Abstract
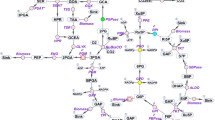
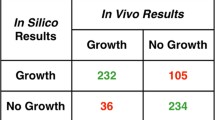
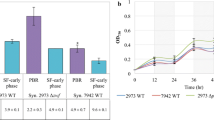
Cyanobacteria produce industrially important secondary metabolites such as lipopeptide, oligosaccharide, fatty acid (esp. sulfolipid),etc. Among them,Synechocystis PCC6803 is the first strain with a publicly available full genome sequence, as of 1996, and is one of the most extensively studied photosynthetic microorganisms. Using this genomic information, the central metabolism ofSynechocystis PCC6803 was reconstructed, including photosynthesis, oxidative phosphorylation, glycolysis, pyruvate metabolism, TCA cycle, carbon fixation, and transport system. Each biochemical reaction was carefully incorporated into the model, taking into consideration the metabolite formula, stoichiometry, charge balance, and thermodynamic properties using information from genomic and metabolic databases as well as biochemical literature. The metabolic flux of the model was calculated using flux balance analysis according to its cultivation with various carbon sources. The results of simulation were in accordance with experimental data, which suggests that the central metabolism model can properly estimate the behavior ofSynechocystis PCC6803. This model would aid in the understanding of the whole cell metabolism ofSynechocystis PCC6803, the first effort of its kind for photosynthetic bacteria.
Similar content being viewed by others
References
Sze, P. (2003)A Biology of the Algae, 3rd ed, pp. 21–38. McGraw-Hill Companies, Boston, MA, USA
Burja, A. M., B. Banaigs, E. Abou-Mansour, J. Grant Burgess, and P. C. Wright (2001) Marine cyanobacteria-a prolific source of natural products.Tetrahedron 57: 9347–9377.
Burja, A. M., S. Dhamwichukorn, and P. C. Wright (2003) Cyanobacterial postgenomic research and systems biology.Trends Biotechnol. 21: 504–511.
Choi, S.-L., I. S. Suh, and C.-G. Lee (2003) Lumostatic operation of bubble column photobioreactors forHaematococcus pluvialis cultures using a specific light uptake rate as a control parameter.Enzyme Microb. Technol. 33: 403–409.
Fazeli M. R., H. Tofighi, N. Samadi, and H. Jamalifar (2006) Effects of salinity on β-carotene production byDunaliella tertiolecta DCCBC26 isolated from the Urmia salt lake, north of Iran.Bioresour. Technol. 97: 2453–2456.
Kim, Z. H., S.-H. Kim, H.-S. Lee, and C.-G. Lee (2006) Enhanced production of astaxanthin by flashing light usingHaematococcus pluvialis.Enzyme Microb. Technol. 39: 414–419.
Lee, H.-S., M.-W. Seo, Z. H. Kim, and C.-G. Lee (2006) Determining the best specific light uptake rates for the lumostatic cultures in bubble column photobioreactors.Enzyme Microb. Technol. 39: 447–452.
Suh, I. S., H.-N. Joo, and C.-G. Lee (2006) A novel double-layered photobioreactor for simultaneousHaematococcus pluvialis cell growth and astaxanthin accumulation.J. Biotechnol. 125: 540–546.
Misawa, N., and H. Shimada (1998) Metabolic engincering for the production of carotenoids in non-carotenogenic bacteria and yeasts.J. Biotechnol. 59: 169–181.
Duarte, N. C., M. J. Herrgard, and B. O. Palsson (2004) Reconstruction and validation ofSaccharomyces cerevisiae iND750, a fully compartmentalized genome-scale metabolic model.Genome Res. 14: 1298–1309.
Raghunathan, A., N. D. Price, M. Y. Galperin, K. S. Makarova, S. Purvine, A. F. Picone, T. Cherny, T. Xie, T. J. Reilly, R. Munson, Jr., R. E. Tyler, B. J. Akerley, A. L. Smith, B. O. Palsson, and E. Kolker (2004)In silico metabolic model and protein expression ofHaemophilus influenzae strain Rd KW20 in rich medium.Omics 8: 25–41.
Reed, J. L., T. D. Vo, C. H. Schilling, and B. O. Palsson (2003) An expanded genome-scale model ofEscherichia coli K-12 (iJR904 GSM/GPR).Genome Biol. 4: R54.
Thiele, I., T. D. Vo, N. D. Price, and B. O. Palsson (2005) Expanded metabolic reconstruction ofHelicobacter pylori (iIT341 GSM/GPR): anin silico genomescale characterization of single- and double-deletion mutants.J. Bacteriol. 187: 5818–5830.
Lee, S. Y., H. M. Woo, D.-Y. Lee, H. S. Choi, T. Y. Kim, and H. Yun (2005) Systems-level analysis of genome-scalein silico metabolic models using MetaFlux-Net.Biotechnol. Bioprocess Eng. 10: 425–431.
