Abstract
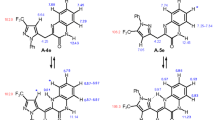
Reaction of 5,5-dimethyl-2-formylcyclohexane-1,3-dione with 4-methyl-, 4-benzoyl-, and 4-nitro-1,2-diaminobenzenes gave the corresponding 2-(2-amino-4-methylphenylaminomethylene)-, 2-(2-amino-5-benzoylphenylaminomethylene)-, and 2-(2-amino-5-nitrophenylaminomethylene)-5,5-dimethylcyclohexane-1,3-diones. When treated with hydrochloric acid, they cyclize to 7-methyl-, 8-benzoyl-, and 8-nitro-3,3-dimethyl-2,3,4,5-tetrahydro-1H-dibenzo[b,e][1,4]diazepinon hydrochlorides. Under hydrolytic conditions the salts of 3,3,7-trimethyl-2,3,4,5-tetrahydro-1H-dibenzo[b,e][1,4]diazepinone and 3,3-dimethyl-2,3,4,5-tetrahydro-1H-dibenzo[b,e][1,4]diazepinone undergo the C11−N10 bond cleavage to give N-(2-aminophenyl)- and N-(2-amino-5-methylphenyl)-substituted 3-amino-2-formyl-5,5-dimethylcyclohex-2-enones. Ring opening of the hydrochlorides of 8-benzoyl-, and 3,3-dimethyl-8-nitro-2,3,4,5-tetrahydro-1H-dibenzo[b,e][1,4]diazepinones occurs at the C−N5 bond and gives the starting enamines.
Similar content being viewed by others
References
A. Ya. Strakov, M. V. Petrova, N. N. Tonkikh, A. I. Gurkovskii, Yu. Popelis, G. P. Kreishman, and S. V. Belyakov,Khim. Geterotsikl. Soedin., No. 3, 379 (1997).
A. Ya. Strakov, M. V. Petrova, A. Dishs, I. A. Strakova, and O. F. Lakhvich,Khim. Geterotsikl. Soedin., No. 3, 336 (1995).
M. T. Shul'tsa and A. Ya. Strakov,Izv. Akad. Nauk Latv. SSR., Ser. Khim., No. 3, 329 (1971).
Additional information
Riga Technical University, Riga LV-1658, Latvia; Translated from Khimiya Geterotsiklicheskikh Soedinenii, N. 5, pp. 696–700, May, 1999.
Rights and permissions
About this article
Cite this article
Gurkovskii, A.I., Tonkikh, N.N., Petrova, M.V. et al. 3-acyl-1,5-benzodiazepines in the reactions of 5,5-dimethyl-2-formylcyclohexane-1,3-dione with certain 1,2-diaminobenzenes. Chem Heterocycl Compd 35, 625–629 (1999). https://doi.org/10.1007/BF02324651
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02324651