Abstract
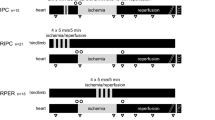
Objectives: Ischemic preconditioning has been used to induce the myocardium to adapt to ischemic stress preceded by short periods of ischemia and reperfusion. We used a sheep right heart bypass model with a conductance catheter to assess the cardioprotective effect of ischemic preconditioning on 30-minute normothermic global myocardial ischemia.Methods: Ischemic preconditioning was conducted in 6 sheep in 3 5-minute aortic cross-clampings intersperesed with 5 minutes of reperfusion during cardiopulmonary bypass, with 6 sheep as time-matched controls. Global myocardial ischemia was subsequently achieved in 30-minute aortic cross-clamping with left ventricular unloading during normothermic cardiopulmonary bypass. Weaning from cardiopulmonary bypass was conducted 40 minutes after reperfusion. Before ischemia and 40, 70, and 100 minutes after reperfusion, left ventricular pressure-volume loops were measured using a conductance catheter during right heart bypass preparation. Left ventricular contractility, diastolic function, and mechanical efficiency were then evaluated. Right heart bypass was instituted to control the preload and to decompress the right ventricle completely, thereby eliminating parallel conductance variation.Results: No differences in the studied parameters were seen between ischemic-preconditioning and control groups before ischemia. Left ventricular contractility, diastolic function, and mechanical efficiency in the ischemic-preconditioning group were significantly superior to those in the control group after reperfusion.Conclusions: Ischemic preconditioning attenuates postischemic myocardial dysfunction in a sheep model using 30-minute unloaded normothermic global myocardial ischemia. Ischemic preconditioning would thus be clinically significant when the ischemic damage is severe.
Similar content being viewed by others
References
Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation 1986; 74: 1124–36.
Reimer KA, Murry CE, Yamasawa I, Hill ML, Jennings RB. Four brief periods of myocardial ischemia cause no cumulative ATP loss or necrosis. Am J Physiol 1986; 251:H1306–15.
Cave AC, Hearse DJ. Ischaemic preconditioning and contractile function: studies with normothermic and hypothermic global ischaemia. J Mol Cell Cardiol 1992, 24: 1113–23.
Hendrikx M, Toshima Y, Mubagwa K, Flameng W. Improved functional recovery after ischemic preconditioning in the globally ischemic rabbit heart is not mediated by adenosine A1 receptor activation. Basic Res Cardiol 1993, 88: 576–93.
Bolling SF, Olszanski DA, Childs KF, Gallagher KP, Ning XH. Stunning, preconditioning, and functional recovery after global myocardial ischemia Ann Thorac Surg 1994, 58: 822–7.
Jenkins DP, Pugsley WB, Yellon DM. Ischaemic preconditioning in a model of global ischaemia infarct size limitation, but no reduction of stunning. J Mol Cell Cardiol 1995, 27: 1623–32.
Valen G, Takeshima S, Vaage J. Preconditioning improves cardiac function after global ischemia but not after cold cardioplegia. Ann Thorac Surg 1996, 62: 1397–403.
Kolocassides KG, Galinanes M, Hearse DJ. Ischemic preconditioning, cardioplegia or both? Differing approaches to myocardial and vascular protection. J Mol Cell Cardiol 1996, 28: 623–34.
Faris B, Pevnet J, Wassef M, Bel A, Mouas C, Duriez M, et al. Failure of preconditioning to improve postcardioplegia stunning of minimally infarcted hearts. Ann Thorac Surg 1997; 64: 1735–41.
Tanoue Y, Sese A, Ueno Y, Joh K, Hijii T. Bidirectional Glenn procedure improves the mechanical efficiency of a total cavopulmonary connection in high-risk Fontan candidates. Circulation 2001, 103: 2176–80.
Tanoue Y, Morita S, Ochiai Y, Haraguchi N, Tominaga R, Kawachi Y, et al. Nitroglycerin as a nitric oxide donor accelerates lipid peroxidation but preserves ventricular function in a canine model of orthotopic heart transplantation. J Thorac Cardiovasc Surg 1999; 118: 547–56.
Tanoue Y, Morita S, Ochiai Y, Hisahara M, Masuda M, Kawachi Y, et al. Inhibition of lipid peroxidation with the lazaroid U74500 A attenuates ischemia-reperfusion injury in a canine orthotopic heart transplantation model. J Thorac Cardiovasc Surg 1996; 112 1017–26.
