Abstract
Purpose: To evaluate the effects of low-dose olprinone, a phosphodiesterase Ill inhibitor, on contractility and its mechanism in nonfatigued and fatigued diaphragm in dogs.
Methods: Thirty six pentobarbitone-anesthetized dogs were studied. In Group la (n=6), animals without fatigue, received no study drug. In Group lb (n=6), dogs were given a bolus injection (10 μg·kg−1) followed by continuous infusion (0.1 μg·kg−1·min−1) of olprinone. In Groups lla, llb, and llc (n=8 each), diaphragmatic fatigue was induced by intermittent supramaximal bilateral electrophrenic stimulation at a frequency of 20-Hz applied for 30 min. After producing fatigue, Group lla received no study drug; Group llb was infused with olprinone (10 μg·kg−1 loading dose plus 0.1 μg·kg−1·min−1 maintenance dose); Group llc was infused with nicardipine (5 μg·kg−1·min−1) during olprinone administration. Diaphragmatic contractility was assessed by transdiaphragmatic pressure (Pdi).
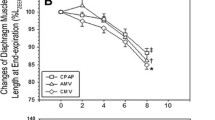
Results: No difference in Pdi was observed between Groups la and lb. After fatigue, in Groups lla, llb, and llc, Pdi at low-frequency (20-Hz) stimulation decreased from prefatigued (baseline) values (P<0.05), whereas there was no change in Pdi at high-frequency stimulation (100-Hz). In Group llb, during olprinone administration, Pdi at both stimuli increased from fatigued values (P<0.05). In Group llc, the augmentation of Pdi to each stimulus in fatigued diaphragm by olprinone was abolished with an infusion of nicardipine.
Conclusion: Low-dose olprinone does not affect contractility in nonfatigued diaphragm, but increases contractility in fatigued diaphragm via its effect on transmembrane calcium movement in dogs.
Résumé
Objectif: Évaluer les effets d’une faible dose d’olprinone, un inhibiteur de la phosphodiestérase Ill, sur la contractilité et sur son mécanisme sur le diaphragme fatigué ou non, chez des chiens.
Méthode: Trente-six chiens anesthésiés au pentobarbital ont été étudiés. Dans le groupe la (n=6), les animaux non fatigués n’ont pas reçu de médicament à l’étude. Dans le groupe lb (n=6), les chiens ont reçu l’injection d’un bolus (10 μg·kg−1) d’olprinone, suivie d’une perfusion continue (0,1 μg·kg−1·min−1). Dans les groupes lla, llb, et llc (n=8 chacun), on a induit la fatigue diaphragmatique par une stimulation électrophrénique intermittente supramaximale bilatérale à une fréquence de 20 Hz appliquée pendant 30 min. Après l’induction de cette fatigue, les chiens du groupe lla n’ont pas reçu de médicament; ceux du groupe llb ont reçu une perfusion d’olprinone (10 μg·kg−1 en dose de charge plus 0,1μg·kg−·min−1 comme dose de maintien); ceux du groupe llc ont reçu de la nicardipine (5 μg·kg−1·min−1) pendant l’administration d’olprinone. La contractilité diaphragmatique a été évaluée par pression transdiaphragmatique (Pdi).
Résultats: Aucune différence de Pdi n’a été observée entre les groupes la et lb. Après la production de fatigue chez les animaux des groupes lla, llb, et llc, la Pdi sous stimulation à basses fréquences (20 Hz) a diminué par rapport aux valeurs précédant la fatigue (valeurs de base),P<0,05, tandis qu’il n’y a pas eu de changement de Pdi sous stimulation à hautes fréquences (100 Hz). Dans le groupe llb, pendant l’administration d’olprinone, la Pdi a augmenté par rapport aux valeurs du muscle fatigué et ce, sous stimulation à toutes les fréquences utilisées (P<0,05). Dans le groupe llc, l’augmentation de Pdi par l’olprinone pour chaque stimulus du diaphragme fatigué a été abolie avec une perfusion de nicardipine.
