Abstract
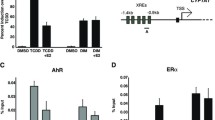
In the current study, our research focused on the estrogenic activity of isoflavonoids, mainly genistein, biochanin A and daidzein. Genistein enhanced the reporter gene expression of MCF-7-ERE-Luc cells, at a concentration as low as 10 nM, with a concentration of 100 nM the achieved gene expression effects were similar to those of 10 pM 17β-estradiol. Based on the estrogenic activities of biochanin A and daidzein, hydroxyl groups at the 4′ and 5 positions are needed for the maximal effect of the genistein. The estrogenic effects of these isoflavonoids were inhibited by the concomitant treatment with tamoxifen. The data showed that the estrogenic effects of isoflavonoids were mediated through estrogen receptors. When the isoflavonoids were tested as mixtures, the estrogenic effects were lower than the arithmetic sum of those induced by each individual isoflavonoid. The estrogenic potency of each isoflavonoid was presented at EC50 levels with a 17β-estradiol equivalent concentration (EEQ) based on the dose response of each chemical. The EC50s and EEQs of genistein, biochanin A and daidzein were 4.15, 0.89 and 0.18 μM, and 15.0, 5.12 and 1.83 μM/M, respectively. Our data clearly demonstrated that the pERE-luciferase reporter gene assay was suited for the sensitive and quantitative measurement, and large scale screening, of the estrogenicity of chemicalsin vitro.
Similar content being viewed by others
References
Andersen, H. R., Andersson, A. M., Arnold, S. F., Autrup, H., Barfoed, M., Beresford, N.A., Bjerregaard, P., Christiansen, L. B., Gissel, B., Hummel, R., Jorgensen, E. B., Korsgaard, B., Le Guevel, R., Leffers, H., McLachlan, J., Moller, A., Nielsen, J. B., Olea, N., Oles-Karasko, A., Pakdel, F., Pedersen, K. L., Perez, P., Skakkeboek, N. E., Sonnenschein, C., and Soto, A. M.et al., Comparison of short-term estrogenicity tests for identification of hormone-disrupting chemicals.Environ, Health Perspect, 107, 89–108 (1999).
Andersson, A. M. and Skakkebaek, N. E., Exposure to exogenous estrogens in food: possible impact on human development and health.Eur. J. Endocrinol., 140, 477–485 (1999).
Ansbacher, R., Selective estrogen receptor modulators.Am. J. Obstet. Gynecol., 181, 1036 (1999).
Bolger, R., Wiese, T. E., Ervin, K., Nestich, S., and Checovich, W., Rapid screening of environmental chemicals for estrogen receptor binding capacity.Environ. Health Prespect, 106, 551–557 (1998).
Breinholt, V. and Larsen, J. C., Detection of weak estrogenic flavonoids using a recombinant yeast strain and a modified MCP7 cell proliferation assay.Chem. Res. Toxicol., 11, 622–629 (1998).
Brzezinski, A. and Debi, A., Phytoestrogens: the “natural” selective estrogen receptor modulators?Eur. J. Obstet. Gynecol. Reprod. Biol., 85, 47–51 (1999).
Cassidy, A., Bingham, S., and Setchell K. D., Biological effects of a diet of soy protein rich in isoflavones on the menstrual cycle of premenopausal women.Am. J. Clin. Nutr., 60, 333–340 (1994).
Choi, Y. H., Zhang, L., Lee, W. H., and Park, K. Y., Genistein-induced G2/M arrest is associated with the inhibition of cyclin B1 and the induction of p21 in human breast carcinoma cells.Int. J. Oncol., 3, 391–396 (1998).
Constantinou, A. I., Kamath, N., and Murley, J. S., Genistein inactivates bcl-2, delays the G2/M phase of the cell cycle, and induces apoptosis of human breast adenocarcinoma MCR-7 cells.Eur. J. Cancer, 34, 1927–1934 (1998)
Delmas, P. D., Clinical use of selective estrogen receptor modulators.Bone, 25, 115–118 (1999).
Rioravanti, L., Cappelletti, V., Miodini, P., Ronchi, E., Brivio, M., and Di Fronzo, G., Genistein in the control of breast cancer cell growth: insights into the mechanism of actionin vitro.Cancer Lett., 130, 143–152 (1998).
