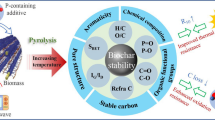
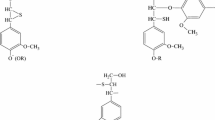
Summary
-
(i)
When cellulose is oxidised with a mixture of nitrogen dioxide and oxygen the products obtained (I) dissolve in dilute alkali when the carboxyl content is about 9% as compared with a minimum of 13·5% required for alkali solubility in case of nitrogen dioxide oxycelluloses (II).
-
(ii)
I usually possesses a higher percentage of combined nitrogen than II.
-
(iii)
The yellow colour developed on treatment with hot dilute caustic soda disappears more or less completely if the oxycelluloses (except I) are treated with chlorous acid. In case of I, the intensity of the colour is diminished considerably.
-
(iv)
There is a greater decrease in cuprammonium fluidity on treatment with chlorous acid with I and periodic acid oxycelluloses as compared with II and dichromate oxalic acid oxycelluloses.
-
(v)
On treatment with alkali there is a fall in copper number both in the case of I and periodic acid oxycellulose while an increase in copper number takes place in the case of II and dichromate-oxalic acid oxycellulose. Based on the above conclusions a tentative mechanism of oxidation has been suggested for the oxidation of cellulose with a mixture of nitrogen dioxide and oxygen.
Similar content being viewed by others
References
Kenyon,et al. J. Amer. Chem. Soc., 1942,64, 121, 127.
—Ibid., 1947,69, 343,347, 349.
Kenyon,et al. Ibid., J. Amer. Chem. Soc., 355.
—Ibid.,, 1948,70, 2100.
HeadJ. Chem. Soc., 1948, 1135.
Rowen, Hunt and PlylerJ. Res. Natl. Bur. Standards, 1947,39, 133.
Maurer and ReiffJ. Makromol. Chemie, 1943,1, 27; cf.Chem. Abstr., 1944,38, 1211.
Nabar and Padmanabhan This Journal, 1950,31, 371.
DavidsonJ. Text. Inst., 1948,39, T 102.
Doreé “Methods of Cellulose Chemistry,” Chapman & Hall, London, 1947, p. 247.
HeyesJ. Soc. Chem. Ind., 1928,47, T 90.
Neale and StringfellowTrans. Faraday. Soc., 1937,33, 881.
Kenyon,et al., Text. Res. J., 1946,16, 1.
DavidsonJ. Text. Inst., 1948,39, T 59.
Pacsu and MehtaText. Res. J. 1948,18, 387.
Pacsu and Mehta “The Viscosity of Cellulose Solutions,” Fabric Research Committee, 1932, London; see alsoJ. Text. Inst., 1936,27, T 285.
Kenyon,et al. J. Amer. Chem. Soc., 1942,64, Fig. 5, p. 125.
TraubeBer., 1885,15, 659.
Manchot and HerzogAnnalen, 1901,316, 318.
Baeyer and WilligerBer., 1900,33, 1569.
KauffmannIbid.,, 1932,65, 179.
Wieland and FränkeAnnalen, 1928,464, 101.
Engler and Weissberg “Krittische Stüdien über die Vorgangen der Auto-oxidation,” Branschweig, 1904.
Engler,et al. Ber., 1897,30, 1699; 1894,31, 3046; 3055; 1900,33, 1090, 1097, 1109; 1901,34, 2933.
BerthelotCompt. rend., 1900,131, 519.
Kenyon and GrayJ. Amer. Chem. Soc., 1936,58, 1422.
Silberad and FarmerJ. Chem. Soc., 1906,89, 1759.
CarlsonBer., 1907,40, 4192.
HaussermannChem. Ztg., 1905,29, 421.
Richardson, Higgin Botham and FarrowJ. Text. Inst., 1936,27, 131 T.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nabar, G.M., Padmanabhan, C.V. Studies in oxycellulose. Proc. Indian Acad. Sci. (Math. Sci.) 32, 212 (1950). https://doi.org/10.1007/BF03170824
Received:
DOI: https://doi.org/10.1007/BF03170824