Abstract
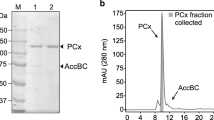
Carnitine acetyltransferase was purified from the citric acid producingA. niger mycelium with a protein band showing a relative molecular weight of 77,000 and a pH optimum of 7.3. TheK m values for the purified enzyme for acetyl-CoA and for carnitine were 0.1 mM and 1 mM, respectively. Carnitine acetyltransferase was located both in the mitochondria and in the cytosol. Both mitochondrial and cytosolic enzyme were purified using ammonium sulfate precipitation, Mono Q and Superose 12 separation. Regarding the localization, except for maximum velocity, there were no differences observed in substrate specificity and inhibition. Inhibition of the enzyme with micromolar concentrations of Cu2+ could contribute to a greater citric acid biosynthesis. Carnitine acetyltransferase can be considered as an enzyme necessary for the transport of acetyl groups through mitochondrial membrane in both directions.
Similar content being viewed by others
References
Srere, P. A. (1965),Nature 205, 766–770.
Bloisi, W., Colombo, I., Caravaglia, B., Giardini R., and Finocchiaro, G.(1990),Eur. J. Biochem. 189, 539–546.
Ratledge, C. and Gilbert, S. C. (1985),FEMS Microbiol. Lett. 27, 88–91.
Kozulic, B., Kappeli, O., Meussdoerffer, F., and Fiechter, A. (1987),Eur. J. Biochem. 168, 245–250.
Kozulic, B., Mosbach, K., and Meussdoerffer, F. (1988),Biochem. J. 253, 845–849.
Ueda, M., Tanaka, A., and Fukui, S. (1984),Eur. J. Biochem. 138, 445–449.
Ueda, M., Tanaka, A., and Fukui, S. (1985),Arch. Microbiol. 141, 29–31.
Pfitzner, A., Kubicek, C. P., and Roehr, M. (1987),Arch. Microbiol. 147, 88–91.
Jernejc, K., Perdih, A., and Cimerman, A. (1991),Appl. Microbiol. Biotechnol. 36, 92–95.
Jernejc, K., Cimerman, A., and Perdih, A. (1982),Eur. J. Appl. Microbiol. Biotechnol. 14, 29–33.
Kohlhaw, G. B. and Tan-Wilson, A. (1977),J. Bacteriol. 129, 1159–1161.
Watson, K. and Smith, J. E. (1968),J. Bacteriol. 96, 1546–1550.
Bradford, M. M. (1976),Anal. Biochem. 72, 148–254.
Laemmli, U. K. (1970),Nature 227, 680–685.
Claus, R., Kappeli, O., and Fiechter, A. (1982),Anal. Biochem. 127, 376–379.
Kispal, G., Cseko, J., Alkonyi, I., and Sandor, A. (1991),Biochem. Biophys. Acta 1085, 217–222.
Colluci, W. J. and Grandour, R. D. (1988),Bioorg. Chem. 16, 307–334.
Ueda, M., Tanaka, A., and Fukui, S. (1982),Eur. J. Biochem. 124, 205–210.
Mijazawa, S., Ozasa, H., Furuta, S., Osumi, T., and Hashimoto, T. (1983),J. Biochem. 93, 439–451.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jernejc, K., Legiša, M. Purification and properties of carnitine acetyltransferase from citric acid producingAspergillus niger . Appl Biochem Biotechnol 60, 151–158 (1996). https://doi.org/10.1007/BF02788069
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02788069