Abstract
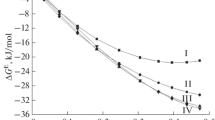
The rate of the chemical potential change of oxygen in a liquid PbO−SiO2 binary solution, with SiO2 contents of 10, 20, and 30 mol pct, and in pure PbO, has been measured at temperatures of 900°, 950°, 1000°, 1050°, and 1100°C. The rate increased with temperature according to the Arrhenius type relation and decreased with the increase of the silica content. It is suggested that the rate-controlling step is the counter diffusion rate of Pb2+ and Pb4+ ions, which are considered to be the most easily movable ions in the PbO−SiO2 solution. The relation between the rate of oxygen chemical potential change and the electrical conductivity is also discussed for the liquid PbO−SiO2 system.
Similar content being viewed by others
References
R. Littlewood:Trans. TMS-AIME, 1965, vol. 233, p. 772.
K. Goto, M. Sasabe, and M. Someno:Trans. TMS-AIME, 1968, vol. 242, p. 1757.
P. Herasymenko:Trans. Faraday Soc., 1938, vol. 34, p. 1245.
P. Herasymenko:Arch. Eisenhuettenw., 1940, vol. 13, p. 369.
Electrical Conductivity of Liquid Slag. A collection of papers presented at the Third Symposium on Liquid Iron and Slag, 1968, Tokyo, Iron and Steel Institutitute of Japan.
M. Ito and T. Yanagase:Denki-Kagaku, 1958, vol. 26, p. 363.
H. Flood and W. J. Knapp:J. Am. Ceram. Soc., 1963, vol. 46, p. 61.
G. W. Toop and C. S. Samis:Trans. TMS-AIME, 1962, vol. 224, p. 818.
C. R. Masson:Proc. Royal Soc., 1965, vol. 287, p. 201.
F. D. Richardson and L. E. Webb:Bull. Inst. Mining Metal, 1955, vol. 584, p. 529.
H. Saito, K. Goto, and M. Someno:Tetsu-to-Hagane, 1969, vol. 55, p. 539.
K. Goto and Y. Matsushita:Tetsu-to-Hagane, 1965, vol. 51, p. 1240.
J. Crank:The Mathematic of Diffusion, p. 42, Oxford Press, 1964.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sasabe, M., Goto, K. & Someno, M. Rate of change of oxygen chemical potential in liquid PbO−SiO2 binary solution. Metall Trans 1, 811–817 (1970). https://doi.org/10.1007/BF02811759
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02811759