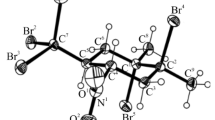
Abstract
The reaction of 6-bromo-7-cyclopropyl-1,4-benzodioxane with N2O4 in methylene chloride does not affect the cyclopropane ring and forms the nitrodebrominated product (ipso-substitution). The same reaction of 6-nitro- and 5,6-dinitro-7-cyclopropyl-1,4-benzodioxanes produces only the products with an modified threemembered ring. The difference in the reaction paths of the studied cyclopropylbenzodioxanes with N2O4 is explained by the different ratio of substrate to one-electron oxidant, the nitrosyl cation.
Similar content being viewed by others
References
S. S. Mochalov, Ya. I. Kuz'min, A. N. Fedotov, E. V. Trofimova, R. A. Gazzaeva, Yu. S. Shabarov, and N. S. Zefirov,Zh. Org. Khim.,34, 1379 (1998).
M. M. Smirnova, A. V. Geiderikh, S. S. Mochalov, and Yu. S. Shabarov,Zh. Org. Khim.,24, 1189 (1988).
S. S. Mochalov, V. N. Atanov, and N. S. Zefirov,Khim. Geterotsikl. Soedin., No. 5, 618 (1998).
E. K. Kim, T. M. Bockman, and J. K. Kochi,J. Am. Chem. Soc.,115, 3091 (1993).
R. G. Clewley, A. Fischer, and G. N. Henderson,Can. J. Chem.,67, 1472 (1989).
J. Feng, X. Zheng and M. C. Zerner,J. Org. Chem.,51, 4531 (1986).
Yu. S. Shabarov, S. S. Mochalov, and S. A. Ermishkina,Dokl. Akad. Nauk SSSR,211, 1135 (1973).
S. S. Mochalov, S. A. Ermishkina, S. K. Erglis, and Yu. S. Shabarov,Zh. Org. Khim.,11, 1409 (1975).
S. S. Mochalov, D. V. Kosynkin, I. D. Yudin, K. A. Zavodskikh, Yu. S. Shabarov, and N. S. Zefirov,Khim. Geterotsikl. Soedin., No. 4, 472 (1994).
Additional information
M. V. Lomonosov Moscow State University, Moscow 119899
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 3, pp. 324–329, March, 1999.
Rights and permissions
About this article
Cite this article
Mochalov, S.S., Gazzaeva, R.A., Atanov, V.N. et al. Reaction of substituted 7-cyclopropyl-1,4-benzodioxanes with dinitrogen tetraoxide. Chem Heterocycl Compd 35, 281–285 (1999). https://doi.org/10.1007/BF02259355
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02259355