Abstract
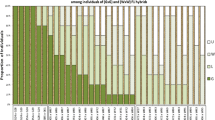
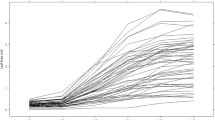
Low tendency to tuber greening under light exposure is a desirable trait for marketing and processing potatoes. Knowledge of the genetic basis of this trait is limited. During two consecutive years tuber greening after 2-week light exposure was evaluated on 17 unselected diploid potato families obtained between parents with varying tuber-greening tendencies. Parents of these families and standard cultivars were evaluated at the same time. External and depth of tuber greening were examined after tubers were exposed to 2 weeks of light in the greenhouse followed by storage at 10 C, 4 C, and after reconditioning at 18 C. Families, storage conditions, and years significantly influenced tuber greening. Distribution of greening indicated it would be possible to select individuals with very low tuber-greening tendency. Generally, external tuber greening was more intense than depth of greening. External tuber greening increased during storage while depth of tuber greening decreased. A significant maternal effect on external tuber greening was found in one of the three sets of reciprocal crosses after storage at 4 C. Both general and specific combining abilities were important in the inheritance of tuber greening. The correlations between external and depth of tuber greening in most families were significant and relatively high. The results indicated that genotypes with low tendency to tuber greening can be selected in diploid families.
Resumen
Un carácter deseable de la papa para comercialización y procesamiento, es su baja tendencia al verdeo de los tubérculos por exposición a la luz. El conocimiento sobre la base genética de esta característica es limitado. El verdeo fue evaluado durante dos años consecutivos en 17 familias de papa diploide no seleccionada, obtenida de progenitores con diferentes tendencias. Se evaluaron al mismo tiempo los progenitores de estas familias y los cultivares estándar. El verdeo externo y profundidad en el tubérculo fue examinado después de que los tubérculos fueron expuestos a dos semanas de luz en el invernadero y luego almacenados a 10 C, 4 C y después que se reacondicionaron a 18 C. Los factores como familias, condiciones de almacenamiento y años influenciaron significativamente el verdeo de los tubérculos. La distribución del verdeo indicó que sería posible seleccionar individuos con tendencia mínima a esta característica. Generalmente el verdeo externo del tubérculo fue más intenso que la profundidad. El verdeo superficial aumentó con el almacenamiento, mientras que la profundidad disminuyó. En uno de los tres grupos de cruzamientos recíprocos se encontró un efecto significativo del progenitor femenino sobre el verdeo externo del tubérculo después del almacenamiento a 4 C. Tanto las aptitudes de combinación general como específica fueron importantes en la herencia del verdeo de los tubérculos. La correlación entre el verdeo externo y profundidad en el tubérculo, en la mayoría de las familias fue significativa y relativamente alta. Los resultados indican que los genotipos con baja tendencia al verdeo del tubérculo pueden ser seleccionados en familias diploides.
Similar content being viewed by others
Literature cited
Akeley RV, GVC Houghland and AE Schark. 1962. Genetic differences in potato-tuber greening. Am Potato J 39:409–417.
Brown E and W Riley. 1976. Greening of potato tubers: varietal response to controlled exposure to light. J Nat Inst Agric Bot 14:70–76.
Brune S and PE Melo. 2001. A quick method to assess greening in potato tubers. Pesq Agropec Bras 36/5:809–814.
Buck RW Jr. and RV Akeley. 1967. Effect of maturity, storage temperature, and storage time on greening of potato tubers. Am Potato J 44:56–58.
Caliński T, S Czajka, Z Kaczmarek, P Krajewski and L Siatkowski. 1998. SERGEN 3. Analiza serii doświadczeńodmianowych i genetyczno-hodowlanych. Instytut Genetyki Roślin PAN, Poznań, Polska. pp 1–58.
Dale MFB, DW Griffiths and H Bain. 1992. Glycoalkaloids in potatoes — shedding light on an important problem. Aspects Appl Biol 33:221–227.
De Maine MJ, H Bain and JL Joyce. 1988. Changes in the total tuber glycoalkaloid content of potato cultivars on exposure to light. J Agric Sci Camb 111:57–58.
Edwards EJ, RE Saint and AH Cobb. 1998. Is there a link between greening and light-enhanced glycoalkaloid accumulation in potato (Solanum tuberosum L.) tubers? J Sci Food Agric 76:327–333.
Griffiths DW, MFB Dale and H Bain. 1994. The effect of cultivar, maturity and storage on photo-induced changes in the total glycoalkaloid and chlorophyll contents of potatoes (Solanum tuberosum). Plant Sci 98:103–109.
