Abstract
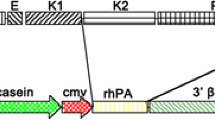
We have recently described the production of large amounts (≤W5 grams per litre) of enzymatically active human alpha 1 antitrypsin in the milk of transgenic sheep (Wrightet al., 1991). Here, we describe in more detail the expression of the human protein in the milk of these animals throughout the lactation period. Human alpha 1 antitrypsin is also found at much lower levels in the plasma of transgenic ewes before, during and after lactation. It is also detected in male plasma at very low levels.
We have previously shown human alpha 1 antitrypsin purified from transgenic sheep milk to be indistinguishable from commercially available human plasma derived alpha 1 antitrypsin in terms of gross sugar content andin vitro activity. Here we extend this comparison to more detailed analyses of glycosylation state, amino-terminal sequence, pI value, and molecular weight determination by mass spectrometry.
Similar content being viewed by others
Abbreviations
- hα1AT:
-
human alpha 1 antitrypsin
- hpα1AT:
-
plasma derived human alpha 1 antitrypsin
- BLG:
-
betalactoglobulin
- LDMS:
-
laser desorption mass spectrometry
References
Archibald AL, McClenaghan M, Hornsey V, Simons JP and Clark AJ (1990) High-level expression of biologically active human α1-antitrypsin in the milk of transgenic mice. Proc. Natl. Acad. Sci. USA 87: 5178–5182.
Bishop JO and Al-Shawi R (1989) Gene expression in transgenic animals. In: Hill WG and Mackay TFC (Eds) Evolution and Animal Breeding, CAB International, pp. 249–260.
Buhler TA, Bruyere T, Went DE, Strazinger G and Burki K (1990) Rabbit β-casein promoter directs secretion of human interleukin-2 into the milk of transgenic rabbits. Bio/Technology 8: 140–143.
Casolaro MA, Fells G, Wewers M, Pierce JE, Ogushi F, Hubbard R, Sellers S, Forstrom J, Lyons D, Kawasaki G and Crystal RG (1987) Augmentation of lung antineutrophil elastase capacity with recombinant human α-1-antitrypsin. J. Appl. Physiol. 63: 2015–2023.
Clark AJ, Bessos H, Bishop JO, Brown P, Lathe R, McClenaghan M, Prowse C, Simons JP, Whitelawn CBA, and Wilmut I (1989) Expression of human anti-hemophilic factor IX in the milk of transgenic sheep. Bio/Technology 7: 487–492.
Courtney M, Buchwalder A, Tessier L-H, Jaye M, Benavente A, Balland A, Kohli V, Lathe R, Tolstoshev P and Lecocq J-P (1984) High-level production of biologically active human α1-antitrypsin inEscherichia coli. Proc. Natl. Acad. Sci. USA 81: 669–673.
Crystal RG (1989) The α1-antitrypsin gene and its deficiency states. Trends Genet. 5: 411–417.
Crystal RG, Brantly ML, Hubbard RC, Curiel DT, States DJ and Holmes MD (1989) The alpha 1 antitrypsin gene and its mutations (1989) Chest. 95: 196–208.
Denman J, Hayes M, O'Day C, Edmunds T, Bartlett C, Hirani S, Ebert KM, Gordon K and McPherson JM (1991) Transgenic expression of a variant of human tissue-type plasminogen activator in goat milk: purification and characterisation of the recombinant enzyme. Bio/Technology 9: 839–843.
Ebert KM, Selgrath JP, DiTullio P, Denman J, Smith TE, Memon MA, Schindler JE, Monastersky GM, Vitale JA and Gordon K (1991) Transgenic production of a variant of human tissue plasminogen activator in goat milk: generation of transgenic goats and analysis of expression. Bio/Technology 9: 835–838.
Garver RI, Chytil A, Karlsson S, Fells GA, Brantly ML, Courtney M, Kantoff PW, Nienhuis AW, French Anderson W and Crystal RG (1987) Production of glycosylated physiologically normal human α1-antitrypsin by mouse fibroblasts modified by insertion of a human α1-antitrypsin cDNA using a retroviral vector. Proc. Natl. Acad. Sci. USA 84: 1050–1054.
Gordon K, Lee E, Vitale JA, Smith AE, Westphal H and Henninghausen L (1987) Production of human tissue plasminogen activator in transgenic mice. Bio/Technology 5: 1183–1187.
Hennighausen L, Ruiz L and Wall R (1990) Transgenic animals-production of foreign proteins in milk. Curr. Op. Biotech. 1: 74–78.
Hoylaerts M, Chuchana P, Verdonck P, Roelants P, Weyens A, Loriau R, DeWilde M and Bollen A (1987) Large scale purification and molecular characterisation of human recombinant α1-proteinase inhibitor produced in yeasts. (1989) J. Biotech. 5: 181–197.
Huber R and Carrell RW (1989) Implications of the three-dimensional structure of α1-antitrypsin for structure and function of serpins. Biochemistry 28: 8951–8966.
Jeppsson JO, Lilja H and Johansson M (1985) Isolation and characterisation of two minor fractions of α1-antitrypsin by high-performance liquid chromatographic chromatofocussing. J. Chromatog. 37: 175–177.
Krimpenfort P, Rademakers A, Eyestone W, van der Schans A, van den Broek S, Kooiman P, Kootwijk E, Platenburgh G, Pieper F, Strijker R and de Boer H (1991) Generation of transgenic dairy cattle usingin vitro embryo production. Bio/technology 9: 844–847.
Lovell-Badge RH (1985) New advances in the field. Nature 315: 628–629.
Meade H, Gates L, Lacy E and Lonberg N (1990) Bovine alphas1-casein gene sequences direct high level expression of active human urokinase in mouse milk. Bio/Technology 8: 443–446.
Palmiter RD, Brinster RL, Hammer RE, Trumbauer ME, Rosenfeld MG, Bimberg NC and Evans RM (1982) Dramatic growth of mice that develop from eggs microinjected with metallothionein-growth hormone fusion genes. Nature 300: 611–615.
Pittius CW, Hennighausen L, Lee E, Westphal H, Nichols E, Vitale J and Gordon K (1989) A milk protein gene promoter directs the expression of human tissue plasminogen activator cDNA to the mammary gland in transgenic mice. Proc. Natl. Acad. Sci USA 85: 5874–5878.
Wright G, Carver A, Cottom D, Reeves A, Scott A, Simons P, Wimut I, Garner I and Colman A (1991) High level expression of active human alpha-1-antitrypsin in the milk of transgenic sheep. Bio/Technology 9: 830–834.
Yu SH, Deen KC, Lee E, Hennighausen L, Sweet RW, Rosenberg M and Westphal H (1989) Functional human CD4 protein produced in milk of transgenic mice. Mol. Biol. Med. 6: 255–261.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Carver, A., Wright, G., Cottom, D. et al. Expression of human α1 antitrypsin in transgenic sheep. Cytotechnology 9, 77–84 (1992). https://doi.org/10.1007/BF02521734
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02521734