Kim, J.-D., and C.-G. Lee (2005) Systemic optimization of microalgae for bioactive compound production.Biotechnol. Bioprocess Eng. 10: 418–424.
Jin, J. H., and J. Lee (2005)In silico analysis of lactic acid secretion metabolism through the top-down approach: Effect of grouping in enzyme kinetics.Biotechnol. Bioprocess Eng. 10: 462–469.
Shastri, A. A., and J. A. Morgan (2005) Flux balance analysis of photoautotrophic metabolism.Biotechnol. Prog. 21: 1617–1626.
Varma, A. and B. O. Palsson (1993) Metabolic capabilities ofEscherichia coli I. Synthesis of biosynthetic precursors and cofactors.J. Theor. Biol. 165: 477–502.
Varma, A. and B. O. Palsson (1993) Metabolic capabilities ofEscherichia coli. II. Optimal growth patterns.J. Theor. Biol. 165: 503–522.
Allen, J. (2002) Photosynthesis of ATP-electrons, proton pumps, rotors, and poise.Cell 110: 273–276.
Gregory, R. P. F. (1989)Biochemistry of Photosynthesis. John Wiley & Sons, Chichester, NY, USA.
Hervas, M., J. A. Navarro, and M. A. De La Rosa (2003) Electron transfer between membrane complexes and soluble proteins in photosynthesis.Acc. Chem. Res. 36: 798–805.
Seelert, H., A. Poetsch, N. A. Dencher, A. Engel, H. Stahlberg, and D. J. Muller (2000) Structural biology, Proton-powered turbine of a plant motor.Nature 405: 418–419.
Kurisu, G., H. Zhang, J. L. Smith, and W. A. Cramer (2003) Structure of the cytochrome b6f complex of oxygenic photosynthesis: Tuning the cavity.Science 302: 1009–1014.
Nelson, N. and C. F. Yocum (2006) Structure and function of photosystems I and II.Annu. Rev. Plant Biol. 57: 521–565.
Munekage, Y., M. Hojo, J. Meurer, T. Endo, M. Tasaka, and T. Shikanai (2002) PGR5 is involved in cyclic electron flow around photosystem I and is essential for photoprotection inArabidopsis.Cell 110: 361–371.
Knowles, V. L. and W. C. Plaxton (2003) From genome to enzyme: analysis of key glycolytic and oxidative pentose-phosphate pathway enzymes in the cyanobacteriumSynechocystis sp. PCC 6803.Plant Cell Physiol. 44: 758–763.
Joset, F., T. Buchou, C.-C. Zhang, and R. Jeanjean (1988) Physiological and genetic analysis of the glucose-fructose permeation system in twoSynechocystis species.Arch. Microbiol. 149: 417–421.
Stal, L. J. and R. Moezelaar (1997) Fermentation in cyanobacteria.FEMS Microbiol. Rev. 21: 179–211.
Tamoi, M., T. Ishikawa, T. Takeda, and S. Shigeoka (1996) Molecular characterization and resistance to hydrogen peroxide of two fructose-1.6-bisphosphatases fromSynechococcus PCC 7942.Arch. Biochem. Biophys. 334: 27–36.
Tamoi, M., A. Murakami, T. Takeda, and S. Shigeoka (1998) Acquisition of a new type of fructose-1,6-bisphosphatase with resistance to hydrogen peroxide in cyanobacteria: molecular characterization of the enzyme fromSynechocystis PCC 6803.Biochim. Biophys. Acta 1383: 232–244.
Scherer, S., E. Sturzl, and P. Boger (1984) Oxygen-dependent proton efflux in cyanobacteria (blue-green algae).J. Bacteriol. 158: 609–614.
Yang, C., Q. Hua, and K. Shimizu (2002) Metabolic flux analysis inSynechocystis using isotope distribution from 13C-labeled glucose.Metab. Eng. 4: 202–216.
Anderson, S. L. and L. McIntosh (1991) Light-activated heterotrophic growth of the cyanobacteriumSynechocystis sp. strain PCC 6803: a blue-light-requiring process.J. Bacteriol. 173: 2761–2767.
Koksharova, O., M. Schubert, S. Shestakov, and R. Cerff (1998) Genetic and biochemical evidence for distinct key functions of two highly divergent GAPDH genes in catabolic and anabolic carbon flow of the cyanobacteriumSynechocystis sp. PCC 6803.Plant Mol Biol. 36: 183–194.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hong, SJ., Lee, CG. Evaluation of central metabolism based on a genomic database ofSynechocystis PCC6803. Biotechnol. Bioprocess Eng. 12, 165–173 (2007). https://doi.org/10.1007/BF03028644
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03028644