Abstract
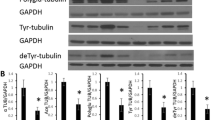
Breakdown or disruption of the cytoskeleton has been implicated in the neurodegenerative processes of a variety of diseases, including Alzheimer disease (AD) and stroke. Studies of such diseases in the human involve the use of postmortem brain tissue. Postmortem delay may vary considerably from a few hours to a few days, and within this period, a degree of cytoskeletal breakdown may occur. It is therefore crucial to examine alterations occurring in the cytoskeleton as a result of postmortem delay and subtract these from those caused by the disease. In this study, the distribution of τ, MAP2, and MAP5 immunohistochemistry was examined following postmortem intervals of 0–72 h in the rat cerebral cortex, corpus callosum, caudate nucleus, and hippocampus. Each microtubule-associated protein (MAP) underwent unique changes that were dependent both on postmortem interval and the brain region examined. Following long postmortem delays, some of the changes in these proteins were similar to those seen in rodent models of cerebral ischemia. These results demonstrate that MAPs are not stable during postmortem delay in the rat. Therefore, caution must be exercised when interpreting changes in MAPs in human postmortem tissue, especially in cases where ischemic injury may be involved. Examination of control tissue carefully matched for postmortem delay is therefore essential to allow meaningful interpretation of cytoskeletal abnormalities in human neurodegenerative disease.
Similar content being viewed by others
References
Abe H., Yagishita S., Amano N., Iwabuchi K., Hasegawa K., and Kowa K. (1992) Argyrophilic glial intracytoplasmic inclusions in multiple system atrophy: Immunocytochemical and ultrastructural study.Acta Neuropathol. 84, 273–277.
Adams J. H. and Graham D. (1989a) Vascular and hypoxic disorders, inAn Introduction to Neuropathology. Churchill Livingstone, pp. 57–82.
Adams J. H. and Graham D. (1989b) Trauma, inAn Introduction to Neuropathology. Churchill Livingstone, pp. 118–136.
Binder L. I., Frankfurter A., and Rebhun L. I. (1985) The distribution of tau in the mammalian central nervous system.J. Cell Biol. 101, 1371–1378.
Brion J-P., Couck A-M., Robertson J., Loviny T. L. F., and Anderton B. H. (1993) Neurofilament monoclonal antibodies RT97 and 8D8 recognize different modified epitopes in paired helical filament-τ in Alzheimer's disease.J. Neurochem. 60, 1372–1382.
Catala I., Ferrer I., Galofre E., and Fabregues I. (1988) Decreased numbers of dendritic spines on cortical pyramidal neurons in dementia. A quantitative golgi study on biopsy samples.Hum. Neurobiol. 6, 255–259.
Conner J. R. and Fine R. E. (1986) The distribution of transferrin immunoreactivity in the rat central nervous system.Brain Res. 368, 319–328.
Davis D. R., Brion J-P, Couck A-M., Gallo J-M., Hanger D., Ladhani K., Lewis C., Miller C. J., Rupniak T., Smith C., and Anderton B. H. (1995) The phosphorylation state of the microtubule-associated protein tau as affected by glutamate, colchicine and β-amyloid in primary rat cortical neuronal cultures.Biochem. J. 309, 941–949.
Dawson D. A. and Hallenbeck J. M. (1996) Acute focal ischemia-induced alterations in MAP2 immunostaining: Description of temporal changes and utilization as a marker for volumetric assessment of acute brain injury.J. Cereb. Blood Flow Metab. 16, 170–174.
Dewar D. and Dawson D. (1995a) Tau protein is altered by focal cerebral ischaemia in the rat: an immunohistochemical and immunoblotting study.Brain Res. 684, 70–78.
Dewar D. and Dawson D. (1995b) Microtubule-associated proteins in focal cerebral ischaemia.J. Neurotrauma 12, 455.
Fischer I., Romano C. G., and Grynspan F. (1991) Calpain-mediated proteolysis of microtubule associated proteins MAP1B and MAP2 in developing brain.Neurochem. Res. 16, 891–898.
