Abstract
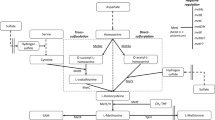
The strainCandida utilis T 20 adapted to a high concentration of ethionine, excretes considerable amounts of methionine in a synthetic medium, about 40 times as much as the original non-adapted strain. At the same time, the amount of methionine in yeast cells incrncreased, predominantly in the pool (9 times as much as in the control). This ability to produce greater amounts of methionine in the pool or to excrete it into the medium is not permanent, since after 5 passages on agar with ut ethionine the amount of methionine was practically not increased as compared with the original non-adapted strain.
An increase in free methionine and of methionine excreted into the medium was found on cultivating the strain in a molasses-containing medium, too.
Abstract
Штамм Candida utilis T 20, адаптированный к высокой концетраации зтонина, выделяет в куль тивационную синтетическую среду значительное количество метионина,—приблизительно в 40 раз больще, чем первоначальный неадаптированный штамн. Одновременно повышается и содержание метионина в дрожжевых клетках, преимущественно в «pool» (в 9 раз больше, чем в контроле). Эта способность образовать большие количества метионина в «pool» или выделять его в среду оказывается непостоянной, так как после 5 пассажей на среде с агаром без этионина количество метионина практически не повышалось по сравнению с первоначальным неадаптированным штаммом.
Повышение содержания свободного метионина и метионина, выделяемого в среду, наблюдалось и при культивации адаптированного штамма на среде с мелассой.
Similar content being viewed by others
References
Adelberg, E. A.:Selection of bacterial mutants which excrete antagonists of antimetabolites. J. Bacteriol. 76: 326, 1958.
Apple, M. A., Brandley, S. G.:Effect of 6-mercaptopurine on induction of α-glucosidase in Candida. Proc. Soc. exp. Biol. & Med. 112: 139, 1963.
Cestari, A., Dessi, P.:Action of ethionine on the bacterial growth. Giorn. microbiol. 5: 39, 1958. C. A. 54: 4769a, 1960.
Dyer, Helen M.:Evidence of the physiological specifity of methionine in regard to the methylthiol group: the synthesis of S-ethylhomocysteine (ethionin) and a study of its availability for growth. J. biol. Chem. 124: 519, 1938.
Halvorson, H. O., Spiegelman, S.:The inhibition of enzyme formation by amido-acid analogues. J. Bacteriol. 64: 207, 1952.
Harrison, E., Lees, K. A., Wood, F.:The assay of vitamin B 12.Part VI. Microbiological estimation with mutant of Escherichia coli by the plate method. Analyst 76: 696, 1951.
Johansonn, K. R.:Response to and assay of vitamin B 12 by a mutant of Escherichia coli. Proc. Soc. exp. Biol. & Med. 83: 448, 1953.
Maw, G. A.:S-methylcysteine and S-ethylcysteine in the sulfur metabolism of yeast. Biochem. J. 80: 28P, 1961a.
Maw, G. A.:Ability of S-methyl-l-cysteine to annual the inhibition of yeast growth by l-ethionine and S-ethyl-l-cysteine. J. gen. Microbiol. 28: 441, 1961b.
Munier, R., Cohen, G. N.:Incorporation d'analogues structuraux d'amino-acides dans les protéines bactériennes au cours de leur synthèse in vivo. Biochem. biophys. Acta 31: 378, 1959.
Oakberg, E. F., Luria, S. E.:Mutation to sulfonamide resistance in Staphylococcus aureus. Genetics 32: 249, 1947.
Parks, L. W.:S-adenosylethionine and ethionine inhibition. J. biol. Chem. 232: 169, 1958.
Petrov, D. F., Grableva, T. I.:Microbiological quantitative determination of methionine. USSR pat 134, 394, 1960. C.A. 55: 14178h, 1961.
Richmond, M. H.:The effect of amino acid analogues on growth and protein synthesis in microorganisms. Bact. Rev. 26: 398, 1962.
Rowbury, R. J., Woods, D. D.:Repression of methionine synthesis in Escherichia coli. J. gen. Microbiol. 22: 129, 1961.
Sato, M., Uemura, T.:Halophilic character of shoyu yeast. IV. Effects of inhibitors on the halophilic behavior of the yeast. 1 Nippon Nôgei Kagaku Kaishi, 33: 13, 1959. C.A. 55: 2367a, 1961.
Scherr, G. H., Rafelson, M. E.:The direct isolation of mutants producing increased amounts of metabolites. J. appl. Bact. 25: 187, 1962.
Wolley, D. W.:A study of antimetabolites. J. Wiley & Sons, London, 1952.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Musílková, M., Fencl, Z. Biosynthesis of methionine in an ethionine-resistant strain ofCandida utilis . Folia Microbiol 9, 374–379 (1964). https://doi.org/10.1007/BF02874189
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02874189