Abstract
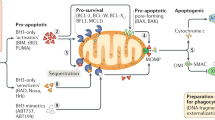
Apoptosis a physiological mechanism that eliminates excessive, damaged or unwanted cells, is a highly regulated pathway important for maintaining homeostasis in multicellular organisms. It can be initiated through various signals via the extrinsic pathway which involves death receptors, or via the intrinsic pathway which is initiated by intracellular damage and involves the mitochondria and release of cytochrome c from it to further activate caspases. The Bcl-2 family of proteins is situated upstream to the irreversible damage of cellular constituents and is an important checkpoint in the fate of a cell. The pro-apoptotic members, BH3 only members include BID, BAD and BIM. They directly or indirectly activate multidomain BAX/BAK that constitute the requisite gateway to the intrinsic pathway which operates at the mitochondrial surface and endoplasmic reticulum. In contrast, antiapoptotic members such as Bcl-2, Bcl-XL bind and sequester activation. Downstream of mitochondria, the apoptosome involvement is seen to generate caspase activity. Post mitochondria regulation involves IAPs, and their inhibitors. The pathogenesis of several diseases such as cancer, neurodegenerative disorders, autoimmune disorders, heart disease, infectious diseases including AIDS is closely related to aberrant apoptosis. Consequently interest has emerged in employing various the rapeutic approaches such as gene therapy, antisense therapy, recombinant biologicals, organic and combinatorial chemistry, to specifically target apoptosis signaling pathways such as death receptors FAS/TRAIL, Bcl-2, p53, IAPs, SMAC and caspases, etc. and are now advancing from preclinical to clinical phase.
Similar content being viewed by others
References
Kerr JF, Wyllie AH, Currie AR. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 1972; 26:239–57.
Sulston JE, Horvitz HR. Post-embryonic cell lineages of the nematode, Caenorhabditis elegans. Dev Biol 1977; 56:110–56.
Singh N, Anand S. Cell death by apoptosis. Indian J Exp Biol 1994; 32:843–7.
Savill J, Fadock V. Corpse clearance defined the meaning of cell death. Nature 2000; 407:784–88.
Philchenkov AA. Caspases as regulators of apoptosis and other cell functions. Biochemistry (Mosc) 2003; 68:365–76.
Hengartner MO. The biochemistry of apoptosis. Nature. 2000; 407:770–76.
Wajant H. The Fas signaling pathway: more than a paradigm. Science 2002; 296:1635–36.
Schultz DR, Harrington WJ Jr. Apoptosis: programmed cell death at a molecular level. Semin Arthritis Rheum 2003; 32:345–69.
Rao RV, Ellerby HM, Bredesen DE. Coupling endoplasmic reticulum stress to the cell death program. Cell Death Diff 2004; 11:372–80.
Singh N. Apoptosis-new concepts in molecular medicine. Ind J Clinical Biochem 1995; 10:54–6.
Singh N, Sun Y, Nakamura K, Smith MR, Colburn NH. C-JUN/AP-1 as possible mediators of tumor necrosis factor-alpha-induced apoptotic response in mouse JB6 tumor cells. Oncology Research 1995; 7:353–62.
Nagata S. Apoptosis by death factor. Cell 1997; 88:355–65.
Reed JC. Bcl-2 family proteins. Oncogene 1998; 17:3225–36.
Wei MC, Zong WX, Cheng EH, Lindsten T, Panoutsakopoulou V, Ross AJ, et al. Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death. Science 2001; 292:727–30.
Scorrano L, Oakes SA, Opferman JT, Cheng EH, Sorcinelli MD, Pozzan T, et al. BAX and BAK regulation of endoplasmic reticulum Ca2+: a control point for apoptosis. Science 2003; 300:135–9.
Cheng EH, Wei MC, Weiler S, Flavell RA, Mak TW, Lindsten T, et al. BCL-2, BCL-X(L) sequester BH3 domain-only molecules preventing BAX- and BAK-mediated mitochondrial apoptosis. Mol Cell 2001; 8:705–11.
Boatright KM, Renatus M, Scott FL, Sperandio S, Shin H, Pedersen IM, et al. A unified model for apical caspase activation. Mol Cell 2003; 11:529–41.
