Abstract
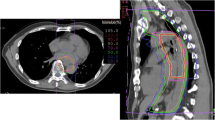
A new dosage formulation consisting of an anticancer drug bound to activated carbon particles was developed for the treatment of digestive cancer in patients in whom operation is contraindicated. The new formulation is designed to distribute higher levels of anticancer drug to the regional lymph nodes and at the injection site compared to distribution of the drug in aqueous solution. In 12 patients with histologically proven carcinoma (7 with superficial esophageal cancer and 5 with early or proper muscle layer-infiltrating gastric cancer), an anticancer drug bound to carbon particles (total dose, 40–100mg peplomycin or 250–500mg methotrexate per person) was injected endoscopically into the primary lesions. Eleven of the 12 patients are currently alive, 12–64 months after therapy, or they died without evidence of cancer 12–98 months after the treatment. One patient has remained cancer-free for 32 months after a second course of the new formulation therapy given to treat a recurrence detected 26 months after the first treatment. Endoscopic injection of this new dosage formulation seems to control these digestive cancers in patients in whom operation is contraindicated.
Similar content being viewed by others
References
Goseki N, Koike M, Yoshida M. Histopathologic characteristics of early stage esophageal carcinoma. Cancer 1992;69:1088–1093.
Ide H. Surgical treatment of superficial esophageal cancer (in Japanese with English abstract). Rinshou Kyoubu-Geka (J Ann Thorac Surg) 1991;11:191–197.
Hioki K, Nakane Y, Yamamoto M. Surgical strategy for early gastric cancer. Br J Surg 1990;77:1330–1334.
Sano T, Kobori O, Muto T. Lymph node metastasis from early gastric cancer: Endoscopic resection of tumour. Br J Surg 1992;79:241–244.
Makuuchi H, Machimura T, Soh Y, et al. Endoscopic mucosectomy for mucosal carcinomas in the esophagus. Jpn J Gastroenterol Surg 1991;24:2599–2603.
Inoue H, Endo M. Endoscopic esophageal mucosal resection using a transparent tube. Surg Endosc 1990;4:198–201.
Takemoto T, Yanai H, Tada M, et al. Evaluation of endoscopic resection for early gastric cancer (in Japanese with English abstract). Stomach and Intestine 1991;26:365–370.
Oguro Y. Laser endoscopic treatment for early gastric cancer. J Gastroenterol 1994;29(Suppl VII):91–95.
Hiki Y, Shimano H, Yamao Y, et al. The concepts, procedures, and problems related to endoscopic laser therapy of early gastric cancer. Surg Endosc 1989;3:1.
Ballard BE. Biopharmaceutical considerations in subcutaneous and intramuscular drug administration. J Pharm Sci 1968;57:357–378.
Hagiwara A, Takahashi T, Ueda T, et al. Activated carbon particles as anticancer drug carrier into regional lymph nodes. Anti-Cancer Drug Des 1987;1:313–321.
Hagiwara A, Takahashi T, Sawai K, et al. Selective delivery of a greater amount of methotrexate to regional lymph node using a new dosage formulation, methotrexate adsorbed on activated carbon particles, in rats. Anti-Cancer Drugs 1994;5:194–198.
Hagiwara A, Takahashi T, Ueda T, et al. Enhanced therapeutic efficacy on lymph node metastasis by the use of peplomycin adsorbed on small activated carbon particles. Anticancer Res 1988;8:287–290.
Hagiwara A, Takahashi T, Sawai K, et al. Methotrexate bound to carbon particles for treating cancers with lymph node metastases, in animal experiments and clinical pilot study. Cancer 1996; 78:2199–2209.
Hagiwara A, Takahashi T, Sawai K, et al. Lymph nodal vital staining with newer carbon particle suspensions compared with india ink—experimental and clinical observations. Lymphology 1992;25:84–89.
Matsuda A, Yoshioka O, Yamashita T, et al. Fundamental and clinical studies on new bleomycin analogues. Recent Results in Cancer Research “Antitumor Antibiotics” 1978;63:191–210.
Japanese classification of gastric carcinoma. Japanese Research Society for Gastric Cancer. First English edition. Tokyo: Kanehara, 1995.
Rusznyak I, Foldi M, Szabo G. Absorption into lymph capillaries. In: Youlten L (ed) Lymphatics and lymph circulation; physiology and pathology. 2nd ed. London: Pergamon, 1967;418–474.
Hagiwara A, Ahn T, Ueda T, et al. Anticancer agents adsorbed by activated carbon particles. A new form of dosage-enhancing efficacy on lymph node metastases. Anticancer Res 1986;6:1005–1008.
Ignoffo RJ, Friedman MA. Therapy of local toxicities caused by extravasation of cancer chemotherapeutic drugs. Cancer Treat Rev 1980;7:17–27.
Terashima T, Umezawa H. Lethal effect of bleomycin on cultured mammalian cells. J Antibiotics 1970;23:300–304.
Keefe DA, Capizzi RL, Rudnick SA. Methotrexate cytotoxicity for L5178Y/Asn lymphoblast: Relationship of dose and duration of exposure to tumor cell viability. Cancer Res 1982;42:1641–1645.
Sikic BI, Collins JM, Mimnaugh EG, et al. Improved therapeutic index of bleomycin when administered by continuous infusion in mice. Cancer Treat Rep 1978;62:2011–2017.
Freeman-Narrod M, Narrod SA. Chronic toxicity of methotrexate in mice. J Natl Cancer Inst 1977;58:735–741.
Blum RH, Carter SK, Agre K. A clinical review of bleomycin—a new anti-neoplastic agent. Cancer 1973;32:903–914.
Hagiwara A, Takahashi T, Ueda T, et al. Reduced pulmonary toxicity of peplomycin in a new drug-delivery system. Anti-Cancer Drug Des 1988;2:319–324.
Hagiwara A, Takahashi T, Sawai K, et al. Reduced toxicity of a new formulation. Anti-Cancer Drugs 1994;5:31–34.
Hagiwara A, Takahashi T, Sawai K, et al. Endoscopic injection of methotrexate bound to activated carbon particles. In: Proceedings of 1st International Gastric Cancer Congress. Bologna: Monduzzi Editore, 1995:1643–1646.
Japanese Society for Esophageal Diseases. Guidelines for the clinical and pathologic studies on carcinoma of the esophagus. 8th ed. Tokyo: Kanehara, 1992.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hagiwara, A., Takahashi, T., Kitamura, K. et al. Endoscopic local injection of a new drug delivery formulation, anticancer drug bound to carbon particles, for digestive cancers: Pilot study. J Gastroenterol 32, 141–147 (1997). https://doi.org/10.1007/BF02936359
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02936359