Abstract
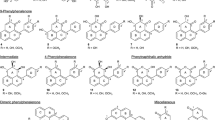
DL-α-Difluoromethylornithine (DFMO), a specific suicide inhibitor of the polyamine biosynthetic enzyme ornithine decarboxylase (EC 4.1.1.17), strongly inhibited mycelial growth and sporidial formation of the wheat pathogen,Neovossia indica, in vitro, while DL-α-difluoromethylarginine (DFMA), the analogous suicide inhibitor of arginine decarboxylase (EC 4.1.1.19), did not. The inhibited mycelial growth and sporidial formation were not only restored by putrescine (polyamine) addition, but were actually enhanced in the putrescine + DFMO cultures. Besides altering mycelial growth and morphology, DFMO also reduced the cell size drastically. The inhibition of fungal polyamine biosynthesis is discussed in relation to selective control of plant disease.
Similar content being viewed by others
References
Bedi, P.S. (1988) Advances in the study of Kamal bunt of wheat — An overview.Pl. Dis. Res. 3:88.
Dhiman, J.S. and Bedi, P.S. (1983) A technique for the isolation ofNeovossia indica — the causal organism of Kamal bunt of wheat.Indian Phytopath. 36:767–768.
Heby, O. (1981) Role of polyamines in the control of cell proliferation and differentiation.Differentiation 19:1–20.
Joshi, L.M., Singh, D.V., Srivastava, K.D. and Wilcoxson, R.D. (1983) Kamal bunt: A minor disease that is now a major threat to wheat.Bot. Rev. 49:309–330.
Mitia, M.A. (1931) A new bunt on wheat in India.Ann. appl. Biol. 18:178–179.
Paulus, T.J. and Davis, R.H. (1981) Regulation of polyamine synthesis in relation to putrescine and spermidine pools inNeurospora crassa.J. Bact. 145:14–20.
Rajam, M.V. and Galston, A.W. (1985) The effects of some polyamine biosynthetic inhibitors on growth and morphology of phytopathogenic fungi.Pl. Cell Physiol. 26:683–692.
Shapira, R., Altman, A. and Henis, Y. (1989) Polyamine and ornithine decarboxylase activity during growth and differentiation inSclerotium rolfsii.J. gen. Microbiol. 135:1361–1367.
Singh, P.P. and Bedi, P.S. (1987) Effect of different physical and cultural factors on the growth and sporidiation ofNeovossia indica.Indian Phytopath. 40:540–542.
Slocum, R.D., Kaur-Sawhney, R. and Galston, A.W. (1984) The physiology and biochemistry of polyamines in plants.Arch. Biochem. Biophys. 235:283–303.
Smilanick, J.L., Hoffmann, J.A., Cashion, N.L. and Prescott, J.M. (1987) Evaluation of seed and foliar fungicides for control of Karnal bunt of wheat.Pl. Dis. 71:94–96.
Smith, T.A. (1985) Polyamines.A. Rev. Pl. Physiol. 36:117–143.
Stevens, L. and Winther, M.D. (1979) Spermine, spermidine and putrescine in fungal development.Adv. Microbiol. Physiol. 19:63–148.
Tabor, G.W. (1981) Mutants ofSaccharomyces cerevisae deficient in polyamine biosynthesis: Studies on the regulation of ornithine decarboxylase.Med. Biol. 59:272–278.
Tabor, G.W. and Tabor, H. (1985) Polyamines in micro-organisms.Microbiol. Rev. 49:81–99.
West, H.M. and Walters, D.R. (1988) The effects of polyamine biosynthesis inhibitors on infection ofHordeum vulgare L. byErysiphe graminis f.sp.hordei Marchal.New Phytol. 110:193–200.
Whitney, P.A. and Morris, D.R. (1978) Polyamine auxotrophs ofSaccharomyces cerevisae.J. Bact. 134:214–220.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Singh, P.P., Singh, S., Basra, A.S. et al. Inhibition of mycelial growth and sporidial formation in the wheat pathogenNeovossia indica by a polyamine biosynthetic inhibitor. Phytoparasitica 17, 323–326 (1989). https://doi.org/10.1007/BF02980762
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02980762