Griffiths DW, H Bain and MFB Dale. 1998. Effect of storage temperature on potato (Solanum tuberosum L.) tuber glycoalkaloid content and the subsequent accumulation of glycoalkaloids and chlorophyll in response to light exposure. J Agric Food Chem 46:5262–5268.
Harkett PJ. 1975. The influence of temperature and skin colour on chlorophyll synthesis in potato tubers exposed to light. 6th Triennial Conference of EAPR, 15–19 September 1975, Wageningen, The Netherlands. pp 179–180.
Haynes KG, RW Goth, SB Sterrett, BJ Christ, DE Halseth, GA Porter, MR Henniger, DR Wilson, RE Webb, DF Hammond, R Moore, FL Haynes, S Arrendell, MJ Wannamaker and SL Sinden. 1992. Coastal Chip: a chipping potato variety resistant to heat stress. Am Potato J 69:515–523.
Holm DG, JC Miller and DG Smallwood. 1992. Russet Nugget: a fresh market and processing potato cultivar with resistance to common scab. Am Potato J 69:331–336.
Jakuczun H. 1993. Zróżnicowanie skłonności do zielenienia bulw u ziemniaków diploidalnych. Biuletyn Instytutu Ziemniaka 43:11–15.
Jakuczun H, E Zimnoch-Guzowska. 2004. Inheritance of glucose content in tubers of diploid potato families. Amer J Potato Res 81:359–370.
Kala R, H Chudzik, A Dobek and H Kiełczewska. 1996. System Analiz Statystycznych Doświadczeń Genetyczno-Hodowlanych, wersja 2.0. Katedra Metod Matematycznych i Statystycznych, Akademia Rolnicza w Poznaniu, Polska.
Lewis WC and RG Rowberry. 1973. Some effects of planting depth and time and height of hilling on Kennebec and Sebago potatoes. Am Potato J 50:301–310.
Liljemark A and E Widoff. 1960. Greening and solanine development of white potato in fluorescent light. Am Potato J 37:379–389.
Łomnicki A. 1995. Wprowadzenie do statystyki dla przyrodników. Wydawnictwo Naukowe PWN, W-wa 1995. pp 245.
Parfitt DE and SJ Peloquin. 1981. The genetic basis for tuber greening in 24-chromosome potatoes. Am Potato J 58:299–304.
Percival GC. 1999. The influence of light upon glycoalkaloid and chlorophyll accumulation in potato tubers (Solanum tuberosum L.). Plant Sci 145:99–107.
Pyke KA 1999. Plastid division and development. Plant Cell 11:549–556.
Reeves AF. 1988. Varietal differences in potato tuber greening. Am Potato J 65:651–658.
Reeves AF, RV Akeley, HJ Murphy, GA Porter, RH Cole, J Watts, BJ Christ, FE Manzer and RH True. 1990. Somerset: a new chipping potato variety. Am Potato J 67:153–161.
Reeves AF, GA Porter, CE Cunningham, RJ Nickeson, FE Manzer, TM Work, AA Davis and ES Plissey. 1994a. Prestile: a new round white potato variety. Am Potato J 71:89–97.
Reeves AF, GA Porter, FE Manzer, TM Work, AA Davis and ES Plissey. 1994b. MaineChip: a new chipping potato variety for cool storage processing. Am Potato J 71:237–247.
Reeves AF, GA Porter, CE Cunningham, RJ Nickeson, FE Manzer, TM Work, AA Davis and ES Plissey. 1995. Portage: a new early-maturing, round white table potato variety. Am Potato J 72:681–688.
Reeves AF, GA Porter, FE Manzer, TM Work, AA Davis, DR Hensel and JR Shumaker 1996. St. Johns: a round white potato variety for fresh market. Am Potato J 73:89–98.
Reeves AF, GA Porter, TM Work, DH Lambert, AA Davis and ES Plissey. 1997. Mainestay: a high-yielding, round white potato variety for fresh market. Am Potato J 74:255–263.
Thomas WW. 1980. Development of nongreen plastids. Ann Rev Plant Physiol 31:375–394.
Vothknecht UC and P Westhoff. 2001. Biogenesis and origin of tylakoid membranes. Biochim Biophys 1541:91–101.
Yamaguchi M, DL Hughes and FD Howard. 1960. Effect of color and intensity of fluorescent lights and application of chemicals and waxes on chlorophyll development of White Rose potatoes. Am Potato J 37:229–236.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jakuczun, H., Zimnoch-Guzowska, E. Inheritance of tuber greening under light exposure in diploid potatoes. Am. J. Pot Res 83, 211–221 (2006). https://doi.org/10.1007/BF02872157
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02872157