Abstract
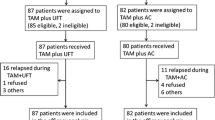
The second 5-year study of postoperative adjuvant therapy in patients with breast cancer between 1985 and 1988 was performed by the Study Group for Adjuvant Chemoendocrine Therapy for Breast Cancer (ACETBC). This report describes the results of a meta-analysis of the outcome. A total of 3012 patients with stage II, estrogen-receptor-positive primary breast cancer who underwent radical surgery (total mastectomy with at least axillary dissection) were eligible for the analyses. On the day of surgery, all patients received 13 mg/m2 of mitomycin C (MMC) intravenously. Patients were then given one of two adjuvant chemoendocrine therapies for 2 years: regimen A, consisting of tamoxifen citrate (TAM) 30 mg/day, or regimen B, consisting of TAM 30 mg/day plus tegafur (Futraful, FT) 600 mg/day. The results from all eligible patients were included in a meta-analysis according to the method of Petoet al. The odds reduction rate was 12 ± 13% (log-rank test,p = 0.35) for the 5-year survival rate and 16±10% (log-rank test, p = 0.093) for the 5-year non-recurrence rate. In terms of the 5-year non-recurrence rate, regimen B (with FT) yielded slightly, but not significantly, better results than regimen A. In addition, stratified analyses were carried out with respect to the 5-year non-recurrence rate. Regimen B (with FT) yielded significantly better results than regimen A in patients with four or more positive axillary nodes (log-rank test,p = 0.039) and yielded slightly, but not significantly, better results than regimen A in premenopausal patients (log-rank test,p = 0.079).
Similar content being viewed by others
Abbreviations
- ACETBC:
-
Adjuvant chemoendocrine therapy for breast cancer
- ER:
-
Estrogen receptor
- MMC:
-
Mitomycin C
- TAM:
-
Tamoxifen citrate
- FT:
-
Futraful
- CMF:
-
Cyclophosphamide+methotrexate+fluorouracil
- CPA:
-
Cyclophosphamide
- EBCTCG:
-
Early Breast Cancer Trialisrs’ Collaborative Group
- QOL:
-
Quality of life
References
Rossi A, Bonadonna G, Valagussa P,et al: Multimodal treatment in operable breast cancer; Five- year results of the CMF programme.Br Med J 282: 1427–1431, 1981.
Yoshimoto M, Kasumi F, Watanabe S,et al: Ten- year follow-up mitomycin C single agent adjuvant chemotherapy for breast cancer. In: Taguchi T, Andrysek O eds, New Trends in Cancer Chemotherapy with Mitomycin C, Excerpta Medica, Tokyo, pp58–67, 1987.
Konda C, Niitani H, Sakauchi N,et al: Chemotherapy of cancer with oral administration of N1-(2-furanidil)- 5-fluorouracil [FT-207].Jpn J Cancer Clin 19:495- 499, 1973 (in Japanese with English summary).
Ansfield FJ, Kallas GJ, Singson JP: Phase III studies of oral tegafur (Futraful).J Clin Oncol 1:107–110, 1983.
Adachi I: Clinical studies on the treatment of the patients with advanced breast cancer.Jpn J Cancer Chemother 21:72–78, 1981 (in Japanese with English summary).
Wada T, Koyama H, Nishizawa Y,et al: Chemoendocrine therapy for advanced breast cancer; A combined treatment with tamoxifen and FT-207.J Jpn Soc Cancer Ther 16:51–55, 1981 (in Japanese with English summary).
Abe O: The role of chemoendocrine agents in postoperative adjuvant therapy for breast cancer; Metaanalysis of the first collaborative studies of postoperative adjuvant chemoendocrine therapy for breast cancer (ACETBC).Breast Cancer 1:1–9, 1994.
Goldhirsch A: Meeting highlights; International panel on the treatment of primary breast cancer.JNCI 87: 1441–1445, 1995.
Early Breast Cancer Trialists’ Collaborative Group: Treatment of Early Breast Cancer, Vol 1; Worldwide Evidence 1985–1990. Oxford University Press, Oxford, pp12–19, 1990.
Sakamoto G: Characteristics and future prospect of breast cancer in Japan.Jpn J Breast Cancer 2:327–338, 1987 (in Japanese with English summary).
Baum M: Prospects for the future in the management of carcinoma of the breast; The biological fall out from clinical trials. In: Jones SE, Salmon SE eds, Adjuvant Therapy of Cancer IV, Groune & Stratton Inc, Orlando, pp321–329, 1984.
