Abstract
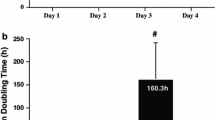
We analyzed changes in polyamine contents and the activities of biosynthetic enzymes during each phase of the cell cycle for a synchronized population of BY-2 cells. Based on our analysis of H3-thymidine incorporation flow cytometry, and the mitotic index, the M and G2 phases seemed to occur at 8 h and from 2.5 to 8 h, respectively, after the release of aphidicolin. The respective activities of arginine decarboxylase (ADC), Ornithine decarboxylase (ODC), and S-adenosyl methionine decarboxylase (SAMDC) at the beginning (7.4, 11.2, and 5.5 nmol mg-1 protein h-1) were increased to 22.6, 22.1, and 15.1 nmol mg-1 protein h-1. However, those increases do not coincide with the general change in polyamines reported from animal cells. In addition, the bi-phasic activation of polyamine biosynthetic enzymes, such as those found in the general animal model, was observed with ADC and ODC but not with SAMDC. These results suggest that the general animal model for explaining polyamine changes and SAMDC activation in the cell cycle cannot be applied to BY-2 cells. Further, our flow-cytometric analysis of cell populations may be a useful tool for evaluating the effects of polyamines on cell cycle progression in BY-2 cells.
Similar content being viewed by others
Abbreviations
- ADC:
-
Arginine decarboxylase
- DcSAM:
-
decarboxylated S-adenosyl methionine
- ODC:
-
Ornithine decarboxylase
- SAM:
-
S-adenosyl methionine
- SAMDC:
-
S-adenosyl methionine decarboxylase
Literature cited
Ackermann JM, Pegg AE, McCloskey DE (2003) Drugs affecting the cell cycle via actions on the polyamine metabolic pathway. Pro Cell Cyc Res5: 461–468
Ahn SM, Jin CD (2004) Protective role of exogenous spermidine against paraquat toxicity in radish chloroplasts. J Plant Biol47: 338–347
Amarantos I, Zarkadis IK, Kalpaxis DL (2002) The identification of spermine binding sites in 16S rRNA allows interpretation of the spermine effect on ribosomal 30S subunit functions. Nucl Acid Res30: 2832–2843
Anderson SE, Bastola DR, Minocha SC (1998) Metabolism of polyamines in transgenic cells of carrot expressing a mouse Ornithine decarboxylase cDNA. Plant Physiol116: 299–307
Anehus S, Pohjanpelto P, Baldetorp B, Längström E, Heyb O (1984) Polyamine starvation prolongs the S and G2 phases of polyamine-dependent (arginase-deficient) CHO cells. Cell Biol4: 915–922
Borrell A, Bedford RT, Altabella T, Masgrau C, Tiburicio AF (1996) Regulation of arginine decarboxylase by spermine in osmotically stressed oat leaves. Physiol Plant98: 105–110
Bradford MM (1976) A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dyebinding. Anal Biochem72: 248–254
Chattopadhyay MK, Tabor CW, Tabor H (2002) Absolute requirement of spermidine for growth and cell cycle progression of fission yeast (Schizosaccharomyces pombe). Proc Natl Acad Sci USA99: 10330–10334
Ehsan H, Roef L, Witters E, Reichheld JP, van Bockstaele D, Inzé D, van Onckeln H (1999) Indomethacin-induced G1/S phase arrest of the plant cell cycle. FEBS Lett458: 349–353
Filiingame RH, Jorstad CM, Morris DR (1975) Increased cellular contents of spermidine or spermine are required for optimal DNA synthesis in lymphocytes activated by concanavalin A. Proc Natl Acad Sci USA72: 4042–4045
Jang SJ, Choi YJ, Park KY (2002) Effects of polyamines on shoot and root development inArabidopsis seedlings and carnation cultures. J Plant Biol45: 230–236
Jang SJ, Shin SH, Yee ST, Hwang B, Im KH, Park KY (2005) Effects of biotic stresses on cell cycle progression in tobacco BY-2 cells. Mol Cells20: 136–141
Kim YJ, Lee SH, Park KY (2004) A leader intron and 115-bp promoter region necessary for expression of the carnation S-adenosylmethionine decarboxylase gene in the pollen of transgenic tobacco. FEBS Lett578: 229–235