Geddes J. W., Chang-Chui H., Cooper S. M., Lott I. T., and Cotman C. W. (1986) Density and distribution of NMDA receptors in the human hippocampus in Alzheimer's disease.Brain Res. 399, 156–161.
Geddes J. W., Lundgren K., and Kim Y. K. (1991) Abberant localization of MAP5 immunoreactivity in the hippocampal formation in Alzheimer's disease.J. Neurosci. Res. 30, 183–191.
Geddes J. W., Schwab C., Craddock S., Wilson J. L., and Creed Petigrew L. (1994) Alterations in τ immunostaining in the rat hippocampus following transient cerebral ischemia.J. Cereb. Blood Flow Metab. 14, 554–564.
Hanks S. D. and Flood D. G. (1991) Region-specific stability of dendritic extent in normal human aging and regression in Alzheimer's disease. I. CA1 of hippocampus.Brain Res. 540, 63–82.
Hasegawa M., Arai T., and Ihara Y. (1990) Immunochemical evidence that fragments of phosphorylated MAP5 (MAP1B) are bound to neurofibrillary tangles in Alzheimer's disease.Neuron 4, 909–918.
Huber G. and Matus A. (1984) Differences in the cellular distributions of two microtubule associated proteins, MAP1 and MAP2, in rat brain.J. Neurosci. 4, 151–160.
Irving E. A., McCulloch J., and Dewar D. (1996) Intracortical perfusion of glutamate in vivo induces alterations of tau and MAP2 immunoreactivity in the rat.Acta Neuropath. 92, 182–196.
Irving E. A., Barnett S. C., Dickinson P., McCulloch J., Dewar D., and Griffiths I. R. (1995) Increased tau immunoreactivity within oligodendrocytes following cytotoxic injury.Soc. Neurosci. Abstract 134, 9.
Iwatsubo T., Hasegawa M., and Ihara Y. (1994) Neuronal and glial tau-positive inclusions in diverse neurologic diseases share common phosphorylation characteristics.Acta Neuropathol. 88, 129–136.
Johnson G. V. W., Jope R. S., and Binder L. I. (1989) Proteolysis of tau by calpain.Biochem. Biophys. Res. Commun. 163, 1505–1511.
Johnson G. V. W., Litersky J. M., and Jope R. S. (1991) Degradation of microtubule-associated protein 2 and brain spectrin by calpain: comparative study.J. Neurochem. 56, 1630–1638.
Kato S., Nakamura H., Hirano A., Ito H., Llena J. F., and Yen S-H. (1991) Agryrophilic ubiquinated cytoplasmic inclusions of Leu-7-positive glial cells in olivopontocerebellar atrophy (multiple system atrophy).Acta Neuropathol. 82, 488–493.
Kitagawa K., Matsumoto M., Niinobe M., Mikoshiba K., Hata R., Ueda H., Handa N., Fukunaga R., Isaka Y., Kimura K., and Kamada T. (1989) Microtubule-associated protein 2 as a sensitive marker for cerebral ischemic damage-immunohistochemical investigation of dendritic damage.Neuroscience 31, 401–411.
Kosik K. S., Duffy L. K., Dowling M. M., Abraham C. R., McCluskey A., and Selkoe D. J. (1984) Microtubule-associated protein 2: monoclonal antibodies demonstrate the selective incorporation of certain epitopes into Alzheimer neurofibrillary tangles.Proc. Natl. Acad. Sci. USA 81, 7941–7945.
Lopresti P., Szuchet S., Papasozomenos S. C., Zinkowski R. P., and Binder L. I. (1995) Functional implications for the microtubule-associated protein tau localized in oligodendrocytes.Proc. Natl. Acad. Sci. USA 92, 10,369–10,373.
Martin S. M., Landel H. B., Lansing A. J., and Vijayan V. K. (1991) Immunocytochemical double labeling of glial fibrillary acidic protein and transferrin permits the identification of astrocytes and oligodendrocytes in the rat brain.J. Neuropathol. Exp. Neurol. 50, 161–170.
