Summary
The sequence of antigen localization and the interaction of immune deposits with the anionic sites of the glomerular basement membrane (GBM) were investigated in an active model of in situ immune complex glomerulonephritis using a cationized ferritin.
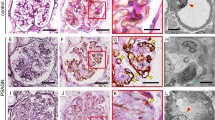
Three weeks after immunization with native horse spleen ferritin, the left kidneys of rats were perfused with 500 μg of cationized ferritin through the left renal artery. One h after renal perfusion, most of ferritin particles localized subendothelially, corresponding to the anionic sites of the lamina rara interna. In the glomerular capillary loops, infiltrating polymorphonuclear leukocytes and monocytes were seen. Some of these monocytes were in direct contact with immune complexes containing ferritin aggregates associated with anionic sites of the lamina rara interna. At 24 h, numerous ferritin aggregates were present subepithelially, preferentially beneath the slit membrane. The subepithelial location of ferritin did not always correspond to the anionic sites of the lamina rara externa.
From days 3 to 7, there was remarkable endocapillary cell proliferation in some loops and pronounced effacement of epithelial foot processes. Focal detachment of epithelium from the GBM was observed occasionally. From days 14 to 28, most of ferritin aggregates were located intramembranously and subepithelially. Membranous transformation has already begun around the subepithelial deposits. This morphological study provides insight into the fate of immune deposits and injury to the GBM in the glomerulonephritis.
Similar content being viewed by others
References
Batsford SR, Takamiya H, Vogt A (1980a) A model of in situ immune complex glomerulonephritis in the rat employing cationized ferritin. Clin Nephrol 14:211–216
Batsford SR, Oite T, Takamiya H, Vogt A (1980b) Anionic binding sites in the glomerular basement membrane: Possible role on the pathogenesis of immune complex glomerulonephritis. Renal Physiol 3:336–340
Batsford SR, Sasaki M, Takamiya H, Vogt A (1983) Cationic macromolecule-induced nephrotic syndrome in rabbits. Lack of immune complex involvement. Lab Invest 49:260–269
Couser WG, Steinmuller DR, Stilmant MM, Salant DJ, Lowenstein LM (1978) Experimental glomerulonephritis in the isolated perfused rat kidney. J Clin Invest 62:1275–1287
Davies M, Coles GA, Hughes KT (1980) Glomerular basement membrane injury by neutrophil and monocyte neutral proteinases. Renal Physiol 3:106–111
Ehrenreich T, Churg J (1968) Pathology of membranous nephropathy. In: Sommers SC (ed) Pathology annual. Appleton-Century-Crofts, New York, vol 3, p 145
Kurtz SM, Feldman JD (1962) Experimental studies on the formation of the glomerular basement membrane. J Ultrastruct Res 6:19–27
Morita T, Kihara I, Oite T, Yamamoto T, Suzuki Y (1978) Mesangiolysis. Sequential ultrastructural study of Habu venom-induced glomerular lesions. Lab Invest 38:94–102
Oite T, Batsford SR, Mihatsch MJ, Takamiya H, Vogt A (1982) Quantitative studies of in situ immune complex glomerulonephritis in the rat induced by planted, cationized antigen. J Exp Med 155:460–474
Oite T, Shimizu F, Kihara I, Batsford SR, Vogt A (1983) An active model of immune complex glomerulonephritis in the rat employing cationized antigen. Am J Pathol 112:185–194
Schurer JW, Hoedemaeker Ph J, Molenaar I (1977) Polyethyleneimine as tracer particle for (immuno)electronmicroscopy. J Histochem Cytochem 25:384–387
Schurer JW, Kalicharan D, Hoedemaeker Ph J (1978) Demonstration of anionic sites in the basement membrane and in collagen fibrils. J Histochem Cytochem 26:688–689
Striker GE, Smuckler EA (1970) An ultrastructural study of glomerular basement membrane synthesis. Am J Pathol 58:531–539
Suzuki Y, Maezawa A, Matsui K, Oite T, Koda Y, Arakawa M (1983) Alteration of glomerular anionic sites by the development of subepithelial deposits in experimental glomerulonephritis in the rat. Virchows Arch [Cell Pathol] 44:209–222
Törnroth T (1976) The fate of subepithelial deposits in acute poststreptococcal glomerulonephritis. Lab Invest 35:461–474
Törnroth T, Skrifvars B (1975) The development and resolution of glomerular basement membrane changes associated with subepithelial immune deposits. Am J Pathol 79:219–230
Unanue ER (1976) Secretory function of mononuclear phagocytes. Am J Pathol 83:396–417
Van Damme BJC, Fleuren GJ, Bakker WW, Vernier RL, Hoedemaeker Ph J (1978) Experimental glomerulonephritis in the rat induced by antibodies directed against tubular antigens. V. Fixed glomerular antigens in the pathogenesis of heterologous immune complex glomerulonephritis. Lab Invest 38:502–510
Vogt A, Bockhorn H, Kozima K, Sasaki S (1968) Electron microscopic localization of nephrotoxic antibody in the glomerulus of purified nephritogenic antibody-ferritin conjugates. J Exp Med 127:867–878
Vogt A, Schmidt U, Takamiya H, Batsford SR (1980) In situ immune complex nephritis and basic proteins. In: Proceedings of European Dialysis and Transplantation Association. Pitman Medical Publishing Co Ltd., Tumbridge Wells, Kent 17:613–620
Vogt A, Rohrbach R, Shimizu F, Takamiya H, Batsford S (1982) Interaction of cationized antigen with rat glomerular basement membrane: In situ immune complex formation. Kidney Int 22:27–35
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Oite, T., Shimizu, F., Suzuki, Y. et al. Ultramicroscopic localization of cationized antigen in the glomerular basement membrane in the course of active, in situ immune complex glomerulonephritis. Virchows Archiv B Cell Pathol 48, 107–118 (1985). https://doi.org/10.1007/BF02890120
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02890120