Abstract
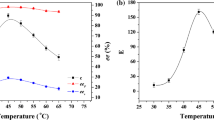
Stereoselective esterification of(R, S) 2-octanol with octanoic acid catalyzed byCandida Sp lipase (CSL) was carried out in cyclohexane. We have studied the effects of factors, such as temperature and the microenvironment of lipase, on this reaction. The results showed that CSL favoredR enantiomer of(R, S) 2-octanol, and that the esterification activity and stereoselectivity of the lipase were dependent on these factors. The higher the temperature, the greater the esterification activity of CSL. A slight increase in stereoselectivity can be seen with temperature decrease. The optimal range of pH value for this reaction was 4.9–6.2. When the salt concentration was between 0 and 0.05 mol/L, CSL showed high activity. The salt concentration in the reaction system and the pH value at which CSL powder was prepared from the aqueous solution had no evident effect on the stereoselectivity of CSL.
The optimal range of the water content in the reaction system was 0.4–1.6%. The esterification activity and the stereoselectivity of CSL were enhanced 1.4-fold and 2.0-fold, respectively, by immediately removing the produced water. (S) 2-octanol with 95.2% enantiomeric excess (ee) was prepared. Based on these results, we have discussed why that all these factors affected this reaction.
Similar content being viewed by others
References
Klibanov, A. M. (1990),Ace. Chem. Res. 23, 114.
Zaks, A. and Russell, A. J. (1988),J. Biotechnol. 8, 259.
Klibanov, A. M. (1989),Trends Biochem. Sci. 14, 141.
Hailing, P. J. (1987),Biotechnol. Adv. 5, 47.
Dordick, J. S. (1989),Enzyme Microb. Technol. 11, 194.
Langrand, G., Secchi, M., Buono, G., Baratti, J., and Triantaphylides, C. (1985),Tetrahedron Lett. 26, 1857.
Langrand, G., Baratti, J., Buono, G., and Triantaphylides, C. (1986),Tetrahedron Lett. 27, 29.
Koshiro, S., Sonomoto, K., and Tanaka, A. J. (1985),Biotechnology 2, 47.
Holmberg, E., Szmulik, P., Norin, J., and Hult, K. (1989),Biocatalysis 2, 217.
Partali, V., Sliwka, H. R., Anthonsen, T., and Synnove, L. J. (1992),Biocatalysis 6, 145.
Hiratake, J., Inagaki, M., Nishioka, T., and Oda, J. (1988),J. Org. Chem. 53, 6130.
Ader, U., Breitgoff, D., Klein, P., Laumen, K. E., and Schneider, M. P. (1989),Tetrahedron Lett. 30, 1793.
Bianchi, D., Cabri, W., Cesti, P., Francalanci, F., and Rama, F. (1988),Tetrahedron Lett. 29, 2455.
Cesti, P., Zaks, A., and Klibanov, A. M. (1985),Appl. Biochem. Biotechnol. 11, 401.
Holmberg, E. and Hult, K. (1991),Biotechnol. Lett. 13, 323.
Thomas, M. S. and Allan, C. O. (1987),Tetrahedron Lett. 28, 2091.
Sakurai, T., Russell, A. J., and Klibanov, A. M. (1988),J. Am. Chem. Soc. 110, 7236.
Kanasawud, P. and Phutrakul, S. (1993),Indian J. Chem. 32, 89.
Hirata, H., Yamashina, T., and Iida, I. (1991),J. Jpn. Oil Chem. Soc. 40(11), 9.
Chen, C. S., Fujimoto, Y., Girdaukas, G., and Sih, C. J. (1982),J. Am. Chem. Soc. 104, 7294.
Klibanov, A. M. (1986),Chemtech. June, 354.
Zale, S. E. (1986),Biochemistry 25, 5432.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yang, H., Cao, S.G., Han, S.P. et al. Studies on the factors that affect stereoselective esterification of (R, S) 2-octanol with octanoic acid catalyzed by lipase in organic solvent. Appl Biochem Biotechnol 59, 177–186 (1996). https://doi.org/10.1007/BF02787819
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02787819