Abstract
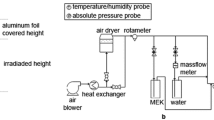
The photocatalytic oxidation of ethyl alcohol vapor in an annulus fluidized bed reactor of 0.06 m I.D. and 1.0 m long was examined. The TiO2 catalyst employed was prepared by the sol-gel method and was coated on the silica gel powder. The UV lamp was installed at the center of the bed as the light source. The effects of the initial concentration of ethyl alcohol, the power of UV-lamp, the photocatalysts with different preparation methods, and the superficial gas velocity on the reaction rate of ethyl alcohol decomposition were determined. It was found that, at 1.2 Umf of flow rate, about 80% of ethyl alcohol was decomposed with initial concentration of 10,000 ppmv and the increase of superficial gas velocity reduced the reaction rate significantly.
Similar content being viewed by others
References
Acker, H. D., French, R. H. and Chiang, Y M., “Comparisons of Hamaker Constants for Ceramic Systems with Intervening Vacuum or Water: from Force Laws and Physical Properties,”J. Coll. Interf. Sci.,179, 460 (1996).
Braun, A. M., Maurette, M. and Oliveros, E., “Photochemical Technology,” John Wiley & Sons, New York, 107 (1991).
Cheremishinoff, N. P. and Cheremisinoff, P. N., “Hydrodynamics of Gas-solids Fluidization,” Gulf Publishing Company, Houston, 137 (1984).
Fujishima, A., Rao, T. N. and Tryk, D. A., “Titanium Dioxide Photocatalysis,”J. Photochem Photobiol, C,1, 1 (2002).
Jung, S. M., Dupont, O. and Grange, P., “TiO2-SiO2 Mixed Oxide Modified with H2SO4. Characterization of the Microstructure of Metal Oxide and Sulfate,”Appl. Cotai, A:gen.,208, 393 (2001).
Karl, M. S. and Marinus, K., “Charge-carrier Dynamics in TiO2 Powder,”J. Phys. Chem.,94, 8222 (1990).
Kunii, D. and Levenspiel, O., “Fluidization Engineering,” Butterworth-Heinemann, Boston, 71 (1991).
Lee, Y D., Ahn, B. H., Lim, K. T., Jung, Y T., Lee, G. D. and Hong, S. S., “Photocatalytic Degradation of Trichloroethylene over Titanium Dioxide,”J. Korean Ind. Eng. Chem.,10, 1035 (1999).
Lim, T. H., Jeong, S. M., Kim, S. D. and Gyenis, J., “Photocatalytic Decomposition of NO by TiO2 Particle,”J. Photochem. Photobiol. A: Chem.,134, 209 (2000).
Lim, T. H. and Kim, S. D., “Photocatalytic Degradation of Trichloroethylene over TiO2/SiO2 in an Annulus Fluidized Bed Reactor,”Korean J. Chem. Eng.,19, 1072 (2002).
Matthews, R. W. and Mxevoy, S. R., “A Comparison of 254 nm and 350 nm Excitation of TiO2 in Simple Photocatalytic Reactors,”J. Photochem. Photobiol. A: Chem.,66, 355 (1992).
Nam, W., Kim, J. and Han, G, “Photocatalytic Oxidation of Methyl Orange in a Three-phase Fluidized Bed Reactor,”Chemosphere,47, 1019 (2002).
Nam, W. and Han, G, “A Photocatalytic Performance of TiO2 Photocatalyst Prepared by the Hydrothermal Method,”Korean J. Chem. Eng.,20, 180 (2003).
Raupp, G. B., Nico, J. A., Annangi, S., Changrani, R. and Annapragada, R., “Two-Flux Radiation-Filed Model for an Annular Packed-Bed Photocatalytic Oxidation Reactor,”AIChE J.,43, 792 (1997).
Ruthven, D. M., “Principles of Adsorption and Adsorption Processes,” John Wiley & Sons, New York, 5 (1984).
Sauer, M. L. and Ollis, D. F, “Photocatalyzed Oxidation of Ethanol and Acetaldehyde in Humidified Air,”J. Catal,158, 570 (1996).
Schiavello, M., “Wiley Series in Photoscience and Photoengineering: Heterogeneous Photocatalysis,” Wiley, 169 (1999).
Skoog D. A, Holler F. J. and Nieman T. A, “Principles of Instrumental Analysis,” Saunders colleges publishing, Philadelphia, 404 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, MJ., Nam, W. & Han, G.Y. Photocatalytic oxidation of ethyl alcohol in an annulus fluidized bed reactor. Korean J. Chem. Eng. 21, 721–725 (2004). https://doi.org/10.1007/BF02705511
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02705511