Abstract
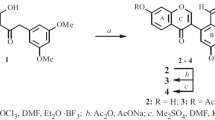
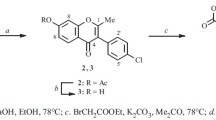
Reaction of Boc-protected amino acids with 3-phenoxy-2-trifluoromethylchromones gave new, previously unknown aminoacyl derivatives.
Similar content being viewed by others
References
M. S. Frasinyuk, A. V. Turov, and V. P. Khilya,Khim. Geterotsikl. Soedin., No. 8, 1072 (1998).
V. V. Arkhipov, M. N. Smirnov, and V. P. Khilya,Khim. Geterotsikl. Soedin., No. 5, 598 (1997).
S. A. Vasil'ev, M. S. Luk'yanchikov, G. I. Molchanov, V. D. Turubarov, and V. P. Khilya,Khim.-Farm. Zh., No. 7, 34 (1991).
A. S. Ogorodniichuk, S. A. Vasil'ev, I. N. Lekhan, V. P. Khilya, and V. V. Shilin, Bioorg.Khim.,17, 1005 (1991).
N. V. Gorbulenko, O. N. Gaiduk, V. P. Khilya, A. S. Ogorodniichuk, and V. V. Shilin,Dokl. Akad. Nauk UkrSSR, Ser. B, No. 9, 149 (1991).
Additional information
T. G. Shevchenko Kiev University, Kiev 252033, Ukraine. Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 6, pp. 744–748, June, 1999.
Rights and permissions
About this article
Cite this article
Garazd, M.M., Arkhipov, V.V., Proskurka, N.K. et al. Chemistry of heteroanalogs of isoflavones. 23. Synthesis of aminoacyl derivatives of 3-phenoxychromone. Chem Heterocycl Compd 35, 658–662 (1999). https://doi.org/10.1007/BF02251621
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02251621