Tormy DC, Taylor SG, Kalish LA,et al: Adjuvant systemic therapy in premenopausal (CMF, CMFP, CMFPT) and postmenopausal (observation, CMFP, CMFPT) women with node positive breast cancer. In: Jones SE, Salmon SE eds, Adjuvant Therapy of Cancer IV, Groune & Stratton Inc, Orlando, pp359- 368, 1984.
Fisher B: Treatment of primary breast cancer with L- PAM/5-FU and tamoxifen; An interim report.Breast Cancer Res Treat 3(Suppl 1):7–17, 1983.
Palshof T, Carstensen B, Mouridsen HT,et al. Reviews on Endocrine-Related Cancer 17(Suppl):43- 50, 1985.
Wallgren A, Baral E, Carstensen J,et al: Should adjuvant tamoxifen be given for several years in breast cancer? In: Jones SE, Salmon SE eds, Adjuvant Therapy of Cancer IV, Groune & Stratton Inc, Orlando, pp331–337, 1984.
Patterson JS, Battersby LA, Edwards DG: Review of the clinical pharmacology and international experience with tamoxifen in advanced breast cancer. In: Lacobelli S, {eaet al} eds, The Role of Tamoxifen in Breast Cancer, Raven Press, ppl7–29, 1982.
Margreiter R, Wiegele J: Tamoxifen (Nolvadex) for premenopausal patients with advanced breast cancer.Breast Cancer Res Treat 4:45–48, 1984.
Tancini G, Bonadonna G, Valagussa P,et al: Adjuvant CMF in breast cancer; Comparative 5-year results of 12 versus 6 cycles.J Clin Oncol 1:2–10, 1983.
Smalley RV, Bartolucci AA, Moore M,et al: Southeastern Cancer Study Group: Breast cancer studies 1972–1982.Int J Radiat Oncol Biol Phys 9:1867–1874, 1983.
Brincker H, Mouridsen HT, Andersen KW: Adjuvant chemotherapy with cyclophosphamide or CMF in premenopausal woman with stage II breast cancer.Breast Cancer Res Treat 3:91–95, 1983.
Howell A, Bush H, George WD,et al: Controlled trial of adjuvant chemotherapy with cyclophosphamide, methotrexate, and fluorouracil for breast cancer.Lancet 2:307–311, 1984.
Nissen-Meyer R, Host H, Kjellgren K,et al: Scandinavian trials with short postoperative course versus a 12-cycle course.Recent Results Cancer Res 96:48- 54, 1984.
Tormy DC, Weinberg VE, Holland JF,et al: A randomized trial of five and three drug chemotherapy and chemoimmunotherapy in women with operable node positive breast cancer.J Clin Oncol 1:138–145, 1983.
Izuo M: Clinicopathological features of recurrent breast cancer.Jpn J Cancer Chem 12(Part I):412–420, 1985 (in Japanese with English summary).
Buyse M: Potential and pitfalls of randomized clinical trials in cancer research.Cancer Surv 8:91–105, 1989.
Early Breast Cancer Trialists’ Collaborative Group: Systemic treatment of early breast cancer by hormonal, cytotoxic or immune therapy.Lancet 339:1–15, 71-85, 1992.
Nomura Y, Tominaga T, Adachi I,et al: Clinical evaluation of cyclophosphamide, methotrexate and 5- fluorouracil (CMF) on advanced and recurrent breast cancer.Jpn J Cancer Chemother 21:1949–1956, 1994 (in Japanese with English summary).
Tormy C: Adjuvant systemic therapy in postoperative node-positive patients with breast carcinoma; The GALGB trial and ECOG premenopausal trial.Recent Results Cancer Res 96:153–165, 1984.
Yasumura T, Oka T, Honjo H: Menstrual abnormality in breast cancer patients receiving adjuvant endocrino-chemotherapy.Jpn J Cancer Chemother 15:2947- 2952, 1988 (in Japanese with English summary).
Author information
Authors and Affiliations
About this article
Cite this article
Yoshida, M., Abe, O., Uchino, J. et al. Meta-analysis of the second collaborative study of adjuvant chemoendocrine therapy for breast cancer (acetbc) in patients with stage ii, estrogen-receptor-positive breast cancer. Breast Cancer 4, 93–101 (1997). https://doi.org/10.1007/BF02967062
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02967062