Abstract
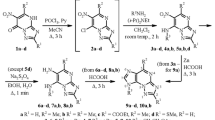
A series of 6-(ω-aroylthio)purines, which have not been described in the literature, has been obtained by the reaction of 6-purinethione with ω-chlorovalerophenone and its substituted derivatives. Some properties of the compounds synthesized have been studied, viz. reaction at the carbonyl group, methylation, and hydrolysis.
Similar content being viewed by others
References
E. V. Kochergina, A. N. Krasovskii, N. A. Klyuev, and V. V. Zamurenko,Farmats. Zh., No. 6, 33 (1981).
E. V. Aleksandrova, A. N. Krasovskii, and N. A. Klyuev,Abstracts of the III conference on Organic Chemistry and Technology, Moscow (1982), p. 86.
P. M. Kochergin, M. Yu. Gromov, E. V. Aleksandrova, and S. Ya. Skachilova,Khim. Geterotsikl. Soedin., No. 11, 1548 (1993).
H. Gunther,Introduction to a Course on NMR Spectroscopy [Russian translation], Mir, Moscow (1984), p. 478.
M. V. Rubtsov and A. G. Baichinov,Synthetic Pharmaceutical Chemistry Prepations [in Russian], Meditsina, Moscow (1971), p. 280.
H. Normant and O. Varenix,Compt. Rend.,231, 703 (1950).
N. K. Zheltukhin, I. A. Tubasheva, E. Ya. Borisova, and S. Ya. Skachilova,Express Information. Series “Foremost Experience in the Pharmaceutical Chemistry Industry”, TsBNTI Medprom, Moscow (1986), p. 6.
E. Fischer, Chem. Ber.,31, 114 (1898).
Aldrich Calalog. Handbook of Fine Chemicals, 1992–1993, p. 719.
G. B. Elion, E. Burgi, and G. H. Hitching,J. Am. Chem. Soc.,74, 411 (1952).
Additional information
All-Russian Scientific Center for the Safety of Biologically Active Substances, Staraya Kupavna, Moscow region 142450. Zaporozhe State Medical University, Zaporozhe 330074, Ukraine. Center for Drug Chemistry, All-Russian Pharmaceutical Chemistry Research Institute, Moscow 119815. Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1407–1411, October, 1999.
Rights and permissions
About this article
Cite this article
Gromov, M.Y., Skachilova, S.Y., Aleksandrova, E.V. et al. Synthesis and some properties of 6-(ω-aroylbutylthio)purines. Chem Heterocycl Compd 35, 1225–1229 (1999). https://doi.org/10.1007/BF02323383
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02323383