Eur. J. Pharmacol., 1991, 203(1): 59–65.
Gerdin, B., McCann, E., Lundberg, C. et al., Selective tissue accumulation of manganese and its effect on regional blood flow and haemodynamics after intravenous infusion of its chloride salt in the rat, Int. J. Tissue React., 1985, 7(3): 373–380.
Sims, N. R., Zaidan, E., Biochemical changes associated with selective neuronal death following short-term cerebral ischaemia, Int. J. Biochem. Cell Biol., 1995, 27(6): 531–550.
Lipton, P., Ischemic cell death in brain neurons, Physiol. Rev., 1999, 79(4): 1431–1568.
Kohno, K., Hoehn-Berlage, M., Mies, G. et al., Relationship between diffusion-weighted MR images, cerebral blood flow, and energy state in experimental brain infarction, Magn. Reson. Imaging, 1995. 13(1): 73–80.
Busch, E., Kruger, K., Allegrini, P. R. et al., Reperfusion after thrombolytic therapy of embolic stroke in the rat: magnetic resonance and biochemical imaging, J. Cereb. Blood Flow Metab., 1998, 18(4): 407–418.
Kidwell, C. S., Saver, J. L., Mattiello, J. et al., Thrombolytic reversal of acute human cerebral ischemic injury shown by diffusion/perfusion magnetic resonance imaging, Ann. Neurol., 2000, 47(4): 462–469.
Harris, R. J., Symon, L., Extracellular pH, potassium, and calcium activities in progressive ischaemia of rat cortex, J. Cereb. Blood Flow Metab., 1984, 4(2): 178–186.
Newland, M. C., Cox, C., Hamada, R. et al., The clearance of manganese chloride in the primate, Fundam. Appl. Toxicol., 1987, 9(2): 314–328.
Aschner, M., Aschner, J. L., Manganese neurotoxicity: cellular effects and blood-brain barrier transport, Neurosci. Biobehav. Rev., 1991, 15(3): 333–340.
Brurok, H., Schjitt, J., Berg, K. et al., Manganese and the heart: acute cardiodepression and myocardial accumulation of manganese, Acta Physiol. Scand., 1997, 159(1): 33–40.
Skjold, A., Vangberg, T., Kristoffersen, A. et al., Preliminary experience with intracellular manganese ions as contrast agents in human myocardium, 12th Annual Meeting of International Society for Magnetic Resonance in Medicine, 2004, Kyoto, Japan.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Fang, K., Li, Y., Liu, H. et al. Tracing neuronal tracts in the olfactory pathway of rat and detecting ischemic core in a rat model of focal ischemia using manganese enhanced magnetic resonance imaging. Chin.Sci.Bull. 49, 1834–1840 (2004). https://doi.org/10.1007/BF03183410
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03183410