Abstract
Purpose
Clinical trials suggest that opioids relieve neuropathic pain and decrease pain-related disability. We conducted a pilot study of current prescribing trends and patients’ attitudes towards opioids for neuropathic pain.
Methods
A patient questionnaire was completed by individuals pursuing enrollment in neuropathic pain clinical trials at our facility.
Results
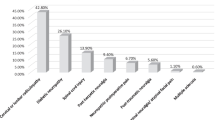
Of 154 patients with diabetic neuropathy (55.2%), postherpetic neuralgia (29.9%), idiopathic peripheral neuropathy (9.7%) and other neuropathies (5.2%), 73.4% complained of inadequate pain control, the mean pain duration was 4.7 (SD = 4.4) yr and the mean pain intensity (0–10) was 7.7 (SD = 2.3). In this group, 40.9% had never tried opioids and 24.7% had never tried any opioids, tricyclic antidepressants or anticonvulsants. Only 9.7% were receiving long-acting opioids or “around the clock” dosing whereas 25.3% were receiving opioids on an “as needed” basis. Opioids combined with tricyclic antidepressants and/or anticonvulsants were used in 11.0%. Fear of addiction and adverse effects were expressed by 31.8% and 46.8% respectively.
Conclusion
These data suggest that barriers to opioid therapy for neuropathic pain include patients’, and possibly physicians’, fears of addiction and adverse effects, which are exaggerated in light of current evidence. The merits of continuous treatment with sustained-release opioids, “as needed” dosing with short-acting preparations, or combining opioids with other agents are discussed. Continued research and communication between health professionals, law enforcement officials and legislators is vital in order to facilitate appropriate opioid use which has a minimal negative impact on the public yet optimally benefits individuals who suffer from disabling neuropathic pain.
Résumé
Objectif
Les essais cliniques montrent que les opioïdes soulagent les douleurs neuropathiques et diminuent l’incapacité reliée à la douleur. Notre étude pilote porte sur les modes courants de prescriptions et les attitudes des patients face aux opioïdes contre la douleur neuropathique.
Méthode
Un questionnaire adressé aux patients a été rempli par des malades intéressés à participer à des essais cliniques sur la douleur neuropathique à notre établissement.
Résultats
Des 154 patients présentant une neuropathie diabétique (55,2 %), une névralgie postherpétique (29,9 %), une neuropathie périphérique idiopathique (9,7 %) ou d’autres neuropathies (5,2 %), 73,4 % se plaignaient d’un soulagement incomplet de la douleur. La durée moyenne des douleurs chroniques était 4,7 ans (écart type = 4,4) et l’intensité moyenne, de 7,7 (écart type = 2,3) sur une échelle de 0–10. Parmi eux, 40,9 % n’avaient jamais pris d’opioïdes et 24,7 % n’avaient jamais pris aucune forme d’opioïdes, d’antidépresseurs ou d’anticonvulsivants tricycliques. Seulement 9,7% avaient des opioïdes à action prolongée ou des dosages continus et 25,3 % prenaient des opioïdes au besoin. Une combinaison d’opioïdes et d’antidépresseurs ou d’anticonvulsivants tricycliques était utilisée par 11,0 % des malades. La crainte d’une accoutumance et des effets indésirables a été exprimée par 31,8 % et 46,8 % respectivement.
Conclusion
Les obstacles au traitement de la douleur neuropathique par des opioïdes comprennent des craintes des patients, et probablement des médecins, d’une accoutumance ou des effets indésirables, ce qui semble exagéré à la lumière des données actuelles. Les mérites du traitement continu avec des opioïdes à libération lente, des dosages “au besoin” avec des préparations à action brève ou des combinaisons d’opioïdes et d’autres médicaments sont discutés. La recherche et les échanges continus entre les professionnels de la santé, les responsables de l’application de la loi et les législateurs sont d’une importance vitale pour faciliter l’usage approprié des opioïdes avec le minimum d’impact négatif sur le public, mais des avantages optimaux pour les malades qui souffrent de douleurs neuropathiques invalidantes.
Article PDF
Similar content being viewed by others
References
Bonica JJ. General considerations of chronic pain.In: JJ Bonica (Ed.), The Management of Pain, 2nd ed. London: Lea & Febiger, 1990.
Verhaak PF, Kerssens JJ, Dekker J, Sorbi MJ, Bensing JM. Prevalence of chronic benign pain disorder among adults: a review of the literature. Pain 1998; 77: 231–9.
Flor H, Fydrich T, Turk DC. Efficacy of multidisciplinary pain treatment centers: a meta-analytic review. Pain 1992; 49: 221–30.
Reisine T, Pasternak G. Opioid analgesics and antagonists.In: Hardman JG, Limbird LE (Eds.). Goodman & Gilman’s The Pharmacological Basis of Therapeutics, 9th ed. New York: McGraw-Hill, 1996: 521–55.
Portenoy RK. Opioid therapy for chronic nonmalignant pain: a review of the critical issues. J Pain Symptom Manage 1996; 11:203–17.
Moulin DE, Iezzi A, Amireh R, Sharpe WK, Boyd D, Merskey H. Randomised trial of oral morphine for chronic non-cancer pain. Lancet 1996; 347: 143–7.
Bowsher D. Neurogenic pain syndromes and their management. Br Med Bull 1991; 47: 644–66.