Philchenkov A. Caspases: potential targets for regulating cell death. J Cell Mol Med 2004; 8:432–44.
Johnson DE. Noncaspase proteases in apoptosis. Leukemia 2000; 14:1695–703.
French LE, Tschopp J. Protein-based therapeutic approaches targeting death receptors. Cell Death Differ 2003; 10:117–23.
Ferri KF, Kroemer G. Organelle-specific initiation of cell death pathways. Nat Cell Biol 2001; 3:E255–63.
Cory S, Huang DC, Adams JM. The Bcl-2 family: roles in cell survival and oncogenesis. Oncogene 2003; 22:8590–607.
Chang DW, Ditsworth D, Liu H, Srinivasula SM, Alnemri ES, Yang X. Oligomerization is a general mechanism for the activation of apoptosis initiator and inflammatory procaspases. J Biol Chem 2003; 278:16466–9.
Guicciardi ME, Leist M, Gores GJ. Lysosomes in cell death. Oncogene 2004; 23:2881–90.
Liston P, Fong WG, Korneluk RG. The inhibitors of apoptosis: there is more to life than Bcl2. Oncogene 2003; 22:8568–80.
Vaux DL, Silke J. IAPs, RINGs and ubiquitylation. Nat Rev Mol Cell Biol 2005; 6:287–97.
Cheng EH, Sheiko TV, Fisher JK, Craigen WJ, Korsmeyer SJ. VDAC2 inhibits BAK activation and mitochondrial apoptosis. Science 2003; 301:513–7.
Pinton P, Ferrari D, Rapizzi E, Di Virgilio F, Pozzan T, Rizzuto R. The Ca2+ concentration of the endoplasmic reticulum is a key determinant of ceramide-induced apoptosis: significance for the molecular mechanism of Bcl-2 action. EMBO J 2001; 20:2690–701.
Kagan VE, Gleiss B, Tyurina YY, Tyurin VA, Elenstrom-Magnusson C, Liu SX, et al. A role for oxidative stress in apoptosis: oxidation and externalization of phospatidylserine is required for macrophage clearance of cell under going Fas-mediated apoptosis. J Immunol 2002; 169:487–99.
Lauber K, Blumenthal SG, Waibel M, Wesselborg S. Clearance of apoptotic cells: getting rid of the corpses. Mol Cell 2004; 14:277–87.
Wu Y, Singh S, Georgescu MM, Brige RB. A role for Mer tyrosine kinase in alphavbeta5 integrin-mediated phagocytosis of apoptotic cell. J Cell Sci 2005; 118:539–53.
Fadeel B. Programmed cell clearance. Cell Mol Life 2003; 60:2575–85.
Huynh ML, Fadok VA, Henson PM. Phosphatidylserine-dependent ingestion of apoptotic cells promotes TGF-beta1 secretion and the resolution of inflammation. J Clin Invest 2002; 109:41–50.
Krammer PH. CD 95's deadly mission in the immune system. Nature 2000; 407:789–95.
Kishimoto H, Sprent J. The thymus and central tolerance. Clin Immunol 2000; 95:S3–7.
Drakesmith H, Chain B, Beverley P. How can dendritic cells cause autoimmune disease? Immunol Today 2000; 21:214–7.
Merry DE, Korsmeyer SJ. Bcl-2 gene family in the nervous system. Annu Rev Neurosci 1997; 20:245–67.
Fadeel B, Orrenius S. Apoptosis: a basic biological phenomenon with wide ranging implication in human disease. J Intern Med 2005; 258:479–517.
Martinon F, Tschopp J. Inflammatory caspases: linking an intracellular innate immune system to autoinflammatory diseases. Cell 2004; 117:561–74.
Ren Y, Tang J, Mok MY, Chan AW, Wu A, Lau CS. Increased apoptotic neutrophils and macrophages and impaired macrophage phagocytic clearance of apoptotic neutrophils in systemic lupus erythematosus. Arthritis Rheum 2003; 48:2888–97.
Mackay F, Browning JL. BAFF: a fundamental survival factor for B cells. Nat Rev Immunol 2002; 2:465–75.