Conclusion: Une faible dose d’olprinone n’a pas d’effet sur la contractilité d’un diaphragme non fatigué, mais accroît celle d’un muscle fatigué par ses effets sur le déplacement du calcium transmembranaire chez les chiens.
Similar content being viewed by others
References
LeJemtel TH, Keung E, Sonnenblick EH, et al. Amrinone: a new non-glycoside, non-adrenergic cardiotonic agent effective in the treatment of intractable myocardial failure in man. Circulation 1979; 59: 1098–104.
Karlsberg RP, DeWood MA, DeMaria AN, Berk MR, Lasher KP. Comparative efficacy of short-term intravenous infusions of milrinone and dobutamine in acute congestive herat failure following acute myocardial infarction. Clin Cardiol 1996; 19: 21–30.
Takaoka H, Takeuchi M, Odake M, et al. Comparison of the effects on arterial-venticular coupling between phospodiesterase inhibitor and dobutamine in the diseased human heart. J Am Coll Cardiol 1993; 22: 598–606.
Fujii Y, Toyooka H, Amaha K. Amrinone improves contractility of fatigued diaphragm in dogs. Can J Anaesth 1995; 42: 80–6.
Fujii Y, Takahashi S, Toyooka H. The effects of milrinone and its mechanism in the fatigued diaphragm in dogs. Anesth Analg 1998; 87: 1077–82.
Fujii Y, Takahashi S, Toyooka H. The effect of olprinone compared with milrinone on diaphragmatic muscle function in dogs. Anesth Analg 1999; 89: 781–5.
Kimata S, Hirosawa K, Kasanuki H, et al. Clinical effects of olprinone on acute heart failure. Optimal dose-finding study. Jpn J Clin Exp Med 1992; 69: 3984–96.
Aubier M, Farkas G, DeTroyer A, Mozes R, Roussos C Detection of diaphragmatic fatigue in man by phrenic nerve stimulation. J Appl Physiol 1981; 50: 538–44.
Grassino A, Goldman MD, Mead J, Sears TA. Mechanics of the human diaphragm during voluntary contraction: statics. J Appl Physiol 1978; 44: 829–39.
Esau SA. Hypoxic, hypercapnic acidosis decreases tension and increases fatigue in hamster diaphragmatic muscle in vitro. Am Rev Respir Dis 1989; 139: 1410–7.
Howell S, Fitzgerald RS, Roussos C Effects of uncompensated and compensated metabolic acidosis on canine diaphragm. J Appl Physiol 1985; 59: 1376–82.
Ide T, Kochi T, Isono S, Mizuguchi T. Effect of sevoflurane on diaphragmatic contractility in dogs. Anesth Analg 1992; 74: 739–46.
Roussos C, Macklem PT. The respiratory muscles. N Engl J Med 1982; 307: 786–97.
Satoh H, Endoh M. Effects of a new cardiotonic agent 1,2-dihydro-6-methyl-2-oxo-5-[imidazol(1,2-a)pyridine-6-yl]-3-pyridine carbonitrile hydrochloride monohydrate (E-1020) on contractile force and cyclic AMP metabolism in canine ventricular muscle. Jpn J Pharmacol 1990; 52: 215–24.
Robertson CH Jr,Foster GH, Johnson RL Jr. The relationship of respiratory failure to the oxygen consumption of, lactate production by, and distribution of blood flow among respiratory muscles during increasing inspiratory resistance. J Clin Invest 1977; 59: 31–42.
Author information
Authors and Affiliations
Corresponding author
Additional information
A retraction note to this article is available at http://dx.doi.org/10.1007/s12630-013-9945-3.
This article has been retracted due to two reasons: Many variables reported in the studies are exceedingly unlikely. The author's institution is not able to attest to the integrity of the study and/or the data conducted under its auspices.
An erratum to this article is available at http://dx.doi.org/10.1007/s12630-013-9945-3.
About this article
Cite this article
Fujii, Y., Toyooka, H. RETRACTED ARTICLE: Different effects of olprinone on contractility in nonfatigued and fatigued diaphragm in dogs. Can J Anesth/J Can Anesth 47, 1243–1248 (2000). https://doi.org/10.1007/BF03019875
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03019875