Abstract
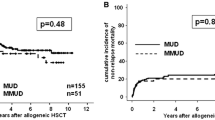
Patients with advanced hematological malignancies ineligible for conventional myeloablative allogeneic hematopoietic stem cell transplantation (HSCT) due to advanced age or medical contraindications were enrolled in a multi-center study to investigate the safety and efficacy of nonmyeloablative HSCT using a 2 Gy total body irradiation (TBI)-based regimen. A total of 192 patients (median age 55) were treated with HLA-matched sibling peripheral blood stem cell (PBSC) grafts, and 63 patients (median age 53) received a 10 of 10 HLA-antigen matched unrelated donor (URD) HSCT (PBSC graft, n=48; marrow graft, n=15). Diagnoses included multiple myeloma (n=61), myelodysplastic syndrome (n=55), chronic myeloid leukemia (n=31), non-Hodgkin lymphoma (n=31), acute myeloid leukemia (n=28), chronic lymphocytic leukemia (n=24), Hodgkin Disease (n=14). The conditioning regimen was fludarabine 30 mg/m2/d × 3 days and 2 Gy TBI. Ninety-five related HSCT patients received 2 Gy TBI without fludarabine. Postgrafting immunosuppression was combined mycophenolate mofetil and cyclosporine. Transplants were well tolerated with a median of 0 days of hospitalization in the first 60 days for eligible patients. For related HSCT recipients, median follow-up was 289 (100–1188) days. Nonfatal graft rejection occurred in 6.8%. Of those with sustained engraftment, graft-versus-host disease (GVHD) occurred in 49% (33% grade II, 11% grade III, 5% grade IV). Day-100 non-relapse mortality was 6%. Overall, 59% (114/192) of patients were alive. The relapse/disease progression mortality was 18%, and non-relapse mortality was 22%. The projected 2-year survival and progression-free survival were 50% and 40%. For the URD HSCT recipients, median follow-up was 190 (100–468) days. Graft rejection occurred in 27% (17/63) of patients, mostly in recipients of marrow grafts (9/15). Acute GVHD occurred in 63% (50% grade II, 13% grade III) of 46 engrafted patients. Chronic GVHD requiring therapy occurred in 50% of patients. Of the 63 URD HSCT patients, 54% were alive, 37% in CR, 3% PR, and 14% with disease progression or relapse. Related and unrelated nonmyeloablative HSCT is feasible and potentially curative in patients with advanced hematological malignancies who have no other treatment options.
Similar content being viewed by others
References
Thomas ED, Storb R, Clift RA, et al. Bone-marrow transplantation.N Engl J Med. 1975;292:832–843, 895–902.
Ringdn O, Horowitz MM, Gale RP, et al. Outcome after allogeneic bone marrow transplant for leukemia in older adults.JAMA. 1993;270:57–60.
Math G, Amiel JL, Schwarzenberg L, Catton A, Schneider M. Adoptive immunotherapy of acute leukemia: Experimental and clinical results.Cancer Res. 1965;25:1525–1531.
Weiden PL, Flournoy N, Thomas ED, et al. Antileukemic effect of graft-versus-host disease in human recipients of allogeneic-marrow grafts.N Engl J Med. 1979;300:1068–1073.
Horowitz MM, Gale RP, Sondel PM, et al. Graft-versus-leukemia reactions after bone marrow transplantation.Blood. 1990; 75:555–562.
Martin PJ. Overview of marrow transplantation immunology. In: Thomas ED, Blume KG, Forman SJ (eds): Hematopoietic Cell Transplantation. Boston: Blackwell Science, p.19–27, 1999.
Storb R, Yu C, Wagner JL, et al. Stable mixed hematopoietic chimerism in DLA-identical littermate dogs given sublethal total body irradiation before and pharmacological immunosuppression after marrow transplantation.Blood. 1997;89:3048–3054.
McSweeney PA, Niederwieser D, Shizuru JA, et al. Hematopoietic cell transplantation in older patients with hematologic malignancies: replacing high-dose cytotoxic therapy with graft-versus-tumor effects.Blood. 2001;97:3390–3400.
Weissinger F, Sandmaier BM, Maloney DG, Bensinger WI, Gooley T, Storb R. Decreased transfusion requirements for patients receiving nonmyeloablative compared with conventional peripheral blood stem cell transplants from HLA-identical siblings.Blood. 2001;98:3584–3588.
Sandmaier BM, Maloney DG, Gooley T, et al. Nonmyeloablative hematopoietic stem cell transplants (HSCT) from HLA-matched related donors for patients with hematologic malignancies: clinical results of a TBI-based conditioning regimen [abstract].Blood. 2001;98 (Part 1):742a-743a.
Sandmaier BM, Hegenbart U, Shizuru J, et al. Nonmyeloablative hematopoietic stem cell transplantation (HSCT) from HLA-identical siblings for treatment of chronic myelogenous leukemia (CML): induction of molecular remissions [Abstract].Blood. 2001;98(Part 2):371b.
Maloney DG, Sahebi F, Stockerl-Goldstein KE, et al. Combining an allogeneic graft-vs- myeloma effect with high-dose autologous stem cell rescue in the treatment of multiple myeloma [Abstract].Blood. 200;98(Part 1):434a–435a.
Maris MB, Sandmaier BM, Niederwieser D, et al. The effect of donor lymphocyte infusions (DLI) on chimerism and persistent disease after nonmyeloablative hematopoietic stem cell transplant (HSCT) [Abstract].Blood. 2000;96(Part 1):477a.
Maris M, Niederwieser D, Sandmaier B, et al. Nonmyeloablative hematopoietic stem cell transplants (HSCT) using 10/10 HLA antigen matched unrelated donors (URDs) for patients with advanced hematologic malignancies ineligible for conventional HSCT [abstract].Blood. 2001;98(Part 1):858a.
Warren EH, Gavin M, Greenberg PD, Riddell SR. Minor histocompatibility antigens as targets for T-cell therapy after bone marrow transplantation.Curr Opin Hematol. 1998;5:429–433.
Greenberg PD, Riddell SR. Cellular Therapy. In: Austen KF, Burakoff SJ, Rosen FS, Strom TB (eds): Therapeutic Immunology. Malden, MA: Blackwell Science, Inc, p.550–573, 2001.
Author information
Authors and Affiliations
About this article
Cite this article
Georges, G.E., Maris, M., Sandmaier, B.M. et al. Related and unrelated nonmyeloablative hematopoietic stem cell transplantation for malignant diseases. Int J Hematol 76 (Suppl 1), 184–189 (2002). https://doi.org/10.1007/BF03165242
Issue Date:
DOI: https://doi.org/10.1007/BF03165242