Neve R. L., Selkoe D., Kurnit D. M., and Kosik K. (1986) A cDNA for a human microtubule associated protein 2 epitope in the Alzheimer neurofibrillary tangle.Mol. Brain Res. 1, 193–196.
Nishimura M., Namba Y., Ikeda K., and Oda M. (1992) Glial fibrillary tangles with straight tubules in the brains of patients with progressive supranuclear palsy.Neurosci. Lett. 143, 35–38.
Papasozomenos S. C. and Binder L. I. (1987) Phosphorylation determines two distinct species of tau in the central nervous system.Cell Motil. Cytoskeleton 8, 210–226.
Papp M. I. and Lantos P. L. (1994) The distribution of oligodendroglial inclusions in multiple system atrophy and its relevance to clinical symptomatology.Brain 117, 235–243.
Pirollet F., Margolis R. L., and Job D. (1992) Ca(2+)-calmodulin regulated effectors of microtubule stability in neuronal tissues.Biochim. Biophys. Acta 1160, 113–119.
Schwab C., Bondada V., Sparks D. L., Cahan L. D., and Geddes J. W. (1994) Postmortem changes in the levels and localization of microtubule associated proteins (tau, MAP2 and MAP1B) in the rat and human hippocampus.Hippocampus 4, 210–225.
Spokes E. G. S. and Koch D. J. (1978) Postmortem stability of dopamine, glutaate decarboxylase and choline acetyltransferase in the mouse brain under conditions simulating the handling of human autopsy material.J. Neurochem. 31, 381–383.
Trojanowski J. Q., Schuck T., Schmidt M. L., and Lee V. M-Y. (1989a) Distribution of phosphate-independent MAP2 epitopes revealed with monoclonal antibodies in microwave-denatured human nervous system tissues.J. Neurosci. Methods 29, 171–180.
Trojanowski J. Q., Schuck T., Schmidt M. L., and Lee V. M-Y. (1989b) Distribution of tau proteins in the normal human central and peripheral nervous system.J. Histochem. Cytochem. 37, 209–215.
Tucker R. P. (1990) The roles of microtubule-associated proteins in brain morphogenesis: a review.Brain Res. Rev. 15, 101–120.
Tucker R. P., Binder L. I., and Matus A. I. (1988) Neuronal microtubule-associated proteins in the embryonic avian spinal cord.J. Comp. Neurol. 271, 44–55.
Ulloa L., Montejo de Garcini E., Gomez-Ramos P., Moran M. A., and Avila J. (1994) Microtubule-associated protein MAP1B showing a fetal phosphorylation pattern is present in sites of neurofibrillary degeneration in brains of Alzheimer's disease patients.Mol. Brain Res. 26, 113–122.
Yamada M., Wada Y., Tsukagoshi H., Otomo E-I., and Hayakawa M. (1988) A quantitative Golgi study of basal dendrites of hippocampal CA1 pyramidal cells in senile dementia of Alzhemier type.J. Neurol. Neurosurg. Psych. 51, 1088–1090.
Yamada T., McGeer P. L., and McGeer E. G. (1992) Appearance of paired nucleated, Tau-positive glia in patients with progressive supranuclear palsy brain tissue.Neurosci. Lett. 135, 99–102.
Yamamoto K., Morimoto K., and Yanagihara T. (1986) Cerebral ischemia in the gerbil: transmission electron microscopic and immunoelectron microscopic investigation.Brain Res. 384, 1–10.
Yanagihara T., Brengman J. M., and Mushynski W. E. (1990) Differential vulnerability of microtubule components in cerebral ischemia.Acta Neuropathol. 80, 499–505.
Yen S. H., Dickson D. W., Crowe A., Butler M., and Shelanski M. L. (1987) Alzheimer's neurofibrillary tangles contain unique epitopes in common with heat-stable microtubule-associated proteins Tau and MAP2.Am. J. Pathol. 126, 81–91.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Irving, E.A., McCulloch, J. & Dewar, D. The effect of postmortem delay on the distribution of microtubule-associated proteins τ, MAP2, and MAP5 in the rat. Molecular and Chemical Neuropathology 30, 253–271 (1997). https://doi.org/10.1007/BF02815102
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02815102