Summary
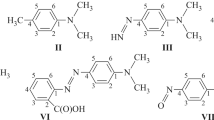
The dipole moments of mono- and disubstitutedpara-xylene derivatives have been measured in benzene and other solvents. The results show clearly that there is interaction between methyl hydrogen atoms and the substituents in the position ortho to the methyl. A comparison with mesitylene and durene derivatives indicates the need to consider also the symmetry of the molecules and the uncertainties of atomic polarsation in a full understanding of the question.
Similar content being viewed by others
References
Anantakrishnan, S. V. and Setu Rao, D.Curr. Sci., 1960,29, 51.
Bell and Buck ..J.C.S., 1956, 1890.
Böttcher, C. J. F. ..Dielectric Polarisation, (Elsevier), 1952.
Chen, J. H. and Petrakaus, A. A.J. Chem. Phys., 1959,30, 304.
Coulson, C. A. .. InHydrogen Bonding, Ed. by Hadzi, Pergamon, 1959, p. 341.
Crawford, M. and Stewart, F. H. C.Trans. Farad. Soc., 1953,49, 752.
Horning .. InOrganic Solvents, Ed. by Weissberger, Interscience, 1955.
Jen Yuan Chein..J. Chem. Educ., 1947,24, 494.
Marzin..J. Prakt Chem., 1933,138, 103.
Paoloni, L...J.A.C.S., 1958,80, 3879.
Smith, J. W. ..Electric Dipole Moments, Butterworths, 1955, p. 46.
----- ..Ibid., 1955, p. 218.
Smyth, C. P. ..Dielectric Behaviour and Structure, Reinhold, 1955.
Sutton, L. E. ..J.C.S., 1938, 1269.
—— and Hampson, G. C.Trans. Farad. Soc., 1935,31, 945.
Szasz, G. J...J. Chem. Phys., 1955,23, 2449.
Varma, P. S...J. Ind. Chem. Soc., 1926,3, 291, 342.
—————.. Ibid., 1935,12, 343.
Wahl..Ann. Chem., 1936,5, 13.
Zanker, V. and Wittwer, A. InHydrogen Bonding, Ed. by Hadzi, Pergamon, 1955, p. 286.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Anantakrishnan, S.V., Rao, D.S. Dipole moments of some derivatives ofpara-xylene—A possible intramolecular hydrogen bonding. Proc. Indian Acad. Sci. 60, 201–210 (1964). https://doi.org/10.1007/BF03046408
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03046408