Abstract
Objectives
To observe the granulosa-luteal cell subpopulations presented within follicular aspirates concerning duration of the follicular phase and the type of IVF protocol.
Design
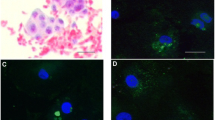
Cells were obtained from dominant follicles of 40 women with natural IVF-ET cycles, in which preovulatory hCG was given when the follicle was mature, and from 40 follicles of 32 women with hMG and hCG stimulated IVF-ET cycles. Granulosa-luteal cell subpopulations were observed by computerized image analysis in which hCG was localized using immunoperoxidase staining.
Results
(1) The nonluteinized granulosa cells from natural developing follicles were larger than those from stimulated ones regardless of the follicular phase duration. (2) The size of each luteinized cell subpopulations was influenced neither by the two IVF protocols nor by the follicular phase duration. (3) The hCG stained cells from natural developing follicles were larger than the ones from stimulated follicles and their relative number in aspirates was higher. Cell areas and distribution were not influenced by the duration of follicular phase. (4) In stimulated conditions. hCG stained cells became larger if follicular phase was longer.
Conclusions
Duration of the follicular phase influences the immunocytochemical hCG localization and the morphometric characteristics of granulosa-luteal cell sub-populations presented within natural developing follicles and stimulated ones.
Similar content being viewed by others
References
Corner GW: On the origin of the corpus luteum of the sow from both granulosa and theca interna. Am J Anat 1919;26:177–183
Fritz MA, Fritz TA: The functional Microscopic anatomy of the corpus luteum: the “small cell”-“large cell” controversy. Clin Obstet Gynecol 1991;24:144–156
Ohara A, Mori T, Taii S, Ban C, Narimoto K: Functional differentiation in steroidogenesis of two types of luteal cells isolated from mature human corpora lutea of menstrual cycle. J Clin Endocrinol Metab 1987;65:1192–1200
Whitman GF, Boldt JP, Martinez JE, Pantazis CG: Flow cytometric analysis of induced human graafian follicles. I. Demonstration and sorting of two luteinized cell populations. Fertil Sterril 1991;56:256–264
Hartshorne GM: Subpopulation of granulosa cells within the human ovarian follicle. J Reprod Fertil 1990;89:773–782
Gersak K: Subpopulations of human granulosa-luteal cells in natural and stimulated IVF cycles. J Assist Reprod Gen 1995;12:Suppl:87S
Gersak K, Meden-Vrtovec H, Tomazevic T: The effect of gonadotrophin-releasing hormone agonist on follicular development in patients with polycystic ovary syndrome in an in-vitro fertilization and embryo transfer programme. Hum Reprod 1994;9:1596–1599
World Health Organization: Laboratory Manual for the examination of human Semen and semen-cervical mucus interaction. Cambridge: Cambridge University Press, 1987
Paulson RJ, Sauer MV, Francis MM: In vitro fertilization in unstimulated cycles: a clinical trial using hCG for timing of follicle aspiration. Obstet Gynecol 1990;76:788–792
Veeck LL: Atlas of Human Oocyte and Early Conceptus. Human Oocytes at the time of Follicular Harvest. Baltimore: Williams and Wilkins, 1986:5–131
Gersak B: Presence of calcium in the vessel walls after end-to-end arterial anastomoses with polydioxanone and poly-propylene sutures in growing dogs. J Thorac Cardiovasc Surg 1993;106:587–591
Whitman GF, Luciano AA, Maier DB, Peluso JJ: Human chorionic gonadotropin localization and morphometric characterization of human granulosa-luteal cells obtained during in vitro fertilization cycles. Fertil Steril 1989;51:475–479
Baurne JA: Handbook of Immunoperoxidase Staining Methods. Santa Barbara: Dako Corporation, 1983:1–37
Robinson G: Immunohistochemistry.In Theory and Practice of Histological Techniques, JD Bancroft, A Stevens (eds). Edinburgh: Churchill Livingstone, 1982, pp 406–427
McNatty KP, Moore Smith D, Makris A, Osathanondh R, Ryan KJ: The microenvironment of the human antral follicle: interrelationships among the steroid levels in antral fluid, the population of granulosa cells, and the status of the oocyte in vivo and in vitro. J Clin Endocrinol Metabol 1979;49:851–860
Rajaniemi HJ, Ronnberg L, Kauppila A, Ylostalo P, Jalkanen M, Saastamoinen J, Selander K, Pystynen P, Vihko R: Luteinizing hormone receptors in human ovarian follicles and corpora lutea during menstrual cycle and pregnancy. J Clin Endocrinol Metab 1981;52:307–313
Yamoto M, Nakano R, Iwasaki M, Ikoma H, Furukawa K: Luteinizing hormone receptors in human ovarian follicles and corpora lutea during the menstrual cycle. Obstet Gynecol 1986;68:200–203
Amsterdam A, Nimrod A, Lamprecht SA, Burstein Y, Lindner HR: Internalization and degradation of receptor-bound hCH in granulosa cell cultures. Am J Physiol 1979;236:E129
Schwall RH, Erickson GF: Inhibition of synthesis of luteinizing hormone (LH) receptors by a down-regulation dose of LH. Endocrinol 1984;114:1114–1123
Lakkakorpi JT, Yang M, Rajaniemi HJ: Processing of the LH/hCG receptor and bound hormone in rat luteal cells after hCG-induced down-regulation as studied by a double immunofluorescence technique in conjunction with confocal laser scanning microscopy. J Histochem Cytochem 1994;42:727–732
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gersak, K., Tomazevic, T. Influence of follicular phase duration on human granulosa-luteal cell subpopulations in natural and stimulated IVF-ET cycles. J Assist Reprod Genet 12, 650–656 (1995). https://doi.org/10.1007/BF02212591
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02212591