Abstract
Purpose
To investigate whether the timing of intrathecal administration of the opioid analgesic fentanyl, alters noxious stimulusevoked neuronal activity in the rat spinal cord.
Methods
A 5% formalin solution was used as the noxious stimulant. For the pretreatment group, adose of 0.00l to 0.5 μg of fentanyl was injected intrathecally ten minutes prior to formalin injection. Early and late post-treatment groups received 0.01 to 0.5 μg fentanyl, five and 60 min after formalin injection respectively. The effect of fentanyl was confirmed with naloxone. The level of c-Fos expression was determined in each treatment group to indicate neuronal activity.
Results
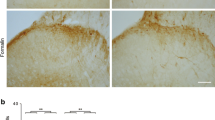
Pretreatment and early post-treatment groups showed suppression of c-Fos activity compared to the vehicle (P < 0.0l). The late post-treatment group showed no difference in c-Fos activity compared to the vehicle (P = NS). Pretreatment with fentanyl showed the most profound suppression of c-Fos expression (P < 0.0l). In addition, pretreatment injection showed a greater suppression of c-Fos activity in the deep (14.6% of control) compared to the superficial laminae (32.7% of control;P < 0.01), whereas the early post-treatment group showed a universal decrease in c-Fos activity (49.2% of control in laminae I and II, 50.4% of control in laminae III and IV and 51.8% of control in laminae V and VI). Naloxone reversed the action of fentanyl on c-Fos activity.
Conclusion
Inasmuch as: 1) c-Fos expression can be equated with behavioural changes; 2) injection of formalin is an appropriate model of surgical trauma; and 3) animal data can be transports to humans, these results suggest that fentanyl would be an effective pre-emptive analgesic.
Résumé
Objectif
Vérifier si le moment choisi pour l’administration intrathécale de l’analgésique opioïde fentanyl agit sur l’activité nerveuse induite par un stimulus nocif dans la moelle épinière du rat.
Méthode
Une solution de formol à 5 % a servi de stimulus nocif Dans le groupe de prétraitement, une dose de 0,001 à 0,5 μg de fentanyl a été administrée en injection intrathécale dix minutes avant l’injection de formol. Dans les groupes de post-traitement hâtif et tardif, on a donné de 0,01 à 0,5 μg de fentanyl, cinq et 60 min après le formol, respectivement, Leffet du fentanyl a été confirmé par la naloxone. Le niveau déxpression de c-Fos a été déterminé dans chaque groupe pour indiquer l’activité neuronale.
Résultats
Léxamen des rats des groupes de prétraitement et de post-traitement hâtif comparés au groupe ayant reçu l’excipient, a montré une suppression de l’activité de c-Fos (P < 0,01). Le groupe de post-traitement tardif, comparé à l’excipient, ne présentait aucune différence d’activité de c-Fos (P = NS). Le prétraitement au fentanyl a permis la suppression la plus profonde de l’expression de c-Fos (P < 0,01). De plus, l’injection de prétraitement a entraîné une suppression plus importante dans les lames profondes (14,6 % des témoins) comparées aux lames superficielles (32,7 % des témoins; P < 0,01), tandis que le post-traitement hâtif a provoqué une baisse générale de l’activité de c-Fos (49,2 % des témoins dans les lames I et II, 50,4 % des témoins dans les lames III et IV et 51,8 % des témoins dans les lames V et VI). La naloxone a renversé l’action du fentanyl sur l’activité de c-Fos.
Conclusion
Étant donné: 1) que l’expression de c-Fos peut être mise en équation avec les changements comportementaux; 2) que l’injection de formol constitue un modèle approprié de traumatisme chirurgical et 3) que les données animales peuvent être extrapolées à l’humain, ces résultats suggèrent que le fentanyl serait un analgésique préventif efficace.
Article PDF
Similar content being viewed by others
References
Coderre TJ, Vaccarino AL, Melzack R. Central nervous system plasticity in the tonic pain response to subcutaneous formalin injection. Brain Res 1990; 535: 155–8.
Honoré P, Chapman V, Buritova J, Besson J-M. Concomitant administration of morphine and an N-methyl-D-aspartate receptor antagonist profoundly reduces inflammatory evoked spinal c-Fos expression. Anesthesiology 1996; 85: 150–60.
Woolf CJ, Chong M-S. Preemptive analgesia — treating postoperative pain by preventing the establishment of central sensitization. Anesth Analg 1993; 77: 362–79.
Katz J, Kavanagh BP, Sandiar AN, et al. Preemptive analgesia. Clinical evidence of neuroplasticity contributing to postoperative pain. Anesthesiology 1992; 77: 439–46.
Tverskoy M, Oz Y, Isakson A, Finger J, Bradley EL Jr, Kissin I. Preemptive effect of fentanyl and ketamine on postoperative pain and wound hyperalgesia. Anesth Analg 1994; 78: 205–9.