Gregory CD, Devitt A. The macrophage and the apoptotic cell: an innate immune interaction viewed simplistically? Immunology 2004; 113:1–14.
Vandivier RW, Fadok VA, Hoffmann PR, Bratton DL, Penvari C, Brown KK, et al. Elastase-mediated phosphatidylserine receptor cleavage impairs apoptotic cell clearance in cystic fibrosis and bronchiectasis. J Clin Invest 2002; 109:661–70.
Henriksen PA, Devitt A, Kotelevtsev Y, Sallenave JM. Gene delivery of the elastase inhibitor elafin protects macrophages from neutrophil elastase-mediated impairment of apoptotic cell recognition. FEBS Lett 2004; 574:80–4.
Orr HT, Zoghbi HY. Reversing neurodegeneration: a promise unfolds. Cell 2000; 101:1–4.
Sanchez I, Xu CJ, Juo P, Kakizaka A, Blenis J, Yuan J. Caspase-8 is required for cell death induced by expanded polyglutamine repeats. Neuron 1999; 22:623–33.
Lambrechts D, Storkebaum E, Morimoto M, Del-Favero J, Desmet F, Marklund SL, et al. VEGF is a modifier of amyotrophic lateral sclerosis in mice and humans and protects motoneurons against ischemic death. Nat Genet 2003; 34:383–94.
Gill C, Mestril R, Samail A. Losing heart: the role of apoptosis in heart disease—a novel therapeutic target? FASEB J 2002; 16:135–46.
Fulda S, Debatin KM. Targeting apoptosis pathways in cancer therapy. Curr Cancer Drug Target 2004; 4:569–76.
Samuels-Lev Y, O'Connor DJ, Bergamaschi D, Triqiante G, Hsieh JK, Zhong S, et al. ASPP protein specifically stimulates the apoptotic function of p53. Mol Cell 2001; 8:781–94.
Reed JC. Apoptosis-based therapies. Nat Rev Drug Discov 2002; 1:111–21.
Fadeel B. Cell clearance and cancer. In: Sluyser M, ed. Applications of apoptosis to Cancer Treatment. The Netherland: Springer, 2005: 51–84.
de la Taille A, Chen MW, Burchardt M, Chopin DK, Buttyan R. Apoptotic conversion: evidence for exchange of genetic information between prostate cancer cells mediated by apoptosis. Cancer Res 1999; 59:5461–3.
Fischer U, Schulze-Osthoff K. Apoptosis-based therapies and drug targets. Cell Death Differ 2005; 12: Suppl. 1:942–61.
Sandborn WJ, Hanauer S, Loftus EV jr, Tremaine WJ, Kane S, Cohen R, et al. An open-label study of the human anti-TNF monoclonal antibody adalimumab in subjects with prior loss of response or intolerance to infliximab for Crohn's disease. Am J Gastroenterol 2004; 99:1984–89.
Daniel PT, Wieder T, Sturm I, Schulze-Osthoff K. The kiss of death: promises and failures of death receptors and ligands in cancer therapy. Leukemia 2001; 15:1022–32.
Nicholson DW. From bench to clinic with apoptosis-based therapeutic agents. Nature 2000; 407:810–16.
Reed JC, Pellecchia M. Apoptosis-based therapies for hematological malignancies. Blood 2005; 106:408–18.
Zangemeister-Wittke U, Leech SH, Olie RA, Simoes-Wust AP, Gautschi, Luedke GH, et al. A novel bispecific antisense oligonucleotide inhibiting both bcl-2 and bcl-xL expression efficiently induces apoptosis in tumor cells. Clin Cancer Res 2000; 6:2547–55.
Sasaki H, Sheng Y, Kotsuji F, Tsang BK. Down-regulation of X-linked inhibitor of apoptosis protein induces apoptosis in chemoresistant human ovarian cancer cells. Cancer Res 2000; 60:5659–66.
Tamm I, Trepel M, Cardo-Vila M, Sun Y, Welsh K, Cabezas E, et al. Peptides targeting caspase inhibitors. J Biol Chem 2003; 278:14401–5.
