Abstract
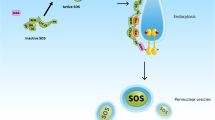
Mutated forms of ras are found in many human tumors and the rate of incidence is significantly higher in colon and pancreatic cancers. The protein product from theras oncogene is a small G-protein, p21ras(Ras) that is known to play a key role in the signal transduction cascade and cell differentiation and proliferation. Mutated Ras is unable to regulate itself and remains constantly activated, leading to uncontrolled cell growth. The function of Ras in signal transduction requires its location near the growth factor receptor at the cell membrane. However, Ras does not have a transmembrane domain. Ras requires farnesylation to increase its hydrophobicity and subsequent plasma membrane association for its transforming activity. This key post-translational modification is catalyzed by the enzyme Ras farnesyltransferase (FTase), which transfers a farnesyl group from famesylpyrophosphate to theC-terminal cysteine of the Ras protein. The requirement has focused attention on FTase as a target for therapeutic intervention. Selective inhibition of FTase will prevent Ras protein from association with the plasma membrane, leading to a disruption of oncogenic Ras function.
Similar content being viewed by others
References
Barbacid, M., Ras gene.Annu. Rev. Biochem., 56, 779–827 (1987).
Barinaga, M., From bench top to bedside.Science, 278, 1036–1039 (1997).
Bolton, G. L., Sebolt-Leopold, J. S. and Hodges, J. C., In Bristol, J. A. (Ed.),Annual reports in Medicinal Chemistry, vol. 29, Academic Press, New York, pp165–174, (1994).
Bos, J. L., Ras Oncogenes in human cancer.Cancer Res., 49, 4682–4689 (1989).
Brown, M. S., Goldstein, J. L., Paris, K. J., Burnier, J. P. and Marsters, J. J., Tetrapeptide inhibitors of protein farnesy-Itransferase: Amino-terminal substitution in phenylalanine-containing tetrapeptides restores farnesylation.Proc. Natl. Acad.Sci. USA, 89, 8313–8316 (1992).
Burn, C. J., Guitton, J.-D., Baudoin, B., Lelievre, Y., Duchesne, M., Parker, F., Fromage, N. and Commercon, A., Novel conformationally extended naphthalene-based inhibitors of farnesyltransferase.J. Med. Chem., 40, 1763–1767 (1997).
Buss, J. E. and Marsters, J. C., Jr., Farnesyl transferase inhibitors: The successes and surprises of new class of potential cancer chemotherapeutics.Chemistry & Biology, 2, 787–791 (1995).
Cadena, D. L. and Gill, G. N., Receptor tyrosine kinases.FASEB J., 6, 2332–2337 (1992).
Casey, P. J., Biochemistry of protein prenylation.J. Lipid Res., 33, 1731–1740 (1992).
Casey, P. J. and Seabra, M. C., Protein prenyltransferases.J. Biol. Chem., 271, 5289–5292 (1996).
Casey, P. J., Solski, P. A., Der, C. J. and Buss, J. E., p21 Ras is modified by a farnesyl isoprenoid.Proc. Natl. Acad. Sci. USA, 86, 8323–8327 (1989).
Clerc, F.-F., Guitton, J.-D., Fromage, N., Lelievre, Y., Duchesne, M., Tocque, B., James-Surcouf, E., Commercon, A. and Becquart, J., Constrained analogues of kcvfmwith improved inhibitory properties against farnesyltransferase.Bioorg. Med.Chem., 5, 1779–1784 (1995).
Cox, A. D. and Der, C. J., Farnesyltransferase inhibitors and cancer treatment: Targeting simply Ras?.Biochim. Biophy. Acta., 1333, F51-F71 (1997).
Der, C. J. and Cox, A. D., Isoprenoid modification and plasma membrane association: critical factors for ras oncogenicity.Cancer Cells, 3, 331–340 (1991).
Dolence, J. M. and Poulter, C. D., A mechanism for posttranslational modifications of proteins by yeast protein farnesyltransferase.Proc. Natl. Acad. Sci. USA, 92, 5008–5011 (1995).
Dunten, P., Kammlott, U., Crowther, R., Weber, D., Palermo, R. and Birktoft, J., Protein farnesyltransferase: Structure and implications for substrate binding.Biochemistry, 37, 7907–7912 (1998).