Stankus SJ, Dlugopolski M, Packer D. Management of herpes zoster (shingles) and postherpetic neuralgia. Am Fam Physician 2000; 61: 2437–48.
Sindrup SH, Jensen TS. Efficacy of pharmacological treatments of neuropathic pain: an update and effect related to mechanism of drug action. Pain 1999; 83: 389–400.
Neil A, Kayser V, Chen YL, Guilbaud G. Repeated low doses of morphine do not induce tolerance but increase the opioid antinociceptive effect in rats with a peripheral neuropathy. Brain Res 1990; 522: 140–3.
Bian D, Nichols ML, Ossipov MH, Lai J, Porreca F. Characterization of the antiallodynic efficacy of morphine in a model of neuropathic pain in rats. Neuroreport 1995; 6: 1981–4.
Suzuki R, Chapman V, Dickenson AH. The effectiveness of spinal and systemic morphine on rat dorsal horn neuronal responses in the spinal nerve ligation model of neuropathic pain. Pain 1999; 80: 215–28.
Dellemijn PL, Vanneste JA. Randomised double-blind active-placebo-controlled crossover trial of intravenous fentanyl in neuropathic pain. Lancet 1997; 349: 753–8.
Sindrup SH, Andersen G, Madsen C, Smith T, Brosen K, Jensen TS. Tramadol relieves pain and allodynia in poly neuropathy: a randomised, double-blind, controlled trial. Pain 1999; 83: 85–90.
Watson CPN, Moulin D, Gordon A, et al. A randomized, double-blind, crossover comparison of the efficacy and safety of oral controlled-release oxycodone and active placebo in patients with painful diabetic neuropathy. Am Pain Soc 2001; 794 (abstract).
Watson CP, Babul N. Efficacy of oxycodone in neuropathic pain. A randomized trial in postherpetic neuralgia. Neurology 1998; 50: 1837–41.
Cleeland CS, Gonin R, Hatfield AK, et al. Pain and its treatment in outpatients with metastatic cancer. N Engl J Med 1994; 330: 592–6.
Ward SE, Goldberg N, Miller-McCauley V, et al. Patient-related barriers to management of cancer pain. Pain 1993; 52: 319–24.
Lewis LM, Lasater LC, Brooks CB. Are emergency physicians too stingy with analgesics? South Med J 1994; 87: 7–9.
American Pain Society Quality of Care Committee. Quality improvement guidelines for the treatment of acute pain and cancer pain. JAMA 1995; 274: 1874–80.
Potter M, Schafer S, Gonzalez-Mendez E, et al. Opioids for chronic nonmalignant pain. Attitudes and practices of primary care physicians in the UCSF/Stanford Collaborative Research Network. J Fam Pract 2001; 50: 145–51.
Schofferman J. Long-term use of opioid analgesics for the treatment of chronic pain of nonmalignant origin. J Pain Symptom Manage 1993; 8: 279–88.
Von Roenn JH, Cleeland CS, Gonin R, Hatfield AK, Pandya KJ. Physician attitudes and practice in cancer pain management. A survey from the Eastern Cooperative Oncology Group. Ann Intern Med 1993; 119: 121–6.
Boulton AJ, Armstrong WD, Scarpello JH, Ward JD. The natural history of painful diabetic neuropathy—a 4-year study. Postgrad Med J 1983; 59: 556–9.
Davies HT, Crombie IK, Macrae WA. Waiting in pain. Delays between referral and consultation in outpatient pain clinics. Anaesthesia 1994; 49: 661–5.
Glajchen M. Chronic pain: treatment barriers and strategies for clinical practice. J Am Board Fam Pract 2001; 14: 211–8.
Petrone D, Kamin M, Olson W. Slowing the titration rate of tramadol HCl reduces the incidence of discontinuation due to nausea and/or vomiting: a double-blind randomized trial. J Clin Pharm Ther 1999; 24: 115–23.
Brownstein MJ. A brief history of opiates, opioid peptides, and opioid receptors. Proc Natl Acad Sci USA 1993; 90: 5391–3.
Porter J, Jick H. Addiction rare in patients treated with narcotics (Letter). N Engl J Med 1980; 302: 123.
Perry S, Heidrich G. Management of pain during debridement: a survey of U.S. burn units. Pain 1982; 13: 267–80.
Savage SR. Long-term opioid therapy: assessment of consequences and risks. J Pain Symptom Manage 1996; 11:274–86.
Passik SD. Responding rationally to recent reports of abuse/diversion of Oxycontin® (Letter). J Pain Symptom Manage 2001; 21: 359.
Max MB. Combining opioids with other drugs: challenges in clinical trial design.In: Gebhart GF, Hammond DL, Jensen TS (Eds.). Progress in Pain Research and Management, vol. 2. Seattle: IASP Press, 1994.
Fields HL. Peripheral neuropathic pain: an approach to management.In: Wall PD, Melzack R (Eds.), Textbook of Pain, 3rd ed. Edinburgh: Churchill Livingstone, 1994.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by CIHR Grant #MCT-38149 and Queen’s University Grant #383-861.
Rights and permissions
About this article
Cite this article
Gilron, I., Bailey, J.M. Trends in opioid use for chronic neuropathic pain: a survey of patients pursuing enrollment in clinical trials. Can J Anesth 50, 42–47 (2003). https://doi.org/10.1007/BF03020185
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03020185