Conclusions
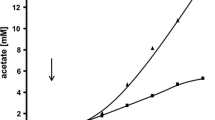
pH-dependent energetic uncoupling ofZymomonas by acetic acid occurs by virtue of the permeability of the plasma membrane to the undissociated form of acetic acid (HAc) and the acidification of the cytoplasm resulting from the uptake of HAc and the consequential diversion of energy away from biosynthetic processes (growth) in order to maintain constant intracellular pH. Energetic uncoupling is manifested by an increase in specific productivity. The degree of uncoupling caused by HAc depends on a rather complex interaction between several different variables including membrane permeability, the transmembrane δpH and the concentraiton of undissociated form of acetic acid in the medium. Within the pH range of 5.0–5.5, maximal energic uncoupling is produced by 30–38 mM HAc. For practical purposes, in terms of the concentration of acetic acid, this corresponds to about 5 and 15 g/L at pH 5.0 and 5.5, respectively. Assuming any upper limit concentration of acetic acid in hydrolysate fermentation media of about 12 g/L, inhibition of Z.mobilis in terms of both ethanol yield and productivity is avoided by controlling the pH in the range of 5.5–6.0.
Similar content being viewed by others
Abbreviations
- HAc:
-
undissociated acetic acid
- Μ:
-
specific growth rate (h−1)
- Yx/s :
-
growth yield co-efficient (g dry wt cells/g glucose)
- Yp/s :
-
product yield (g ethanol/g glucose)
- Qp :
-
average volumetric productivity (g ethanol/L/h)
- qp :
-
specific productivity (g ethanol/g cell/h).
References
Wright, J. D. (1988),Chem. Eng. Progress 84, 62–68.
Wyman, C. E. and Hinman, N. D. (1990),Appl. Biochem. Biotechnol. 24/25, 735–753.
Hinman, N. D., Wright, J. D., Hoagland, W., and Wyman, C. E. (1989),Appl. Biochem. Biotechnol. 20/21, 391–401.
Keim, C. R. (1983),Enzyme Microbiol. Technol. 5, 103–114.
Busche, R., Scott, C. D., Davidson, B. H., and Lynd, L. R. (1992),Appl. Biochem. Biotechnol. 34/35, 395–417.
Maiorella, B. L., Blanch, H. W., and Wilke, C. R. (1984),Bioeng. Biotechnol. 26, 1003–1025.
Ohta, K., Supanwong, K., and Hayashida, S. (1981),J. Ferment. Technol. 59, 435–439.
Lawford, H. G. (1988),Appl. Biochem. Biotechnol. 17, 203–219.
Lawford, H. G. (1988), Proc. VIII Int’l Symp. on Alcohol Fuels, Pub. by NEDO, Tokyo, pp. 21–27.
Lawford, H. G. and Ruggiero, A. (1990), inBioenergy, Proc. 7th Cdn. Bioenergy R&D Seminar, Hogan, E., ed., NRC Canada, pp. 401–408.
Rogers, P. L., Lee, K. J., Skotnicki, M. L., and Tribe, D. E. (1982),Adv. Biochem. Eng. 23, 37–84.
Doelle, H. W., Kirk, L., Crittenden, R., Toh, H., and Doelle, M. B. (1993),CRC Rev. Biotechnol. 13(1), 57–98.
Rodriguez, E. and Callieri, D. A. S. (1986),Biotechnol. Letts 8, 745–748.
Doelle, M. B., Greenfield, P. F., and Doelle, H. W. (1990),Proc. Biochem. 25(5), 151–156.
Beavan, M., Zawadzki, B., Droniuk, R., Fein, J., and Lawford, H. G. (1989),Appl. Biochem. Biotechnol. 20/21, 319–326.
Bringer, S., Sahm, H., and Swyzen, W. (1984),Biotechnol. Bioeng. Symp. 14, 311–319.
Lee, G. M., Kim, C. H., Lee, K. J., Zainal Abidin Mohd. Yusof, Han, M. H., and Rhee, S. K. (1986),J. Ferment Technol. 64, 293–297.
Parekh, S. R., Parekh, R. S., and Wayman, M., (1989),Proc. Biochem. 24, 88–91.
Freese, E., Sheu, C. W., and Galliers, E. (1973),Nature 241, 321.
Lynd, L. R. (1990),Appl. Biochem. Biotechnol. 24/25, 695–719.
Grethlein, H. E. (1985),Bio/Technolgy 3, 155–160.
Lawford, H. G. and Rousseau, J. D. (1993), inEnergy from Biomass & Wastes XVI, Klass, D. L., ed., Institute of Gas Technology, Chicago, IL, pp. 559–597.
Lawford, H. G. and Rousseau, J. D. (1993),Appl. Biochem. Biotechnol. 39/40, 301–322.
Lawford, H. G. and Rousseau, J. D. (1992),Appl. Biochem. Biotechnol. 34/35, 205–216.
Lawford, H. G. and Rousseau, J. D. (1993),Appl. Biochem. Biotechnol. 39/40, 687–699.
Nicholls, D. G. (1982), Bioenergetics—anintroduction to the chemiosmotic theory, Academic, Toronto, pp. 56–58.
Swings, J. and De Ley, J. (1977),Bacteriol. Rev. 41, 1–46.
Lawford, H. G. and Ruggiero, A. (1990),Biotechnol. Appl. Biochem. 12, 206–211.
Pampulha, M. E. and Louriero, V. (1989),Biotechnol. Letts. 11, 269–274.
Mitchell, P. (1973),J. Bioenergetics 4, 63–91.
Pankova, L. M., Shvinka, J. E., and Beker, M. J. (1988),Appl. Microbiol. Biotechnol. 28, 583–588.
Pirt, S. J. (1975),Principles of Microbe and Cell Cultivation, Blackwell, London, UK, pp. 66–68.
Maiorella, B. L., Blanch, H. W., and Wilke, C. R. (1983),Biotechnol. Bioeng. 25, 103–121.
Vega, J. L., Claussen, E. C., and Gaddy, J. L. (1987),Biotechnol. Bioeng. 29, 429–435.
Lavers, B. H., Pang, P., MacKenzie, C. R., Lawford, G. R., Pik, J., and Lawford, H. G. (1981), inAdvances in Biotechnology, Moo-Young, M. and Robinson, C. W., eds., vol. II, Pergamon, Toronto, pp. 195–204.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lawford, H.G., Rousseau, J.D. The pH-dependent energetic uncoupling ofZymomonas by acetic acid. Appl Biochem Biotechnol 45, 437–448 (1994). https://doi.org/10.1007/BF02941818
Issue Date:
DOI: https://doi.org/10.1007/BF02941818