Summary
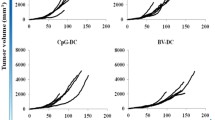
The specific anti-tumor immune response induced by mouse bone marrow dendritic cells (DCs) transfected with recombinant adenovirus carrying mutant k-ras genes was investigated. DCs were generated from mouse bone marrow in the presence of rmGM-CSF (3.3 ng/mL) and rmIL-4 (1.3 ng/mL) and detected by FACS, and then transfected with the recombinant adenovirus encoding mutant k-ras gene. The efficacy of transfection and T cell stimulating activity of DCs were detected. CTL activity of the mice vaccinated with DCs was observed. The results showed that DCs had dendritic veiled morphology. BmDCs highly expressed B7-1 (80%), B7-2 (77%), MHC II (70%), CD11c (65%), CD40 (70%) and CD54 (96%) with FACS, and no significant difference in the expression was observed before and after the transfection (P>0.05). The DCs transfected by mutant k-ras gene could significantly stimulate lymphocytes proliferation as compared with those transfected by Ad-c or non-modified DCs (P<0.05). DC vaccine transfected by mutant k-ras gene could induce CTL activity against Lewis lung cancer, but not against B16. The specific cytotoxicity against Lewis lung cancer in Ad-k-ras/12-transduced DC group was significantly higher than those in the control, vector and non-transfected DCs groups (P<0.05). It was concluded that special antitumor response could be induced by DCs transfected with recombinant adenovirus carrying mutant k-ras genes.
Similar content being viewed by others
References
Abrams S I, Stanziale S F, Lunin S Det al. Identification of overlapping epitopes in mutant ras oncogene peptides that activate CD4+ and CD8+ T cell responses. Eur J Immunol. 1996,26:435
Bergmann-Leitner E S, Kantor J A, Lesley Shupert Wet al. Identification of a human CD8+ T lymphocyte neo-epitope created by a ras codon 12 mutation which is restricted by the HLA-A2 allele. Celluar Immunology, 1998,187:103
Bos J L. ras oncogenes in human cancer: a review. Cancer Res, 1989,49:4682
Ramakrishna G, Sithanandam G, Cheng R Yet al. K-ras p21 expression and activity in lung and lung tumors. Exp Lung Res, 2000,26:659
Abrams S I, Dobrzanski M J, Wells D Tet al. Peptide-specific activation of cytolytic CD4 + T lymphocytes against tumor cells bearing mutated epitopes of K-ras p21. Eur J Immunol, 1995,25:2588
Banchereau J, Steinman R M. Dendritic cells and the control of immunity. Nature, 1998,392:245
Zhao F, Zhou Q H, Qing Yet al. Construction of mutant k-ras gene recombinant adenovirus. Chin J Lung Cancer (Chinese), 2002,5(1):14
Jackson J H, Cochrane C G, Bourne J Ret al. Farnesol modification of Kirsten-ras exon 4B protein is essential for transformation. Proc Natl Acad Sci U S A, 1990,87:3042
He T C, Zhou S, Costa L Tet al. A simplified system for generating recombinant adenoviruses. Proc Natl Acad Sci U S A, 1998,95:2509
Inaba K, Inaba M, Romani Net al. Generation of large number of dendritic cells from mouse bone marrow cultures supplement with GM-CSF. J Exp Med, 1992, 176(6):1693
Hunger R E, Brand C U, Streit Met al. Successful induction of immune responses against mutant ras in melanoma patients using intradermal injection of peptides and GM-CSF as adjuvant. Exp Dermatol, 2001,10(3): 161
Linard B, Bezieau S, Benlalam Het al. A ras-mutated peptide targeted by CTL infiltrating a human melanoma lesion. J Immunol, 2002, 168:4802
Zhong L, Granelli-Piperno A, Choi Yet al. Recombinant adenovirus is an efficient and nonperturbing genetic vector for human dendritic cells. Eur J Immunol, 1999,29:964
Author information
Authors and Affiliations
Additional information
Zhao Feng, male, born in 1971, Doctor in Charge, M. D., Ph.D.
This project was supported by a grant from National Sciences Foundation of China (No. 30000206).
Rights and permissions
About this article
Cite this article
Feng, Z., Qinghua, Z., Yanrong, L. et al. In vitro anti-tumor immune response induced by dendritic cells transfected with recombinant adenovirus carrying mutant k-ras genes. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 25, 378–381 (2005). https://doi.org/10.1007/BF02828201
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02828201