Abstract
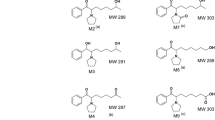
Mechanism of the monoamine oxidase inhibition by tranylcypromine was studied in relation to its metabolism to reactive species. A metabolic study performed to collect general biotransformation pathway in rats provided GC/MS evidence for the detection of two new metabolites, N-acetyl and hydroxylated N-acetyltranylcypromine.
Similar content being viewed by others
Literature Cited
Kang, G. I.: Mechanism of the Monoamine Oxidase Inhibition.Yakhak Hoeji 27, 321 (1983).
Paech, C. P., Salach, J. I., and Singer, T. P.: Suicide Inactivation of Monoamine Oxidase by trans-Phenylcyclopropylamine.J. Biol. Chem. 255, 2700 (1980).
Fuller, R.W., Hemrick-Luecke, S.K., Molloy, B.B.: N-[2-(o-iodophenoxy) ethyl] cyclopropylamine hydrochloride (LY 121768), A potent and selective irresversible inhibitor of type A monoa-mine oxidase.Biochem. Pharmacol. 32, 1243 (1983).
Neal, R. A.: Chemically Reactive Metabolites as Suicide Enzyme Inhibitors.Drug Metab. Rev. 14, 49 (1983).
Author information
Authors and Affiliations
Additional information
Mechanism of the Monoamine Oxidase Inhibition I
Rights and permissions
About this article
Cite this article
Kang, G.I., Chung, S.Y. Identfiication of N-acetyl and hydroxylated N-acetyltranylcypromine from tranylcypromine-dosed rat urine. Arch. Pharm. Res. 7, 65–68 (1984). https://doi.org/10.1007/BF02856926
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02856926