Edelstein, R. L., Weller, V. A. and Distefano, M. D., Stereo-chemical analysis of the reaction catalyzed by yeast protein farnesyltransferase.J. Org. Chem., 63, 5298–5299 (1998).
Fearon, E. R., Human cancer syndromes.Science, 278, 1043–1050 (1997).
Fu, H.-W., Beese, L. S. and Casey, P. J., Kinetic analysis of zinc ligand mutants of mammalian protein farnesyltransferase.Biochemistry, 37, 4465–4472 (1998).
Gibbs, J. B., Graham, S. L., Hartman, G. D., Koblan, K. S., Kohl, N. E., Omer, C. A. and Oliff, A., Farnesyltransferase inhibitors versus Ras inhibitors.Curr. Opin. Chem. Biol., 1, 197–203 (1997).
Grand, R. J. A. and Owen D., The biochemistry of ras p21.Biochem. J., 279, 609–631 (1991).
Gutierrez, L., Magee, A. I., Marshall, C. J. and Hancock, J. F., Post-translational processing of p21 ras is two-step and involves carboxyl-methylation and carboxyterminal proteolysis.EMBO J., 8, 1093–1098 (1989).
Hamilton, A. D. and Sebti, S. M., Inhibitors of Ras farnesyltransferase as novel antitumor agents.Drug News Perspect., 8, 138–145 (1995).
Hancock, J. F., Magee, A. I., Childs, J. E. and Marshall, C. J., All ras proteins are polyisoprenylated but only some are palmitoylated.Cell, 57, 1167–1177 (1989).
Heldin, C.-H. and Westermark B., Signal transduction by the receptors for plateleWerived growth factor.J. Cell Sci., 96, 193–196 (1990).
Hightower, K. E. and Fierke, C. A., Zinc-catalyzed sulfur alkylation: Insights from protein farnesyltransferase..Curr. Opin. Chem. Biol., 3, 176–181 (1999).
Hightower, K. E., Huang, C.-C., Casey, R J. and Fierke, C. A., H-Ras peptide and protein substrates bind protein farnesyltransferase as an ionized thiolate.Biochemistry, 37, 15555–15562 (1998).
Hinterding, K., Alonso-Diaz, D. and Waldmann, H., Organic synthesis and biological signal transduction.Angew. Chem. Int. Ed. Engl., 37, 688–749 (1998).
Hunt, J. T., Lee, Y. G., Leftheris, K., Seizinger, B., Carboni, J., Mabus, J., Ricca, C., Yan, N. and Manne, V., Potent, cell active, non-thiol tetrapeptide inhibitors offarnesyltransferase.J. Med. Chem., 39, 353–358 (1996).
Kobran, K. S., Culberson, J. C., Desolms, S. J., Giuliani, E. A., Mosser, S. D., Omer, CA., Pitzenberger, S. M. and Bogusky, M. J., NMR studies of novel inhibitors bound to farnesyl protein transferase.Protein Sci., 4, 681–688 (1995).
Krengel, U., Schlichting, I., Scherer, A., Schumann, R., French, M., John, J., Kabsch, W., Pai, E. F. and Wittinghofer, A., Three-dimensional structures of H-ras p21 mutants: Molecular basis for their inability to function as signal switch molecules.Cell, 62, 539–548 (1990).
Leonard, D. M., Ras farnesyltransferase: A new therapeutic target.J. Med. Chem., 40, 2971–2990 (1997).
Leonard, D. M., Shuler, K. R., Poulter, C. J., Eaton, S. R., Sawyer, T. K., Hodges, J. C., Su, T.-Z., Scholten, J. D., Gowan, R. C., Sebolt-Leopold, J. S. and Doherty, A. M., Structure-activity relationships of cysteine-locking pentapeptide derivatives that inhibit ras farnesyltransferase.J. Med. Chem., 40, 192–200 (1997).
Long, S. B. and Casey, P. J., Cocrystal structure of protein farnesyltransferase complexed with a farnesyl diphosphate substrate.Biochemistry, 37, 9612–9618 (1998).
Lowy, D. R. and Willumsen, B. M., Function and regulation of Ras.Annu. Rev. Biochem., 62, 851–891 (1993).
Maignan, S., Guilloteau, J.-P., Fromage, N., Arnoux, B., Becquart, J. and Ducruix, A., Crystal structure of the mammalian Grb2 adaptor.Science, 268, 291–293 (1995).
