Abstract
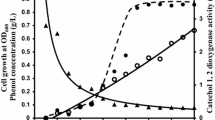
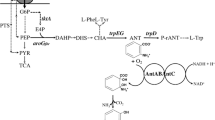
For enhancement ofcis,cis-muconate productivity from benzoate, catechol 1,2-dioxygenase (C12O) which catalyzes the rate-limiting step (catechol conversion tocis,cis-muconate) was cloned and expressed in recombinantPseudomonas putida BCM114. At higher benzoate concentrations (more than 15 mM),cis,cis-muconate productivity gradually decreased and unconverted catechol was accumulated up to 10 mM in the case of wildtypeP. putida BM014, whereascis,cis-muconate productivity continuously increased and catechol was completely transformed tocis,cis-muconate forP. putida BCM114. Specific C12O activity ofP. putida BCM114 was about three times higher than that ofP. putida BM014, and productivity was enhanced more than two times.
Similar content being viewed by others
References
Mizuno, S., N. Yoshikawa, M. Seki, T. Mikawa, and Y. Imada (1988) Microbial production ofcis,cis-muconic acid from benzoic acid.Appl. Microbiol. Biotechnol. 28:20–25.
Smith, M. R. and C. Ratledge (1989) Quantitative biotransformation of catechol tocis,cis-muconate.Biotechnol. Lett. 11:105–110.
Yoshikawa, N., S. Mizuno, K. Ohta and M. Suzuki (1990) Microbial production ofcis,cis-muconic acid.J. Biotechnol. 14:203–210.
Draths, K. M. and J. W. Frost (1994) Environmentally compatible synthesis of adipic acid from D-glucose.J. Am. Chem. Soc. 116:399–400.
Bang, S. G. and C. Y. Choi (1995) DO-stat fedbatch production ofcis,cis-muconic acid from benzoic acid byPseudomonas putida BM014.J. Ferment. Bioeng. 79:381–383.
Choi, W. J., E. Y. Lee, M. H. Cho and C. Y. Choi (1997) Enhanced production ofcis,cis-muconate in a cell-recycle bioreactor.J. Ferment. Bioeng. 84:70–76.
Bayly, R. C. and D. L. Mckenzie (1976) Catechol oxygenases ofPseudomonas putida mutant strains.J. Bacteriol. 127:1098–1107.
Kukor, J. J., R. H. Olsen, and D. P. Ballou (1989) Cloning and expression of thecatA andcatBC gene clusters fromPseudomonas aurogenosa PAO.J. Bacteriol. 170:4458–4465.
Simon, R., U. Priefer, and A. Pühler (1983) A broad host range mobilization system forin vivo genetic engineering: Transposon mutagenesis in Gram negative bacteria.Biotechnol. 1:784–791.
Knauf, B. C. and E. W. Nester (1982) Wide host range cloning vectors: A cosmid clone bank of anAgrobacterium Ti plasmid.Plasmid 8:45–54.
Bang, S. G., W. J. Choi, C. Y. Choi and M. H. Cho (1996) Production ofcis,cis-muconic acid from benzoic acidvia microbial transformation.Biotechnol. Bioprocess Eng. 1:36–40.
Sambrook, J., E. F. Fritsch, and T. Maniatis (1989) Molecular Cloning, USA: Cold Spring Habor Laboratory Press.
Friedrich, B., C. Hogrefe and H. G. Schlegel (1981) Naturally occurring genetic transfer of hydrogen-oxidizing ability between strainsof Alcaligenes eutrophus.J. Bacteriol. 147:198–205.
Neidle, E. L. and L. N. Ornston (1986) Cloning and expression ofAcinetobacter calcoaceticus catechol 1,2-dioxygenase structural genecatA inEscherichia coli.J. Bacteriol. 168:815–820.
Kivisaar, M., L. Kasak and A. Nurk (1991) Sequence of the plasmid-encoded catechol 1,2-dioxygenase-expressing gene,pheB of phenoldegradingPseudomonas sp. Strain EST1001.Gene 98:15–20.
Nakai, C., H. Uyeyama, H. Kagamiyama, T. Nakazawa, S. Inouye, F. Kishi, A. Nakazawa, and M. Nozaki (1995) Cloning, DNA sequencing, and amino acid sequencing of catechol 1,2-dioxygenases (Pyrocatechase) fromPseudomonas putida mt-2 andPseudomonas arvilla C-1.Archiv. Biochem. Biophys. 321:353–362.
Ditta, G., S. Stanfield, D. Corbin and D. R. Helinski (1980) Broad host range DNA cloning system for Gram-negative bacteria: Construction of a gene bank ofRhizobium meliloti.Proc. Natl. Acad. Sci. USA 77:7347–7351.
Yamaguchi, M. and H. Fujisawa (1980) Purification and characterization of an oxygenase component in benzoate 1,2-dioxygenase system fromPseudomonas arvilla C-1.J. Biol. Chem. 255:5058–5063.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, B.J., Choi, W.J., Lee, E.Y. et al. Enhancement ofcis,cis-muconate productivity by overexpression of catechol 1,2-dioxygenase inPseudomonas putida BCM114. Biotechnol. Bioprocess Eng. 3, 112–114 (1998). https://doi.org/10.1007/BF02932513
Issue Date:
DOI: https://doi.org/10.1007/BF02932513