Hoglen NC, Chen LS, Fisher CD, Hirakawa BP, Groessi T, Contreras PC. Characterization of IDN-6556 (3-[2-(2-tert-butyl-phenylaminooxalyl)-amino]-propionylamino]-4-oxo-5-(2,3,5,6-tetrafluoro-phenoxy)-pentanoic acid): a livertargeted caspase inhibitor. J Pharmacol Exp Ther 2004; 309:634–40.
Yang W, Guastella J, Huang JC, Wang Y, Zhang L, Xue D, et al. MX 1013, a dipeptide caspase inhibitor with potent in vivo antiapoptotic activity. Br J Pharmacol 2003; 140:402–12.
Linton SD, Aja T, Allegrini PR, Deckwerth TL, Diaz JL, Hengerer B, et al. Oxamyl dipeptide caspase inhibitors developed for the treatment of stroke. Bioorg Med Chem Lett. 2004; 14:2685–91.
Philchenkov A, Zavelevich M, Kroczak TJ, Los M. Caspases and cancer: mechanisms of inactivation and new treatment modalities. Exp Oncol 2004; 26:82–97.
Xie X, Zhao X, Liu Y, Zhang J, Matusik RJ, Slawin KM, et al. Adenovirus-mediated tissue-targeted expression of a caspase-9-based artificial death switch for the treatment of prostate cancer. Cancer Res 2001; 61:6795–804.
Komata T, Kondo Y, Kanzawa T, Hirohata S, Koga S, Sumiyoshi H, et al. Treatment of malignant glioma cells with the transfer of constitutively active caspase-6 using the human telomerase catalytic subunit (human telomerase reverse transcriptase) gene promoter. Cancer Res 2001; 61:5796–802.
Hu Y, Cherton-Horvat G, Dragowska V, Baird S, Korneluk RG, Durkin JP, et al. Antisense oligonucleotides targeting XIAP induce apoptosis and enhance chemotherapeutic activity against human lung cancer cells in vitro and in vivo. Clin. Cancer Res. 2003; 9:2826–36.
Stoll R, Renner C, Hansen S, Palme S, Klein C, Belling A, et al. Chalcone derivatives antagonize interactions between the human oncoprotein MDM2 and p53. Biochemistry. 2001; 40:336–44.
Butz K, Denk C, Ullmann A, Scheffner M, Hoppe-Seyler F. Induction of apoptosis in human papillomavirus positive cancer cells by peptide aptamers targeting the viral E6 oncoprotein. Proc Natl Acad Sci U S A 2000; 97:6693–7.
Green DR, Kroemer G. The pathophysiology of mitochondrial cell death. Science 2004; 305:626–9.
Breckenridge DG, Xue D. Regulation of mitochondrial membrane permeabilization by BCL-2 family proteins and caspases. Curr Opin Cell Biol. 2004; 16:647–52.
Armstrong JS, Jones DP. Glutathione depletion enforces the mitochondrial permeability transition and causes cell death in Bcl-2 overexpressing HL60 cells. FASEB J. 2002; 16:1263–65.
Armstrong JS. Mitochondria: a target for cancer therapy. Br J Pharmacol 2006; 147:239–48.
Marcucci G, Stock W, Dai G, Klisovic RB, Liu S, Klisovic MI, et al. Phase I study of oblimersen sodium, an antisense to Bcl-2, in untreated older patients with acute myeloid leukemia: pharmacokinetics, pharmacodynamics, and clinical activity. J Clin Oncol 2005; 23:3404–11.
Armstrong JS, Steinauer KK, Hornung B, Irish JM, Lecane P, Birrell GW, et al. Role of glutathione depletion and reactive oxygen species generation in apoptotic signaling in a human B lymphoma cell line. Cell Death Diffe. 2002; 9:252–63.
Costantini P, Jacotot E, Decaudin D, Kroemer G. Mitochondrion as a novel target of anticancer chemotherapy. J Natl Cancer Inst 2000; 92:1042–53.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singh, N. Apoptosis in health and disease and modulation of apoptosis for therapy: An overview. Indian J Clin Biochem 22, 6–16 (2007). https://doi.org/10.1007/BF02913307
Issue Date:
DOI: https://doi.org/10.1007/BF02913307