Masrers, J. C., Jr., McDowell, R. S., Reynilds, M. E., Oare, D. A., Somers, T. C., Stanley, M. S., Rawson, T. E., Struble, M. E., Burdick, D. J., Chan, K. S., Duarte, C. M., Paris, K. J., Tom, J. Y., Wan, D. T., Xue, Y. and Burnier, J. P., Benzodiazepine peptidomimetic inhibitors of farnesylferase.Bioorg. Med. Chem., 2, 949–957 (1994).
Matthews, R. G. and Goulding, C. W., Enzyme-catalyzed methyl transfers to thiols: Therole of zinc.Curr. Opin. Chem. Biol., 1, 332–339 (1997).
McCormick, F., Ras GTPase activating protein: Signal transmitter and signalterminator.Cell, 56, 5–8 (1989).
McCormick, F., How receptors turn Ras on.Nature, 363, 15–16 (1993).
McCormick, F., Raf: The holy grail of Ras biology?.Trends Cell Biol., 1994, 4, 347–350 (1994).
Moodie, S. A. and Wolfman, A., The 3Rs of life: Ras, Raf and Growth regulation.TIG, 10, 44–48 (1994).
Mu, Y., Omer, C. A. and Gibbs, R. A., On the stereochemical course of human protein farnesyltransferaes.J. Am. Chem. Soc., 118, 1817–1823 (1996).
Pai, E. F., Krengel, U., Gregoty, A. P., Goody, R. S., Kabsch, W. and Wittinghofer, A., Refiend crystal Structure of the triphosphate conformation of H-ras p21 at 1.35 Aresolution: Implications for the mechanism of GTP hydrolysis.EMBO J., 9, 2351–2359 (1990).
Park, H.-W. and Beese, L. S., Protein farnesyltransferase.Curr. Opin. Struct. Biol., 7, 873–880 (1997).
Park, H.-W., Boduluri, S. R., Moornaw, J. P., Casey, P. J. and Beese, L. S., Crystal structure of protein farnesyltransferase at 2.25 angstrom resolution.Science, 275, 1800–1805 (1997).
Patel, D. V., Gordon, E. M., Schmidt, R. J., Weller, H. N., Young, M. G., Zahler, R., Barbacid, M., Carboni, J. M., Gullo-Brown, J. L., Huniham, L., Ricca, C., Robinson, S., Seizinger, B. R., Tuomari, A. V. and Mann, V., Phosphinyl acid-based bisubstrate analog inhibitors of Ras farnesyl protein transferase.J. Med. Chem., 38, 435–42 (1995).
Pawson, T. and Schlessinger, J., SH2 and SH3 domains.J. Curr. Biol. 3, 434–442 (1993).
Reiss, Y., Brown, M. S. and Goldstein, J. L., Divalent cation and prenyl pyrophosphate specificities of the protein farnesyl-transferase from rat brain, a zinc metalloenzyme.J. Biol. Chem., 267, 6403–6408 (1992).
Reiss, Y., Goldstein, J. L., Seabra, M. C., Casey, P. J. and Brown, M. S., Inhibition of purified p21 ras farnesyl protein transferase by Cys-AAX tetrapeptide.Cell, 62, 81–88 (1990).
Strickland, C. L., Windsor, W. T., Syto, R., Wang, L., Bond, R., Wu, Z., Schwartz, J., Le, H. V., Beese, L. S. and Weber, P. C., Crystal structure of farnesyl protein transferase complexed with a CaaX peptide and farnesyl diphosphate analogue.Biochemistry, 37, 16601–16611 (1998).
Weller, V. A. and Distefano, M. D., Measurement of the α-Secondary kinetic isotope effect for a prenyltransferase by MALDI mass spectrometry.J. Am. Chem. Soc., 120, 7975–7976 (1998).
Willumsen, B. M., Christensen, A., Hubert, N. L., Papageorge, A. G. and Lowy, D. R., The p2.ras C-terminus is required for transformation and membrane association.Nature, 310, 583–586 (1984).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cho, K.N., Lee, K.I. Chemistry and biology of ras farnesyltransferase. Arch Pharm Res 25, 759–769 (2002). https://doi.org/10.1007/BF02976989
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02976989