Baan J, van der Velde ET, de Bruin HG, Smeenk GJ, Koops J, van Dijk AD, et al. Continuous measurement of left ventricular volume in animals and humans by conductance catheter. Circulation 1984, 70: 812–23.
Suga H. Ventricular energetics. Physiol Rev 1990, 70: 247–77.
Glower DD, Spratt JA, Snow ND, Kabas JS, Davis JW, Olsen CO, et al. Linearity of the Frank-Starling relationship in the intact heart the concept of preload recruitable stroke work. Circulation 1985; 71: 994–1009.
Little WC. The left ventricular dP/dtmax-end-diastolic volume relation in closed-chest dogs. Circ Res 1985; 56: 808–15.
Gaasch WH, Battle WE, Oboler AA, Banas JS, Jr. Levine HJ. Left ventireular stress and compliance in man With special reference to normalized ventricular function curves. Circulation 1972, 45: 746–62.
Mirsky I, Pasipoularides A. Clinical assessment of diastolic function. Prog Cardiovasc Dis 1990, 32: 291–318.
Sunagawa K, Maughan WL, Burkhoff D, Sagawa K. Left ventricular interaction with arterial load studied in isolated canine ventricle. Am J Physiol 1983, 245 H773–80.
Nozawa T, Yasumura Y, Futaki S, Tanaka N, Uenishi M, Suga H. Efficiency of energy transfer from pressure-volume area to external mechanical work increases with contractile state and decreases with afterload in the left ventricle of the anesthetized closed-chest dog. Circulation 1988; 77: 1116–24.
Perrault LP, Menasche P, Bel A, de Chaumaray T, Peynet J, Mondry A, et al. Ischemic preconditioning in cardiac surgery a word of caution. J Thorac Cardiovasc Surg 1996, 112: 1378–86.
Kaukoranta PK, Lepojarvi MP, Ylitalo KV, Kiviluoma KT, Peuhkurinen KJ. Normothermic retrograde blood cardioplegia with or without preceding ischemic preconditioning. Ann Thorac Surg 1997; 63: 1268–74.
Cremer J, Steinhoff G, Karck M, Ahnsell T, Brandt M, Teebken OE, et al. Ischemic preconditioning prior to myocardial protection with cold blood cardioplegia in coronary surgery. Eur J Cardiothorac Surg 1997, 12: 753–8.
Jenkins DP, Pugsley WB, Alkhulaifi AM, Kemp M, Hooper J, Yellon DM. Ischaemic preconditioning reduces troponin T release in patients undergoing coronary artery bypass surgery. Heart 1997, 77: 314–8.
Alkhulaifi AM, Yellon DM, Pugsley WB. Preconditioning the human heart during aorto-coronary bypass surgery. Eur J Cardiothorac Surg 1994; 8: 270–6.
Flameng W. New strategies for intraoperative myocardial protection. Curr Opin Cardiol 1985, 10: 577–83.
Galinanes M, Argano V, Hearse DJ. Can ischemic preconditioning ensure optimal myocardial protection when delivery of cardioplegia is impaired? Circulation 1995, 92: II389–94.
Liu GS, Thornton J, Van Winkle DM, Stanley AW, Olsson RA, Downey JM. Protection against infarction afforded by preconditioning is mediated by Al adenosine receptors in rabbit heart. Circulation 1991, 84: 350–6.
Goto M, Liu Y, Yang XM, Ardell JL, Cohen MV, Downey JM. Role of bradykinin in protection of ischemic preconditioning in rabbit hearts. Circ Res 1995, 77: 611–21.
Schultz JJ, Hsu AK, Gross GJ. Ischemic preconditioning and morphine-induced cardioprotection involve the delta (δ)-opioid receptor in the intact rat heart. J Mol Cell Cardiol 1997; 29: 2187–95.
Author information
Authors and Affiliations
Additional information
Read at the Fifty-fourth Annual Meeting of The Japanese Association for Thoracic Surgery Osaka October 3–5, 2001.
Rights and permissions
About this article
Cite this article
Tanoue, Y., Herijgers, P., Meuris, B. et al. Cardioprotective effect of ischemic preconditioning on global myocardial ischemia in a sheep right heart bypass model. Jpn J Thorac Cardiovasc Surg 50, 23–29 (2002). https://doi.org/10.1007/BF02913482
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02913482