Lee MM, Lee SH, Park KY (1997) Characterization and expression of two members of the S-adenosylmethionine decarboxylase gene family in carnation flower. Plant Mol Biol34: 371–382
Lee SH, Kim YB, Lee MM, Park KY (1996) The stimulation of arginine decarboxylation by dfmo in tobacco suspension cultured cells. J Plant Biol39: 107–112
Mad Arif SA, Taylor MA, George LA, Butler AR, Burch LR, Davis HV, Stark MJR, Kumar A (1994) Characterization of the S-adenosylmethionine decarboxylase (SAMDC) gene of potato. Plant Mol Biol26: 327–338
Marco F, Carrasco P (2002) Expression of the pea S-adenosylmethionine decarboxylase gene is involved in developmental and environmental responses. Planta214: 641–647
Martin-Tanguy J (2001) Metabolism and function of polyamines in plants: Recent development (new approaches). Plant Growth Reg34: 135–148
Menges M, Murray JAH (2002) SynchronousArabidopsis suspension cultures for analysis of cell-cycle gene activity. Plant J30: 203–212
Nagata T, Kumagai F (1999) Plant cell biology through the window of the highly synchronized tobacco BY-2 cell line. Meth Cell Sci21: 123–127
Nagata T, Nemoto Y, Hasezawa S (1992) Tobacco BY-2 cell line as the “Hela” cell in the cell biology of higher plants. Intl Rev Cytol123: 1–30
Oredsson SM (2003) Polyamine dependence of normal cell-cycle progression. Biochem Soc Trans31: 366–370
Park KY, Lee SH (1994) Effects of ethylene and auxin on polyamine contents in suspension-cultured cells. Physiol Plant90: 382–390
Park WK, Lee SH, Park KY (2001) Characterization and light-induced expression of the S-adenosylmethionine decarboxylase gene fromIpomoea nil. J Plant Biol44: 111–117
Pohjanpelto P, Nordling S, Knnutila S (1994) Flow cytometric analysis of the cell cycle in polyamine-depleted cells. Cytometry16: 331–338
Porceddu A, Stals H, Reichheld JP, Segers G, De Veylder L, Barroco RP, Casteels P, van Montagu M, Inze D, Mironov V (2001) A plant-specific cyclin-dependent kinase is involved in the control of C2/M progression in plants. J Biol Chem276: 36354–36360
Raney A, Lynn LG, Mize GJ, Morris DR (2002) Regulated translation termination at the upstream open reading frame in S-adenysylmethionine decarboxylase mRNA. J Biol Chem277: 5988–5994
Reichheld JP, Vernoux T, Lardon F, Van Montagu M, Inzé D (1999) Specific checkpoints regulate plant cell cycle progression in response to oxidative stress. Plant J17: 647–656
Setiady YY, Sekine M, Hariguchi N, Kouchi H, Shinmyo A (1996) Molecular cloning and characterization of a cDNA clone that encodes a cdc2 homolog fromNicotiana tabacum. Plant Cell Physiol37: 369–376
Sorrell DA, Menges M, Healy JMS, Deveaux Y, Amano C, Su Y, Nakagami H, Shinmyo A, Doonan JH, Sekine M, Murray JAH (2001) Cell cycle regulation of cyclindependent kinases in tobacco cultivar bright yellow-2 cells. Plant Physiol126: 1214–1223
Swiate A, Lenjou M, Van Bockstaele D, Inzé D, Van Onckelen H (2002) Differential effect of jasmonic acid and abscisic acid on cell cycle progression in tobacco BY-2 cells. Plant Physiol128: 210–211
van Dam L, Korolev N, Nordenskiöld L (2002) Polyamine: Nucleic acid interactions and the effects on structure in oriented DNA fibers. Nucl Acids Res30: 419–428
Waiden R, Cordeiro A, Tiburicio AF (1997) Polyamines: Small molecules triggering pathways in plant growth and development. Plant Physiol113: 1009–1013
Wi SJ, Park KY (2002) Antisense expression of carnation cDNA encoding ACC synthase or ACC oxidase enhances polyamine content and abiotic stress tolerance in transgenic tobacco plants. Mol Cells13: 209–220
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Jang, S.J., Cho, H.W., Park, K.Y. et al. Changes in cellular polyamine contents and activities of their biosynthetic enzymes at each phase of the cell cycle in by-2 cells. J. Plant Biol. 49, 153–159 (2006). https://doi.org/10.1007/BF03031011
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03031011