Potsis, T., Pepper, M. S., Aktas, E., Breit, S., Rasku, S., Adlercreutz, H., Wahala, K., Montesano, R., and Schweigerer, L., Flavonoids, dietary-derived inhibitors of cell proliferation andin vitro angiogenesis.Cancer Res., 57, 2916–2921 (1997).
Gaido, K. W., Leonard, L. S., Lovell, S., Gould, J. C., Babai, D., Portier, C. J., and MacDonell, D. P., Evaluation of chemicals with endocrine modulating activity in a yeast-based steroid hormone receptor transcription assay.Toxicol. Appl. Pharmacol., 143, 205–212 (1997).
Hochner-Celnikier, D., Pharmacokinetics of raloxifene and its clinical applications.Eur. J. Obstet. Gynecol. Reprod. Biol., 85, 47–51 (1999).
Jorgensen, M., Vendelbo, B., Skakkebaek, N. E., and Leffers, H., Assaying estrogenicity by quantitating the expression of endogenous estrogen-regulated genes.Environ. Health Perspect, 108, 403–412 (2000).
Klein, K. O., Baron, J., Colli, M. J., McDonnell, D. P., and Cutler, G. B. Jr., Estrogen levels in childhood determined by an ultrasensitive recombinant cell bioassay.J. Clin. Invest., 94, 2475–2480 (1994).
Kuzumaki, T., Kobayashi, T., and Ishikawa, K., Genistein induces p21(Cip1/WAP1) expression and blocks the G1 to S phase transition in mouse fibroblast and melanoma cells.Biochem. Biophys. Res. Commun., 251, 291–295 (1998).
Masamura, S., Santner, S. J., Heitjan, D. P., and Santen, R. J., Estrogen deprivation causes estradiol hypersensitivitiy in human breast cancer cells.J. Clin. Endocrinol. Metab., 80, 2918–2925 (1995).
Meyer, T., Koop, R., Von Angerer, E., Schonenberger, H., and Holler, E., A rapid luciferase transfection assay for transcription activation effects and stability control of estrogenic drugs in cell cultures.J. Cancer Res. Clin. Oncol., 120, 359–364 (1994).
Morito, K., Aomori T., Hirose, T., Kinjo J., Hasegawa, J., Ogawa, S., Inoue, S., Muramatsu, M., and Masamune, Y., Interaction of Phytoestrogens with Estrogen Receptors α and β (II).Biol. Pharm. Bull., 25, 48–52 (2002).
Nishikawa, J., Saito, K., Goto, J., Dakeyama, F., Matsuo, M., and Nishihara, T., New screening methods for chemicals with hormonal activities using interaction of nuclear hormone receptor with coactivator.Toxico. Appl. Pharmacol., 154, 76–83 (1999).
Noroozi, M., Angerson, W. J., and Lean, M. E., Effects of flavonoids and vitamin C on oxidative DNA damage to human lymphocytes.Am. J. Clin. Nutr., 67, 1210–1218 (1998).
Soto, A. M., Sonnenschein, C., Chung, K. L., Fernandes, M. F., Olea, N., and Serrano, F. O. The E-SCREEN assay as a tool to identify estrogens: an update on estrogenic environmental pollutants.Environ. Health Perspect, 103, 113–122 (1995).
Sumpter, J. P. and Jobling, S., Vitellogenesis as a biomarker for estrogenic contamination of the aquatic environment.Environ. Health Perspect, 103, 173–178 (1995).
Wang, T. T., Sathyamoorthy, N., and Phang, J. M., Molecular effects of genistein on estrogen receptor mediated pathways.Carcinogenesis, 17, 271–275 (1996).
Weryha, G., Pascal-Vigneron, V., Klein, M., and Leclere, J., Selective estrogen receptor modulators.Curr. Opin. Rheumatol., 11, 301–306 (1999).
Xu, X., Duncan, A. M., Merz, B. E., and Kurzer, M. S., Effects of soy isoflavones on estrogen and phytoestrogen metabolism in premenopausal women.Cancer Epidemiol. Biomarkers Prev., 7, 1101–1108 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Joung, K.E., Kim, Y.W. & Sheen, Y.Y. Assessment of the estrogenicity of isoflavonoids, using MCF-7-ERE-Luc cells. Arch Pharm Res 26, 756–762 (2003). https://doi.org/10.1007/BF02976687
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02976687