Abstract
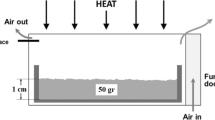
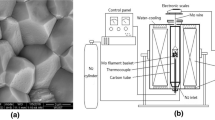
The thermal behavior and kinetic parameters of decomposition reaction of limestone in a temperature-programmed mode were investigated by means of TG. The experimental results show that the kinetic model functions in different forms for the thermal decomposition reactions of different limestone grain sizes in dispersing state under the atmosphere of static air are 4(1−α)3/4 for small size limestone and (1−α) for large size limestone. Information was obtained on the relationship among the decomposition temperature, decomposition time, decomposition fraction, decomposition reaction rate constant and grain size of limestone.
Similar content being viewed by others
References
Vosteen B. Vorwarmung and Vollkommene Caalcinations Von Zementrohmehl in Einem Schwebegassystem.Zemen-Kalk-Gips, 1974, 9: 443
Müller A, Dabm B, Stark J, Zur Kinetik der. Calciumcarbonatzerstetzung. Muller A, Dabm B, J Stork.Silikattechnik, 1977, 28: 4
Müller A, Dabm B, Weimar J S. Contribution to the Kinetics of Calcium Carbonate Dissociation.Zement-Kalk-Gips 1979, 2: 78
Hu R Z, Shi Q Z.Thermal Analysis Kinetics. Beijing: Science Press, 2001
Hu R Z, Yang Z, Liang Y J. The Determination of the Most Probable Mechanism Function and Three Kinetic Parameters of Exothermic Decomposition Reaction of Energetic Materials by a Single Non-isothermal DSC Curre.Thermochim. Acta, 1998: 123–135
Author information
Authors and Affiliations
Additional information
Xu De-long: Born in 1952
Funded by the National Science Foundatin of China (No. 50272050)
Rights and permissions
About this article
Cite this article
De-long, X., Hong-en, W. & Yong-qin, L. The influence of grain size on decomposition reaction of limestone in dispersing state. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 19, 1–5 (2004). https://doi.org/10.1007/BF02841354
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02841354