Yaksh TL, Rudy TA. Chronic catheterization of the spinal subarachnoid space. Physiol Behav 1976; 17: 1031–6.
Presley RW, Menétrey D, Levine JD, Basbaum AI. Systemic morphine suppresses noxious stimulus-evoked Fos protein-like immunoreactivity in the rat spinal cord. J Neurosci 1990; 10: 323–35.
Molander C, Xu Q, Grant G. The cytoarchitechtonic organization of the spinal cord in the rat. I. The lower thoracic and lumbosacral cord. J Comp Neurol 1984; 230: 133–41.
Swett JE, Woolf CJ. The somatotopic organization of primary afferent terminals in the superficial laminae of the dorsal horn of the rat spinal cord. J Comp Neurol 1985; 231: 66–77.
Besson JM, Chaouch A. Peripheral and spinal mechanisms of nociception. Physiol Rev 1987; 67: 67–186.
Atweh SF, Kuhar MJ. Autoradiographic localization of opiate receptors in rat brain. I. Spinal cord and lower medulla. Brain Res 1977; 124: 53–67.
Abram SE, Yaksh TL. Morphine, but not inhalation anesthesia, blocks post-injury facilitation. The role of preemptive suppression of afferent transmission. Anesthesiology 1993; 78: 713–21.
Fields HL, Basbaum AI. Central nervous system mechanisms of pain modulation.In: Wall PD, Melzack R (Eds.). Textbook of Pain, 4th ed., New York: Churchill Livingstone Inc., 1999: 309–29.
Reuben SS, Dunn SM, Duprat KM, O’Sullivan P. An intrathecal fentanyl dose-response study in lower extremity revascularization procedures. Anesthesiology 1994; 81: 1371–5.
Joris JL, Dubner R, Hargreaves KM. Opioid analgesia at peripheral sites: a target for opioids released during stress and inflammation? Anesth Analg 1987; 66: 1277–81.
Hunt SP, Pini A, Evan G. Induction of c-Fos-like protein in spinal cord neurons following sensory stimulation. Nature 1987; 328: 632–4.
Menétrey D, Gannon A, Levine JD, Basbaum AI. Expression of c-fos protein in interneurons and projection neurons of the rat spinal cord in response to somatic, articular, and visceral stimulation. J Comp Neurol 1989; 285: 177–95.
Dubuisson D, Dennis SG. The formalin test: a quantitative study of the analgesic effects of morphine, meperidine, and brain stimulation in rats and cats. Pain 1977; 4: 161–74.
Tashpal K, Mason P, Mckenna JE, Sharma SK, Henry JL, Coderre TJ. Comparison of the effects of treatment with intrathecal lidocaine given before and after formalin on both nociception and Fos expression in the spinal cord dorsal horn. Anesthesiology 1998; 88: 157–64.
Girlon I, Quirion R, Coderre TJ. Preversus postformalin effects of ketamine or large-dose alfentanil in the rat: discordance between pain behavior and spinal Fos-like immunoreactivity. Anesth Analg 1999; 89: 128–35.
Dickenson AH, Sullivan AF. Subcutaneous formalin-induced activity of dorsal horn neurons in the rat: differential response to an intrathecal opiate administered pre or post formalin. Pain 1987; 30: 349–60.
Siddall PJ, Cousins MJ. Introduction to pain mechanisms. Implications for neural blocakde.In: Cousins MJ, Bridenbaugh PO (Eds.). Neural Blockade in Clinical Anesthesia and Management of Pain, 3rd ed., Philadelphia: Lippincott-Raven Inc., 1998: 675–99.
Griffin MJ, Hughes D, Knaggs A, Donnelly MB, Boylan JF. Late-onset preemptive analgesia associated with preincisional large-dose alfentanil. Anesth Analg 1997; 85: 1317–21.
Williams S, Evan GI, Hunt SP. Changing patterns of c-fos induction in spinal neurons following thermal cutaneous stimulation in the rat. Neuroscience 1990; 36: 73–81.
Herdegen T, Kovary K, Leah J, Bravo R. Specific temporal and spatial distribution of JUN, FOS, and KROX-24 proteins in spinal neurons following noxious transsynaptic stimulation. J Comp Neurol 1991; 313: 178–91.
Abbadie C, Taylor BK, Peterson MA, Basbaum AI. Differential contribution of the two phases of the formalin test to the pattern of c-fos expression in the rat spinal cord: studies with remifentanil and lidocaine. Pain 1997; 69: 101–10.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nakamura, T., Takasaki, M. Intrathecal pre-administration of fentanyl effectively suppresses formalin evoked c-Fos expression in spinal cord of rat. Can J Anaesth 48, 993–999 (2001). https://doi.org/10.1007/BF03016590
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03016590