Abstract
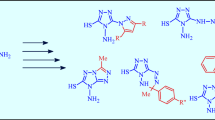
Addition of benzylmagnesium chloride to cyclopentylidenecyanoacetic ester gives 1-benzyl-1-(cyanocarbethoxymethyl)cyclopentane and this can be cyclized to 4-amino-3-carbethoxy-1,2-dihydrospiro-(naphthalene-2,1′-cyclopentane). Acylation of the obtained aminoester with carboxylic acid chlorides produces the corresponding amides (which can react with an excess of benzoyl chloride). It can also react with orthoformic ester. In both cases the product can then react with hydrazine hydrate. Treatment of the same aminoester with caprolactam gives a tetrahydrospiro(benzo[h]quinazoline-5,1′-cyclopentane) derivative.
Similar content being viewed by others
References
R. A. Kuroyan, A. I. Markosyan, A. Sh. Oganisyan, and M. G. Oganisyan, Arm. Khim. Zh.,42, No. 8, 527 (1989).
A. I. Markosyan, S. V. Dilanyan, R. A. Kuroyan, A. A. Chachoyan, and B. T. Garibdzhanyan, Khim.-Farm. Zh.,29, No. 4, 32 (1995).
A. I. Markosyan, R. A. Kuroyan, S. V. Dilanyan, and L. A. Shirkhanyan, Khim. Geterotsikl. Soedin., No. 4, 530 (1996).
Additional information
A. L. Mndzhoyan Institute of Fine Organic Chemistry, National Academy of Sciences of the Republic of Armenia, Yerevan 375014. Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 6, pp. 820–823, June, 1998.
Rights and permissions
About this article
Cite this article
Markosyan, A.I., Kuroyan, R.A. & Dilanyan, S.V. Synthesis and reactions of 4-amino-3-carbethoxy-1,2-dihydrospiro(naphthalene-2,1′-cyclopentane). Chem Heterocycl Compd 34, 716–718 (1998). https://doi.org/10.1007/BF02252282
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02252282